Is Oil And Water A Heterogeneous Mixture
Muz Play
Apr 05, 2025 · 5 min read
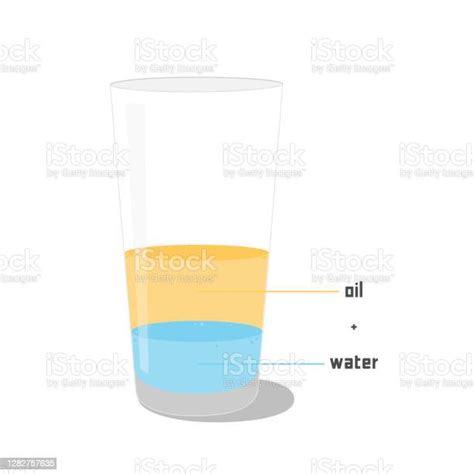
Table of Contents
Is Oil and Water a Heterogeneous Mixture? A Deep Dive into Mixtures and Separations
The question, "Is oil and water a heterogeneous mixture?" is a fundamental one in chemistry, touching upon concepts of mixtures, phases, and separation techniques. The answer is a resounding yes, and understanding why delves into the fascinating world of molecular interactions and physical properties. This article will explore this seemingly simple question in detail, examining the characteristics of heterogeneous mixtures, the properties of oil and water that lead to their incompatibility, and various methods used to separate these two substances.
Understanding Mixtures: Homogeneous vs. Heterogeneous
Before diving into the specifics of oil and water, let's establish a clear understanding of mixtures. A mixture is a substance composed of two or more components that are not chemically bonded. Crucially, the components retain their individual chemical properties within the mixture. Mixtures are categorized into two main types:
Homogeneous Mixtures: A Uniform Blend
In a homogeneous mixture, the components are uniformly distributed throughout the mixture. This means that the composition is the same throughout the sample, regardless of where you take a sample from. Think of saltwater: the salt is dissolved uniformly in the water, and you wouldn't be able to visually distinguish salt crystals from the water itself. Other examples include air (a mixture of gases) and sugar dissolved in water. At the microscopic level, the components are thoroughly intermixed.
Heterogeneous Mixtures: A Visible Difference
A heterogeneous mixture, on the other hand, exhibits visibly distinct components. The composition is not uniform, and different regions of the mixture may have different proportions of the components. A salad is a classic example: you can clearly see the individual pieces of lettuce, tomatoes, and cucumbers. Other examples include sand and water, or oil and water (our focus for this article). The components are not evenly dispersed, and the different phases are readily distinguishable.
The Immiscibility of Oil and Water: Polarity and Intermolecular Forces
The reason oil and water don't mix lies in their contrasting polarities and the resulting intermolecular forces. Polarity refers to the distribution of electrical charge within a molecule. Water (H₂O) is a highly polar molecule; it has a partial positive charge on the hydrogen atoms and a partial negative charge on the oxygen atom. This polarity leads to strong hydrogen bonds between water molecules, creating a cohesive network.
Oils, on the other hand, are typically nonpolar. They consist primarily of long hydrocarbon chains with relatively even charge distribution. The dominant intermolecular forces in oils are weak London dispersion forces.
The fundamental principle here is "like dissolves like." Polar substances tend to dissolve in polar solvents, while nonpolar substances dissolve in nonpolar solvents. Because oil is nonpolar and water is polar, their intermolecular forces are incompatible. The strong hydrogen bonds in water resist the intrusion of nonpolar oil molecules, resulting in the separation of the two liquids into distinct layers.
Visualizing the Heterogeneity
When you mix oil and water, you observe two distinct layers: the less dense oil floats on top of the denser water. This clear separation of phases is a hallmark of a heterogeneous mixture. No matter how vigorously you shake the mixture, the two liquids will eventually separate again. Microscopic observation would further confirm the heterogeneity, revealing distinct oil and water domains, rather than a uniform blend at the molecular level.
Methods for Separating Oil and Water: Harnessing Density Differences
The heterogeneous nature of oil and water allows for relatively simple separation techniques based on their differing densities. Here are a few common methods:
1. Decantation: A Simple Gravity-Based Separation
Decantation is the simplest method. It involves carefully pouring off the top layer (oil) after allowing the mixture to settle completely. This method is effective but not entirely complete, as some oil may remain emulsified within the water.
2. Separatory Funnel: Precise Phase Separation
A separatory funnel is a specialized piece of laboratory glassware used to separate immiscible liquids. The mixture is added to the funnel, allowed to settle, and then the stopcock at the bottom is carefully opened to drain the lower layer (water), leaving the oil behind. This method is more efficient than decantation and minimizes contamination.
3. Centrifugation: Accelerating Separation
Centrifugation uses centrifugal force to accelerate the separation process. The mixture is placed in a centrifuge tube and spun at high speed. The denser water is forced to the bottom, while the less dense oil forms a separate layer at the top. This method is particularly useful for separating small amounts of oil from water or for accelerating the separation process.
4. Other Advanced Techniques
More advanced techniques like distillation (exploiting differences in boiling points) can also achieve complete separation, although they are generally more complex and energy-intensive than the simpler methods mentioned above.
Applications and Real-World Examples
The heterogeneous nature of oil and water, and the consequent need for separation techniques, is relevant across various fields:
-
Environmental Science: Oil spills are a significant environmental concern. Understanding the immiscibility of oil and water, and employing effective separation methods, is crucial for cleaning up these spills and mitigating their environmental impact. Various techniques are used to remove oil from contaminated water bodies, including booms, skimmers, and bioremediation.
-
Chemical Engineering: Many industrial processes involve the separation of oil and water mixtures. The efficiency of these separation processes directly impacts the overall efficiency and cost-effectiveness of the industrial operation. Understanding the principles of phase separation and employing optimal methods are crucial for industrial applications.
-
Food Industry: In the food industry, oil and water emulsions are frequently encountered. The stability and separation of these emulsions impact the texture, appearance, and shelf-life of many food products. Understanding the interplay between oil and water is fundamental to developing food processing technologies.
Conclusion: Heterogeneity Confirmed
In conclusion, the answer to the question, "Is oil and water a heterogeneous mixture?" is an unequivocal yes. The immiscibility of oil and water, stemming from their differing polarities and intermolecular forces, results in the formation of two distinct phases that are readily observable. This heterogeneous nature allows for the application of various separation techniques, which are crucial in environmental remediation, industrial processes, and food production. Understanding this fundamental concept provides a strong basis for comprehending more complex mixture systems and separation technologies. The seemingly simple combination of oil and water serves as a potent illustration of fundamental chemical principles and their practical implications.
Latest Posts
Latest Posts
-
Anatomy And Physiology Nervous System Practice Test
Apr 06, 2025
-
Critical Temperature And Pressure Of Nitrogen
Apr 06, 2025
-
The Hormone Of Hyposecretion Is
Apr 06, 2025
-
How Was The First Industrial Revolution Different From The Second
Apr 06, 2025
-
Number Of Atoms In Face Centered Cubic
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is Oil And Water A Heterogeneous Mixture . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
