Give The Iupac Name Of The Carboxylic Acid Below.
Muz Play
Apr 02, 2025 · 6 min read
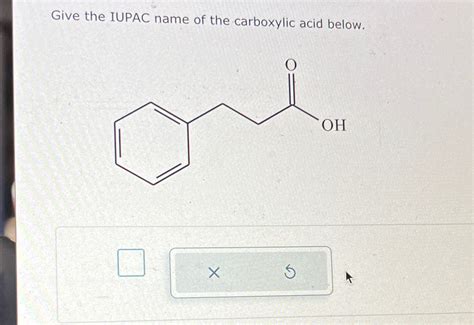
Table of Contents
Giving the IUPAC Name of Carboxylic Acids: A Comprehensive Guide
Carboxylic acids are a fundamental class of organic compounds characterized by the presence of a carboxyl group (-COOH) attached to an alkyl or aryl group. Naming these compounds correctly, using IUPAC nomenclature, is crucial for clear communication in organic chemistry. This comprehensive guide will walk you through the process, covering various complexities and providing examples to solidify your understanding.
Understanding the Basics of IUPAC Nomenclature for Carboxylic Acids
The IUPAC (International Union of Pure and Applied Chemistry) system provides a standardized method for naming organic compounds, ensuring universal understanding. For carboxylic acids, the naming follows a specific set of rules:
1. Identifying the Parent Chain:
The parent chain is the longest continuous carbon chain containing the carboxyl group (-COOH). This chain is numbered starting from the carbon atom of the carboxyl group. This carbon is always considered carbon number 1.
2. Naming the Parent Chain:
The parent chain's name is based on its number of carbon atoms. The suffix "-oic acid" is added to the alkane name corresponding to the number of carbons.
- 1 carbon: Methanoic acid
- 2 carbons: Ethanoic acid
- 3 carbons: Propanoic acid
- 4 carbons: Butanoic acid
- 5 carbons: Pentanoic acid
- and so on...
3. Incorporating Substituents:
Substituents, which are atoms or groups attached to the parent chain, are named and their positions are indicated using numbers. The numbering starts from the carboxyl carbon (always carbon 1). Substituents are listed alphabetically before the parent chain name.
4. Handling Multiple Carboxyl Groups:
If a molecule contains multiple carboxyl groups, the suffix "-dioic acid," "-trioic acid," etc., is used. The positions of the carboxyl groups are indicated using numbers.
5. Dealing with Unsaturation:
If the parent chain contains double or triple bonds, the position and type of unsaturation are indicated using the appropriate prefixes (e.g., "2-ene" for a double bond at position 2, "3-yne" for a triple bond at position 3). The numbering of the parent chain starts from the carboxyl carbon, even if this results in a lower number for the double or triple bond compared to other potential numbering schemes.
6. Cyclic Carboxylic Acids:
For cyclic carboxylic acids, the carboxyl group is considered part of the ring, and the numbering starts at the carboxyl carbon. The name of the cyclic structure is used as the parent name, followed by "-carboxylic acid".
Step-by-Step Guide with Examples
Let's walk through several examples to illustrate the process of naming carboxylic acids using the IUPAC system.
Example 1: A simple aliphatic carboxylic acid
Consider the following structure:
CH₃CH₂CH₂COOH
-
Identify the parent chain: The longest chain containing the carboxyl group has four carbon atoms.
-
Name the parent chain: The four-carbon alkane is butane. Adding the carboxylic acid suffix gives us butanoic acid.
-
Substituents: There are no substituents in this structure.
Therefore, the IUPAC name of this carboxylic acid is butanoic acid.
Example 2: Carboxylic acid with alkyl substituents
Consider the following structure:
CH₃CH(CH₃)CH₂COOH
-
Identify the parent chain: The longest chain containing the carboxyl group has four carbon atoms.
-
Name the parent chain: The four-carbon alkane is butane, resulting in butanoic acid.
-
Substituents: There is a methyl group (CH₃) attached to the carbon at position 2.
-
Numbering: Numbering begins at the carboxyl carbon (C-1).
Therefore, the IUPAC name of this carboxylic acid is 2-methylbutanoic acid.
Example 3: Carboxylic acid with multiple substituents
Consider the following structure:
CH₃CH(CH₃)CH(Cl)CH₂COOH
-
Identify the parent chain: The longest chain containing the carboxyl group has five carbon atoms.
-
Name the parent chain: The five-carbon alkane is pentane, resulting in pentanoic acid.
-
Substituents: There is a methyl group (CH₃) at position 2 and a chloro group (Cl) at position 3.
-
Numbering and Alphabetical Ordering: Numbering begins at the carboxyl carbon (C-1). The substituents are listed alphabetically: 3-chloro, 2-methyl.
Therefore, the IUPAC name of this carboxylic acid is 3-chloro-2-methylpentanoic acid.
Example 4: A carboxylic acid with unsaturation
Consider the following structure:
CH₂=CHCH₂CH₂COOH
-
Identify the parent chain: The longest chain containing the carboxyl group has four carbon atoms.
-
Name the parent chain: The four-carbon alkane is butane. However, there's a double bond.
-
Unsaturation: The double bond is located at position 2 (counting from the carboxyl group).
Therefore, the IUPAC name of this carboxylic acid is pent-3-enoic acid. Note that the double bond is at position 3 in the alkene chain. Always number from the carboxylic acid carbon.
Example 5: A dicarboxylic acid
Consider the following structure:
HOOCCH₂CH₂COOH
-
Identify the parent chain: The longest chain containing both carboxyl groups has four carbon atoms.
-
Name the parent chain: The four-carbon alkane is butane. Since there are two carboxyl groups, the suffix "-dioic acid" is used.
Therefore, the IUPAC name of this dicarboxylic acid is butanedioic acid. (Also known as succinic acid, a common name).
Example 6: A cyclic carboxylic acid
Consider the following structure (cyclohexanecarboxylic acid):
COOH
|
/ \
/ \
/ \
/ \
----------------
-
Identify the parent chain: The ring is cyclohexane.
-
Carboxylic Acid Group: The carboxyl group is directly attached to the ring.
Therefore, the IUPAC name is cyclohexanecarboxylic acid. No numbering is needed since the carboxylic acid carbon is inherently part of the ring's numbering system.
Advanced Considerations and Common Pitfalls
While the basic principles are relatively straightforward, some situations can present challenges:
-
Complex Substituents: When dealing with complex substituents, remember to prioritize the longest chain and systematically name each part, following IUPAC rules for naming branched alkyl groups, halogens, and other functional groups.
-
Prioritizing Functional Groups: Carboxylic acids have a higher priority than many other functional groups. This means that when a molecule contains both a carboxylic acid and another functional group (e.g., an alcohol or ketone), the carboxylic acid group dictates the numbering and the principal functional group suffix.
-
Isomerism: Be mindful of isomerism (structural, geometrical, and stereoisomerism). Specify the type of isomerism using the appropriate prefixes and designations.
-
Consult the IUPAC Blue Book: For extremely complex molecules or ambiguous situations, refer to the official IUPAC nomenclature rules (often referred to as the "Blue Book") for definitive guidance.
Conclusion
Mastering IUPAC nomenclature for carboxylic acids is essential for any student or professional working in organic chemistry. By systematically following the guidelines outlined in this guide, you can confidently assign the correct IUPAC name to any carboxylic acid, regardless of its complexity. Remember to always identify the parent chain, incorporate substituents, handle unsaturation and multiple functional groups properly, and use the appropriate suffixes to reflect the specific structure. Practicing with various examples will solidify your understanding and improve your proficiency. With consistent effort, you'll become adept at navigating the nuances of IUPAC nomenclature and effectively communicate the structures of these important organic compounds.
Latest Posts
Latest Posts
-
Range And Kernel Of A Linear Transformation
Apr 03, 2025
-
The Chemistry Of Living Organisms Is Called Chemistry
Apr 03, 2025
-
Finding Domain Of A Composite Function
Apr 03, 2025
-
Image Formation In A Plane Mirror
Apr 03, 2025
-
Us Air Force Tongue And Quill
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Give The Iupac Name Of The Carboxylic Acid Below. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
