How Does Temperature Affect Diffusion Rate And Why
Muz Play
Apr 05, 2025 · 6 min read
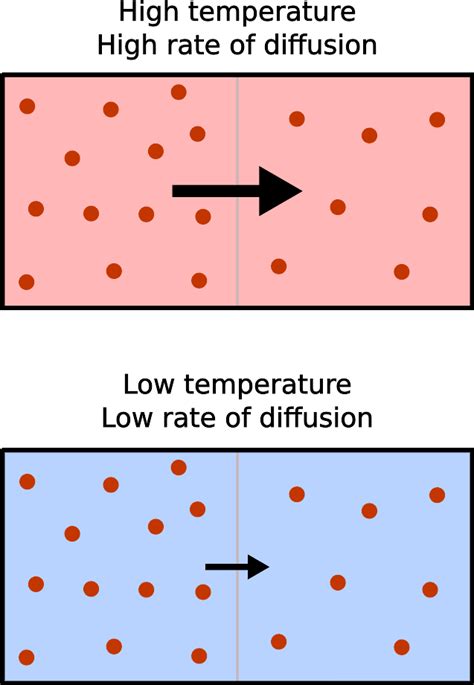
Table of Contents
How Does Temperature Affect Diffusion Rate and Why?
Diffusion, the net movement of particles from a region of higher concentration to a region of lower concentration, is a fundamental process in numerous natural phenomena and technological applications. Understanding how various factors influence diffusion rates is crucial in fields ranging from biology and chemistry to materials science and environmental engineering. One of the most significant factors impacting diffusion rate is temperature. This article delves deep into the relationship between temperature and diffusion rate, exploring the underlying mechanisms and providing practical examples.
The Kinetic Energy Connection: The Heart of the Matter
The primary reason temperature affects diffusion rate lies in its influence on the kinetic energy of particles. Temperature is a measure of the average kinetic energy of the particles within a system. At higher temperatures, particles possess greater kinetic energy, meaning they move faster and more vigorously. This increased movement directly translates to a higher frequency of collisions and a greater probability of particles overcoming the energy barriers that impede their movement.
Increased Kinetic Energy Leads to Faster Diffusion
Imagine a crowded room. At a low temperature (think a chilly winter evening), people might move slowly and cautiously, bumping into each other less frequently. This corresponds to a slower diffusion rate. Now, imagine the same room during a lively party (higher temperature). People are moving rapidly, colliding more frequently, and spreading out across the room much faster – a direct analogy to faster diffusion.
The increased kinetic energy at higher temperatures not only increases the speed of individual particles but also enhances their ability to overcome intermolecular forces. These forces, whether attractive or repulsive, can hinder the movement of particles. Higher kinetic energy allows particles to break free from these attractive forces more easily, facilitating their movement and accelerating the diffusion process.
The Mathematical Relationship: Quantifying the Impact of Temperature
The relationship between temperature and diffusion rate is often described mathematically using the Arrhenius equation:
D = D₀ * exp(-Ea/RT)
Where:
- D is the diffusion coefficient (a measure of how quickly diffusion occurs)
- D₀ is the pre-exponential factor (related to the frequency of particle collisions)
- Ea is the activation energy (the energy barrier particles must overcome to diffuse)
- R is the ideal gas constant
- T is the absolute temperature (in Kelvin)
This equation highlights the exponential relationship between temperature and diffusion rate. As temperature (T) increases, the exponential term (-Ea/RT) decreases, leading to a significant increase in the diffusion coefficient (D). Even a small increase in temperature can result in a noticeable increase in diffusion rate.
Activation Energy: The Energy Hurdle
The activation energy (Ea) represents the energy barrier that particles need to overcome to move from one location to another. This barrier can be due to intermolecular forces, lattice structures in solids, or other obstacles. The Arrhenius equation shows that a higher activation energy results in a slower diffusion rate at a given temperature. Conversely, a lower activation energy facilitates faster diffusion.
Examples Across Different States of Matter:
The impact of temperature on diffusion rate manifests differently across various states of matter:
Gases: The Most Dramatic Effect
In gases, the effect of temperature on diffusion rate is most pronounced. Gas particles are already relatively far apart and experience weak intermolecular forces. A temperature increase significantly boosts their kinetic energy, leading to a dramatic increase in diffusion speed. This is evident in the rapid spreading of scents or smoke in warm air compared to cold air.
Liquids: A More Subtle but Significant Influence
In liquids, the effect is slightly less dramatic than in gases because liquid molecules are closer together and experience stronger intermolecular forces. However, increasing the temperature still increases the kinetic energy, weakening intermolecular attractions and allowing for faster diffusion. The dissolving of sugar in hot tea versus cold tea is a prime example.
Solids: Diffusion at a Glacial Pace, Still Temperature-Dependent
In solids, diffusion occurs much more slowly due to the strong intermolecular forces and the rigid structure of the solid. Atoms or molecules are essentially "locked" in place within a lattice structure. However, even in solids, increasing temperature provides the energy for atoms to overcome the energy barriers and move through the lattice, albeit at a much slower rate compared to gases or liquids. This is crucial in processes like metal alloying, where diffusion at high temperatures is essential for creating homogeneous mixtures.
Practical Applications and Real-World Examples:
The influence of temperature on diffusion has far-reaching implications across numerous fields:
Biology: Enzyme Activity and Cellular Processes
In biological systems, temperature significantly affects the rate of diffusion-dependent processes. Enzyme activity, for example, is highly temperature-dependent. Enzymes facilitate biochemical reactions, and their effectiveness depends on the diffusion of substrates to the active site. Increasing temperature initially increases the diffusion rate, leading to higher enzyme activity. However, excessively high temperatures can denature enzymes, rendering them inactive, thereby decreasing the reaction rate.
Cellular processes such as nutrient uptake and waste removal are also dependent on diffusion, with temperature playing a vital role in regulating these processes. Maintaining optimal temperatures is crucial for cellular health and function.
Metallurgy: Heat Treatment and Alloy Formation
In metallurgy, controlled heating and cooling processes (heat treatment) leverage the effect of temperature on diffusion. Heat treatment modifies the microstructure of metals and alloys, influencing their properties such as strength and hardness. Diffusion at high temperatures is essential for the formation of homogeneous alloys, where different metallic elements intermix at an atomic level.
Food Science: Preservation and Cooking
Temperature significantly impacts the diffusion of flavors, aromas, and moisture in food. Preservation techniques often involve lowering the temperature to reduce the diffusion rate of microorganisms and slow down spoilage processes. Conversely, cooking involves increasing the temperature to accelerate diffusion processes, facilitating the even cooking of food and the release of aromas.
Environmental Science: Pollutant Dispersion and Atmospheric Processes
In environmental science, temperature plays a crucial role in pollutant dispersion and atmospheric processes. The rate at which pollutants diffuse into the atmosphere and spread across geographical areas depends significantly on temperature. Understanding this relationship is critical for environmental modeling and pollution control strategies.
Conclusion: Temperature – A Master Regulator of Diffusion
In conclusion, temperature is a pivotal factor influencing diffusion rates across all states of matter. The exponential relationship described by the Arrhenius equation highlights the significant impact of temperature increases on the kinetic energy of particles, enabling them to overcome energy barriers and move more rapidly. This fundamental principle underpins a wide array of phenomena in diverse fields, making the study of temperature's impact on diffusion a critical area of research and application. From biological processes to industrial applications, understanding this relationship is key to optimizing processes, controlling outcomes, and making informed decisions in various sectors. Further research continues to refine our understanding of this relationship, leading to advancements in technologies and insights into natural processes worldwide.
Latest Posts
Latest Posts
-
Critical Temperature And Pressure Of Nitrogen
Apr 06, 2025
-
The Hormone Of Hyposecretion Is
Apr 06, 2025
-
How Was The First Industrial Revolution Different From The Second
Apr 06, 2025
-
Number Of Atoms In Face Centered Cubic
Apr 06, 2025
-
Two Reactions Between A Grignard Reagent And A Carbonyl Compound
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Does Temperature Affect Diffusion Rate And Why . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
