Example Of Stoichiometry In Real Life
Muz Play
Apr 04, 2025 · 6 min read
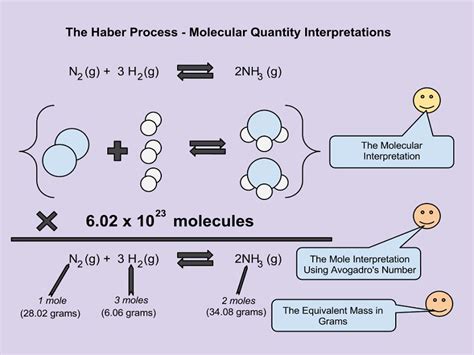
Table of Contents
Examples of Stoichiometry in Real Life: From Baking a Cake to Saving the Planet
Stoichiometry, at its core, is the science of measuring the quantities of reactants and products in chemical reactions. While it might sound like a dry academic subject, stoichiometry is a fundamental principle underpinning countless aspects of our daily lives. From the simplest tasks to complex industrial processes, understanding the quantitative relationships between substances is crucial. This article delves into various real-world examples of stoichiometry, demonstrating its practical applications and importance.
Stoichiometry in Everyday Life: Baking and Cooking
One of the easiest ways to understand stoichiometry is through baking. Baking a cake, for instance, involves a precise chemical reaction between ingredients. The recipe itself acts as a stoichiometric equation.
The Cake Recipe as a Stoichiometric Equation
Let's consider a simple cake recipe:
- 2 cups flour
- 1 cup sugar
- 1 teaspoon baking powder
- 1/2 teaspoon salt
- 4 eggs
- 1 cup milk
- 1 cup butter
This recipe is essentially a stoichiometric equation, defining the ratios of reactants (ingredients) needed to produce the desired product (cake). If you double the recipe, you double the amount of every ingredient; altering the ratio will likely lead to a less desirable outcome. Too much baking powder, for example, might result in a dense or dry cake.
Beyond Baking: Cooking and Everyday Chemistry
The principle extends beyond cakes to all forms of cooking. The correct proportions of ingredients in a sauce, the ratio of coffee grounds to water, even the perfect blend of spices – these all depend on an intuitive understanding of stoichiometry. Changing these ratios, even slightly, can significantly alter the taste and texture of the dish.
Stoichiometry in Industrial Processes: Manufacturing and Production
The industrial world heavily relies on stoichiometric calculations for efficient and safe production.
Fertilizer Production: Feeding the World
The Haber-Bosch process, a crucial method for producing ammonia (NH₃) for fertilizers, is a prime example. This process uses stoichiometric calculations to determine the optimal ratios of nitrogen (N₂) and hydrogen (H₂) gases needed to synthesize ammonia. The precise control of these ratios is vital for maximizing ammonia production while minimizing waste and energy consumption. The global food supply fundamentally relies on this stoichiometrically controlled process. Without precise calculations, the agricultural industry wouldn't be able to produce enough food to meet the world’s population needs.
Pharmaceutical Manufacturing: Precision Medicine
Pharmaceutical production demands exceptional precision. Stoichiometry is crucial in determining the exact amounts of reactants needed to synthesize drugs. The incorrect ratios of compounds can lead to impurities, reduced efficacy, and potentially harmful side effects. The entire process, from research and development to quality control, hinges on accurate stoichiometric calculations. The potency and safety of every medicine depend on these calculations.
Metal Refining: Extracting Pure Metals
Metal refining often involves chemical reactions where stoichiometry plays a critical role. For example, the extraction of pure metals from their ores often requires precise chemical reactions controlled by stoichiometric principles. Ensuring the correct ratios of reactants guarantees efficient metal extraction while minimizing waste and environmental impact.
Stoichiometry and Environmental Protection: Pollution Control and Sustainability
Stoichiometry isn't just about creating products; it's equally important in mitigating environmental issues.
Combustion Analysis: Monitoring Emissions
The combustion of fuels, whether in vehicles or power plants, involves stoichiometric relationships between fuel and oxygen. Understanding these relationships is crucial for controlling emissions. By meticulously measuring the ratios of fuel and oxygen, engineers can optimize combustion processes to minimize pollutants like carbon monoxide (CO) and unburned hydrocarbons. This is critical for reducing air pollution and addressing climate change.
Wastewater Treatment: Cleaning Up Our Water
Stoichiometry is crucial in wastewater treatment plants. The removal of pollutants, such as nitrates and phosphates, often involves controlled chemical reactions where the precise amounts of chemicals used are dictated by stoichiometric principles. Efficient treatment requires the precise calculation of reagent quantities to ensure effective removal of pollutants and safeguarding our water resources. Improper stoichiometric control can lead to ineffective treatment and environmental damage.
Carbon Capture and Storage: Combating Climate Change
Carbon capture and storage (CCS) technologies rely heavily on stoichiometric principles. These technologies involve capturing CO₂ from industrial emissions and storing it underground. The reactions involved in capturing and storing CO₂ require precise stoichiometric calculations to ensure efficiency and safety. Advancements in CCS technologies are essential in mitigating climate change, and stoichiometry plays a central role.
Stoichiometry in Advanced Technologies: Nanotechnology and Beyond
The applications of stoichiometry aren't limited to traditional industries; they are equally important in cutting-edge technologies.
Nanotechnology: Building at the Atomic Level
Nanotechnology involves manipulating materials at the atomic and molecular levels. Creating nanomaterials with specific properties often requires precise control over the stoichiometric ratios of constituent atoms or molecules. The creation of nanoparticles with desired characteristics relies on accurate stoichiometric calculations.
Biomedical Engineering: Targeted Drug Delivery
In biomedical engineering, stoichiometry plays a crucial role in developing targeted drug delivery systems. Precise control of drug concentration and release requires a detailed understanding of the stoichiometric relationships between different components in these systems. This ensures the safe and effective delivery of medication to specific locations in the body.
Energy Storage: Developing Efficient Batteries
The development of efficient energy storage systems, such as batteries, relies heavily on stoichiometric principles. The chemical reactions within batteries involve precise ratios of reactants to maximize energy storage and longevity. The performance of batteries directly depends on the careful balance and control of stoichiometric relationships. Innovation in battery technology is crucial in the transition to renewable energy sources, and stoichiometry is fundamental to this advancement.
Beyond the Basics: Advanced Stoichiometric Concepts
While the examples above highlight the fundamental applications of stoichiometry, more advanced concepts are also employed in various fields.
Limiting Reactants and Percent Yield
In many real-world scenarios, reactants are not present in stoichiometrically ideal ratios. One reactant will be completely consumed (the limiting reactant), while others remain in excess. Calculating the limiting reactant and the theoretical yield (based on the limiting reactant) is crucial in optimizing reactions and predicting the amount of product formed. The actual yield, compared to the theoretical yield, gives the percent yield, an important indicator of reaction efficiency.
Equilibrium Calculations
Chemical reactions often don't proceed to completion. Instead, they reach a state of equilibrium where the rates of forward and reverse reactions are equal. Stoichiometry is used in conjunction with equilibrium constants to predict the concentrations of reactants and products at equilibrium. This is particularly important in industrial processes where understanding equilibrium is key to optimizing production.
Thermodynamics and Stoichiometry
Thermodynamics relates to the energy changes associated with chemical reactions. Stoichiometric calculations are often combined with thermodynamic data to determine the feasibility and efficiency of a reaction, considering factors such as enthalpy and entropy changes.
Conclusion: The Ubiquitous Nature of Stoichiometry
Stoichiometry, although often perceived as a purely theoretical concept, is a fundamental principle with far-reaching practical implications. Its applications span numerous fields, from everyday cooking to advanced technologies, emphasizing its importance in understanding and controlling chemical processes. Whether it's baking a cake or developing life-saving medications, stoichiometry ensures efficiency, safety, and optimal outcomes. Understanding and mastering stoichiometry is essential for anyone seeking to delve deeper into the chemical world and its impact on our daily lives and the future of technology. The examples provided here only scratch the surface of the wide-ranging applications of this vital scientific principle. Its continuing relevance across various fields underscores its enduring importance in science and technology.
Latest Posts
Latest Posts
-
When Does Dna Replication Occur In A Eukaryotic Cell
Apr 04, 2025
-
What Was Shown By Both Redis And Pasteurs Experiments
Apr 04, 2025
-
The Elements In Group 1 Are Called The
Apr 04, 2025
-
Proof Of The Inverse Function Theorem
Apr 04, 2025
-
Nonmetal Halogen Family Atomic Mass 35
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Example Of Stoichiometry In Real Life . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
