Elements That Are Gas At Room Temp
Muz Play
Apr 05, 2025 · 5 min read
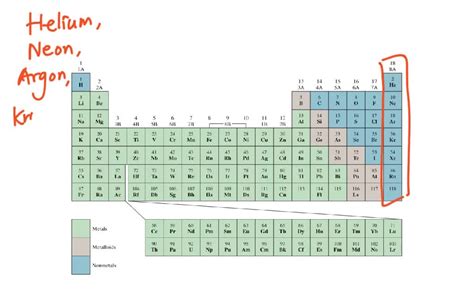
Table of Contents
Elements That Are Gases at Room Temperature: A Comprehensive Overview
The periodic table showcases a diverse array of elements, each with unique properties. Understanding these properties is crucial in various fields, from chemistry and physics to engineering and medicine. One fundamental property is the state of matter at room temperature (approximately 25°C or 77°F). This article delves into the fascinating world of elements that exist as gases under standard conditions, exploring their characteristics, applications, and importance.
The Noble Gases: Inert and Unreactive
The most well-known group of gaseous elements at room temperature are the noble gases. Located in Group 18 of the periodic table, these elements are characterized by their exceptional stability and low reactivity. This inertness stems from their complete valence electron shells, meaning they have little tendency to gain, lose, or share electrons with other atoms.
Helium (He)
Helium, the lightest noble gas, is renowned for its low density and inertness. It's extensively used in:
- Balloons and Airships: Its lightness makes it ideal for inflating balloons, providing buoyancy.
- Cryogenics: Helium's extremely low boiling point (-268.93°C) allows it to be used as a coolant for superconducting magnets in MRI machines and other scientific instruments.
- Welding and Leak Detection: Helium's inertness makes it suitable for shielding welds from atmospheric gases and detecting leaks in high-pressure systems.
- Breathing Mixtures: Helium-oxygen mixtures are used by deep-sea divers to reduce the risk of decompression sickness.
Neon (Ne)
Neon, known for its vibrant red-orange glow when electrically excited, finds primary applications in:
- Lighting: Neon signs, a ubiquitous symbol of advertising, utilize neon gas to produce bright, colorful lights. While many "neon" signs use other gases to achieve different colors, the name persists.
- Lasers: Neon is a component in several types of gas lasers, used in various applications including barcode scanners and laser surgery.
Argon (Ar)
Argon, the most abundant noble gas in the Earth's atmosphere, is largely inert and used in:
- Welding: Argon provides an inert atmosphere during welding, preventing oxidation and improving weld quality.
- Lighting: It's used in incandescent light bulbs to prevent filament oxidation, prolonging the bulb's lifespan.
- Winemaking: Argon is used to displace oxygen in wine bottles, preventing oxidation and preserving the wine's quality.
Krypton (Kr)
Krypton, though less abundant, boasts several niche applications:
- Lighting: Krypton gas emits a bright white light and is sometimes used in high-intensity lamps.
- Photography: Krypton flash lamps are used in high-speed photography.
Xenon (Xe)
Xenon, a heavier noble gas, has specialized uses:
- Lighting: Xenon arc lamps are used in high-intensity projectors and automotive headlights.
- Anesthesia: Certain xenon compounds are being investigated for use in anesthesia.
- Medical Imaging: Xenon is used in some medical imaging techniques.
Radon (Rn)
Radon, a radioactive noble gas, is naturally occurring and poses a health risk due to its radioactivity. It's not generally used in any applications but is important to understand and mitigate its presence in buildings due to its potential carcinogenic effects.
Other Gaseous Elements at Room Temperature
Beyond the noble gases, several other elements exist as gases under standard conditions.
Hydrogen (H₂)
Hydrogen, the lightest element, is a highly reactive gas and is found extensively in various compounds, particularly water. It's emerging as a significant energy carrier and has many industrial applications, including:
- Ammonia Production (Haber-Bosch Process): Hydrogen is a crucial component in producing ammonia, a vital fertilizer.
- Petroleum Refining: Hydrogen is used in various refining processes to improve the quality of fuels.
- Fuel Cells: Hydrogen fuel cells convert chemical energy directly into electrical energy, offering a clean energy alternative.
Nitrogen (N₂)
Nitrogen, making up approximately 78% of the Earth's atmosphere, is a relatively inert gas with several vital roles:
- Fertilizers: Nitrogen is a key component in fertilizers, essential for plant growth.
- Food Packaging: Nitrogen gas is used to preserve food and prevent oxidation.
- Electronics Manufacturing: Nitrogen is used in semiconductor manufacturing to provide an inert atmosphere.
Oxygen (O₂)
Oxygen, essential for respiration in most living organisms, is a highly reactive gas. Its applications are vast and include:
- Medical Applications: Oxygen therapy is used to treat respiratory conditions.
- Welding and Cutting: Oxygen is used in oxy-fuel welding and cutting processes.
- Industrial Processes: Oxygen is used in numerous industrial processes, including steelmaking.
Chlorine (Cl₂)
Chlorine, a highly reactive halogen, is a yellowish-green gas with several important applications, but also significant safety considerations due to its toxicity:
- Water Purification: Chlorine is widely used to disinfect water supplies, eliminating harmful bacteria and viruses.
- Industrial Bleaching: Chlorine is used as a bleaching agent in the paper and textile industries.
- Chemical Synthesis: Chlorine is a reactant in many chemical synthesis processes.
Fluorine (F₂)
Fluorine, the most reactive element, is a pale yellow gas. Its reactivity dictates specialized applications and handling requirements:
- Fluorocarbons: Fluorine is used in the production of fluorocarbons, which have applications as refrigerants and in other industrial processes.
- Dental Applications: Fluoride, a compound of fluorine, is added to toothpaste and water to help prevent tooth decay.
Safety Considerations
Working with gaseous elements requires careful attention to safety. Many of these gases are either highly reactive, toxic, or present a fire hazard. Proper ventilation, safety equipment (such as respirators and protective clothing), and appropriate handling procedures are paramount. Consult the appropriate safety data sheets (SDS) before handling any gaseous element.
Conclusion: The Importance of Gaseous Elements
Elements that are gases at room temperature play a crucial role in various aspects of modern life. From the inert noble gases used in lighting and medical applications to the highly reactive elements like oxygen and chlorine involved in vital biological and industrial processes, their properties and applications are diverse and impactful. Understanding their characteristics and handling them safely is essential for scientists, engineers, and anyone working with these substances. Further research and innovation continue to expand the applications of these essential elements, shaping our technological advancements and improving our quality of life. The study of these gaseous elements remains a vibrant and evolving field, with ongoing discoveries promising further breakthroughs in various sectors.
Latest Posts
Latest Posts
-
Volume Of A Solid With A Known Cross Section
Apr 05, 2025
-
Bacterial Growth Curves Experiment With Bacterial Growth
Apr 05, 2025
-
What Is A Secondary Alkyl Halide
Apr 05, 2025
-
Is Oil And Water A Heterogeneous Mixture
Apr 05, 2025
-
Difference Between Basic Science And Applied Science
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Elements That Are Gas At Room Temp . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
