How Many A Columns Are On The Periodic Table
Muz Play
Apr 03, 2025 · 5 min read
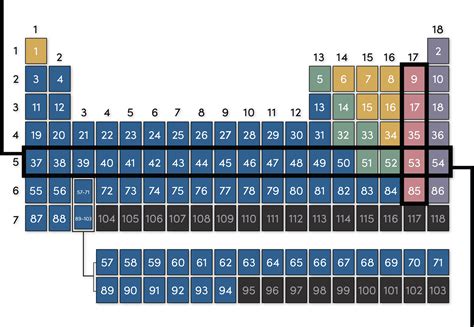
Table of Contents
How Many Columns Are on the Periodic Table? A Deep Dive into the Organization of Elements
The periodic table, a cornerstone of chemistry, isn't just a random arrangement of elements. Its structure, with its rows and columns, reflects fundamental properties and behaviors of atoms. Understanding the organization, particularly the number of columns (or groups), is crucial to grasping the relationships between elements and predicting their chemical characteristics. So, how many columns are on the periodic table? The simple answer is 18. However, the story behind that number and the meaning of each column is far more intricate.
The 18 Columns: Groups and Families of Elements
The periodic table is organized into 18 vertical columns, also known as groups or families. Elements within the same group share similar chemical properties due to having the same number of valence electrons – the electrons in their outermost shell. These valence electrons are the primary participants in chemical bonding, dictating how an element will interact with others.
Understanding Valence Electrons and Group Properties
The number of valence electrons directly corresponds to the group number. For example:
- Group 1 (Alkali Metals): These elements (like Lithium, Sodium, Potassium) have one valence electron, leading to their high reactivity and tendency to lose that electron to form +1 ions.
- Group 2 (Alkaline Earth Metals): With two valence electrons, these elements (like Beryllium, Magnesium, Calcium) are also reactive, though less so than alkali metals, typically forming +2 ions.
- Group 17 (Halogens): These elements (like Fluorine, Chlorine, Bromine) have seven valence electrons. They are highly reactive nonmetals, tending to gain one electron to form -1 ions and achieve a stable electron configuration.
- Group 18 (Noble Gases): These elements (like Helium, Neon, Argon) have a full valence shell (eight electrons, except for Helium with two). This full shell makes them extremely unreactive, hence their name "noble gases."
This pattern continues across the table, with the group number (excluding the transition metals, which have a more complex electron configuration) directly reflecting the number of valence electrons and thus influencing the element's chemical behavior.
Beyond the 18 Main Groups: Transition Metals and Inner Transition Metals
While the 18 main groups provide a straightforward understanding of valence electron configurations, the periodic table also includes the transition metals and inner transition metals, which add complexity.
Transition Metals: D-Block Elements
The transition metals occupy the central block of the periodic table (Groups 3-12). These elements are characterized by partially filled d orbitals, which allows for variable oxidation states – meaning they can lose different numbers of electrons to form ions with varying charges. This contributes to their diverse and often colorful chemistry, as well as their importance in catalysis and numerous industrial applications. The number of valence electrons isn't as straightforward to define here as in the main groups due to the involvement of d-electrons.
Inner Transition Metals: F-Block Elements
The inner transition metals, also known as the lanthanides and actinides, reside at the bottom of the periodic table. They are characterized by partially filled f orbitals. Their chemistry is even more complex than the transition metals, with similar properties within each series (lanthanides and actinides) due to the gradual filling of the f orbitals. These elements are often radioactive and have important applications in nuclear technology and specialized materials science. These elements don't add to the main 18 columns but rather represent a separate expansion of the periodic table.
Historical Development and Evolution of the Periodic Table's Structure
The periodic table's structure, with its 18 columns, wasn't arrived at overnight. It's the result of centuries of scientific inquiry, culminating in Dmitri Mendeleev's groundbreaking work in the late 19th century.
Mendeleev's Contributions and Early Periodic Tables
Mendeleev's genius lay in recognizing patterns in the elements' properties and arranging them according to increasing atomic weight (later refined to atomic number). His early periodic table, although less refined than modern versions, accurately predicted the properties of undiscovered elements based on their predicted positions within the table. While not explicitly having 18 columns as we know them today, his organization laid the groundwork for the modern structure.
The Modern Periodic Table: Refining the Structure
Over time, advancements in atomic theory, particularly the understanding of electron configuration and quantum mechanics, led to a more accurate and refined periodic table. The discovery of noble gases, initially unknown to Mendeleev, also necessitated a modification of the structure. The inclusion of the transition metals and inner transition metals, coupled with a more precise understanding of electron shell filling, eventually resulted in the current 18-column structure.
The Significance of the 18 Columns in Chemistry and Beyond
The 18 columns of the periodic table are more than just a convenient arrangement; they represent a deep-seated connection between the fundamental properties of elements and their chemical behavior.
Predicting Chemical Reactions and Properties
The column organization allows chemists to predict the reactivity and bonding characteristics of elements with remarkable accuracy. Knowing an element's group provides crucial information about its potential to form ionic or covalent bonds, its oxidation states, and its overall chemical behavior.
Understanding Chemical Trends and Periodic Properties
The periodic table's organization reveals trends in properties like electronegativity, ionization energy, and atomic radius. These trends are directly related to the arrangement of electrons and are crucial for understanding chemical reactions and predicting the outcome of chemical processes.
Applications in Materials Science, Technology, and Medicine
The understanding of the periodic table is essential in various fields. Materials scientists use it to design new materials with specific properties, engineers utilize it to select elements for technological applications, and medical professionals employ its principles in understanding the role of trace elements in biological processes.
Conclusion: A Dynamic and Ever-Evolving System
The periodic table, with its 18 columns, is a testament to the power of scientific inquiry and the elegance of nature's underlying patterns. While the 18-column structure provides a framework for understanding elemental relationships, it’s also important to remember that the periodic table is not static. Ongoing research may lead to even more nuanced understandings of the elements and perhaps even further refinements to its organization. However, the core principle of organizing elements based on their electronic structure and resulting properties remains the bedrock of chemistry and countless other scientific disciplines. The 18 columns, therefore, represent not just a numerical fact, but a fundamental organizing principle of our understanding of the material world.
Latest Posts
Latest Posts
-
Do Double Bonds Increase Boiling Point
Apr 03, 2025
-
Staphylococcus Epidermidis Hemolysis On Blood Agar
Apr 03, 2025
-
Formula For Rate Of Natural Increase
Apr 03, 2025
-
Are All Bronsted Acids Lewis Acids
Apr 03, 2025
-
What Is The Purpose Of An Indicator
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many A Columns Are On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
