How Are The Elements Organized In The Periodic Table
Muz Play
Apr 06, 2025 · 6 min read
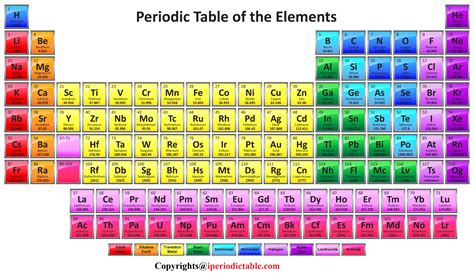
Table of Contents
How Are the Elements Organized in the Periodic Table?
The periodic table, a cornerstone of chemistry, isn't just a random arrangement of elements. Its organization reflects a deep understanding of atomic structure and the resulting properties of elements. Understanding this organization is crucial to grasping the behavior of matter and predicting chemical reactions. This comprehensive guide delves into the intricacies of the periodic table's structure, explaining the principles behind its arrangement and the significance of its various sections.
The Genesis of Organization: Mendeleev's Insight
Before the discovery of subatomic particles, Dmitri Mendeleev, a Russian chemist, revolutionized chemistry in the late 19th century. He meticulously arranged the then-known elements based on their atomic weight and reoccurring chemical properties. This arrangement, though initially based on incomplete data, revealed a striking pattern: elements with similar properties appeared at regular intervals. This pattern led him to predict the existence and properties of elements yet to be discovered, a testament to the power of his organizational system. His table was a pioneering step, but it wasn't perfect, and modern understanding has refined it.
Modern Organization: Beyond Atomic Weight
While Mendeleev's work was groundbreaking, the modern periodic table is organized primarily by atomic number, which represents the number of protons in an atom's nucleus. This is fundamental because the number of protons defines an element's identity. Isotopes, which have varying numbers of neutrons, still belong to the same element because their proton count remains constant.
The periodic table arranges elements in rows (periods) and columns (groups or families).
Periods: Reflecting Energy Levels
The periods, or rows, represent the principal energy levels or shells of electrons surrounding the atom's nucleus. Each period corresponds to a new energy level being filled. As you move across a period, electrons are added to the outermost shell, gradually changing the element's properties. For example, the first period (containing only hydrogen and helium) has electrons only in the first energy level, while the seventh period has electrons filling the seventh energy level.
Groups: Shared Chemical Behaviors
The groups, or columns, are where the real power of the periodic table lies. Elements within the same group share similar outer electron configurations, leading to strikingly similar chemical behavior. The outer electrons, called valence electrons, are primarily responsible for the interactions between atoms, determining how an element will bond with others. This is why elements in the same group often exhibit similar reactivity, oxidation states, and compound formation patterns.
The Major Blocks of the Periodic Table
The periodic table can be further divided into distinct blocks, each representing the type of atomic orbital being filled with electrons.
s-block: Alkali Metals and Alkaline Earth Metals
The s-block, located on the far left, includes groups 1 and 2. Group 1 elements, the alkali metals (lithium, sodium, potassium, etc.), are highly reactive, readily losing one electron to form +1 ions. Group 2 elements, the alkaline earth metals (beryllium, magnesium, calcium, etc.), are also reactive, though less so than alkali metals, losing two electrons to form +2 ions. These elements are crucial in biological systems and industrial processes.
p-block: Diverse Properties and Abundant Elements
The p-block, occupying the largest portion of the table's right side, encompasses groups 13-18. This block contains a wide array of elements with diverse properties. It includes nonmetals, metalloids, and some metals. Group 17, the halogens (fluorine, chlorine, bromine, etc.), are highly reactive nonmetals that readily gain one electron to form -1 ions. Group 18, the noble gases (helium, neon, argon, etc.), are exceptionally unreactive due to their complete outer electron shells. Many essential elements for life, like carbon, nitrogen, and oxygen, reside in this block.
d-block: Transition Metals and Their Unique Qualities
The d-block, situated in the middle of the table, houses the transition metals. These elements are known for their variable oxidation states, meaning they can lose different numbers of electrons to form ions with varying charges. This contributes to their rich and diverse chemistry, with many forming colorful compounds and exhibiting catalytic properties. They are essential components of alloys and are widely used in industry and technology.
f-block: Lanthanides and Actinides - A Separate Story
The f-block, usually placed separately at the bottom of the table, comprises the lanthanides (rare earth elements) and actinides. These elements have electrons filling the f orbitals, leading to unique properties. Many actinides are radioactive, and their chemistry is often complex due to the effects of relativity on their electronic structure. These elements have found applications in nuclear technology and specific industrial applications.
Trends Across the Periodic Table: A Periodic Dance of Properties
The periodic table's organization allows us to predict certain trends in elemental properties. These trends arise from changes in effective nuclear charge, atomic size, and electron shielding.
Atomic Radius: Size Matters
Atomic radius, the size of an atom, generally decreases across a period (left to right) due to increased nuclear charge pulling electrons closer. It generally increases down a group (top to bottom) as new energy levels are added.
Ionization Energy: The Energy of Removal
Ionization energy is the energy required to remove an electron from an atom. It generally increases across a period as the nuclear attraction strengthens and decreases down a group as the outer electrons become further from the nucleus.
Electronegativity: Sharing or Stealing?
Electronegativity measures an atom's ability to attract electrons in a chemical bond. It generally increases across a period and decreases down a group, following similar trends to ionization energy.
Electron Affinity: Accepting Electrons
Electron affinity is the energy change associated with adding an electron to an atom. It shows a less clear trend across the periodic table than ionization energy and electronegativity but generally exhibits some correlation with electronegativity.
Beyond the Basics: Advanced Concepts and Applications
The organization of the periodic table extends beyond the fundamental trends discussed above. More advanced concepts such as electron configuration, orbital hybridization, and molecular orbital theory build upon this foundational framework to explain more complex chemical phenomena. These concepts are integral to understanding chemical bonding, reactivity, and the properties of compounds.
Furthermore, the periodic table isn't just a static organizational tool; it's a dynamic resource with ongoing applications in various fields:
- Material Science: The table guides the development of new materials with specific properties, from lightweight alloys to high-temperature superconductors.
- Medicine: Understanding the properties of elements is critical in designing drugs and understanding their interactions with biological systems.
- Environmental Science: The periodic table assists in monitoring pollutants, understanding environmental cycles, and developing sustainable solutions.
- Nuclear Chemistry: It is fundamental to understanding nuclear reactions, radioactive decay, and the applications of radioactive isotopes.
Conclusion: A Powerful Tool for Understanding Matter
The periodic table, far from being a mere list of elements, is a powerful organizational tool reflecting the underlying principles of atomic structure and chemical behavior. Its arrangement, based on atomic number and electron configuration, allows us to predict and understand trends in elemental properties, paving the way for advancements across a wide array of scientific and technological fields. Mastering the organization of the periodic table is a crucial step towards a deeper understanding of chemistry and the world around us. Its elegance and predictive power continue to inspire awe and drive scientific discovery. It is a testament to the power of observation, pattern recognition, and the human desire to unravel the mysteries of the universe.
Latest Posts
Latest Posts
-
The Study Of Matter And Its Changes
Apr 06, 2025
-
Find The Limit Of Trigonometric Functions
Apr 06, 2025
-
How Did Lincolns Assassination Affect Reconstruction
Apr 06, 2025
-
What Elements Cycle Between Living And Nonliving
Apr 06, 2025
-
Test Statistic For Hypothesis Test Calculator
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Are The Elements Organized In The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
