How To Draw A Water Molecule
Muz Play
Apr 05, 2025 · 5 min read
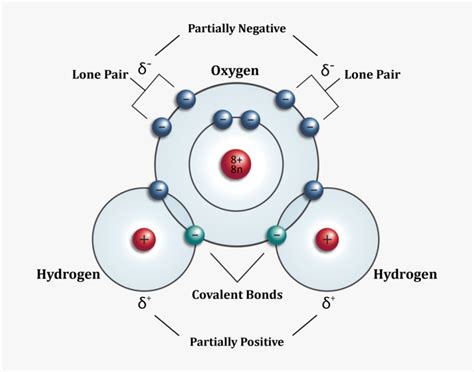
Table of Contents
How to Draw a Water Molecule: A Comprehensive Guide for Beginners and Beyond
Drawing a water molecule might seem deceptively simple, but accurately representing its structure reveals a fascinating glimpse into the world of chemistry. This comprehensive guide will take you through various methods, from basic representations to more advanced models, equipping you with the skills to draw a water molecule effectively for any purpose, whether for school projects, presentations, or simply satisfying your scientific curiosity.
Understanding the Water Molecule: H₂O
Before we delve into the drawing techniques, let's understand what we're representing. A water molecule (H₂O) consists of two hydrogen atoms and one oxygen atom. The key to a realistic depiction lies in understanding the arrangement of these atoms:
-
Covalent Bonds: The atoms are connected via covalent bonds, where they share electrons. This sharing isn't equal; oxygen is more electronegative, meaning it attracts the shared electrons more strongly. This leads to a polar molecule.
-
Bent Shape: The molecule isn't linear; it has a bent or V-shaped geometry due to the lone pairs of electrons on the oxygen atom. These lone pairs repel the bonding pairs, pushing the hydrogen atoms closer together.
-
Bond Angles: The angle between the two hydrogen-oxygen bonds is approximately 104.5 degrees, not 180 degrees as in a linear structure. This bent shape is crucial for many of water's unique properties.
Method 1: The Simple Stick-and-Ball Model
This is the most basic representation, ideal for beginners or when simplicity is prioritized.
Steps:
-
Draw the Oxygen Atom: Start by drawing a large circle to represent the oxygen atom (O). You can label it with 'O'.
-
Add the Hydrogen Atoms: Draw two smaller circles, representing the hydrogen atoms (H), slightly below and to either side of the oxygen atom. Ensure they are not directly in line.
-
Connect with Lines: Draw straight lines connecting each hydrogen atom to the oxygen atom. These lines represent the covalent bonds.
-
Labeling (Optional): Add labels (O and H) to clearly identify each atom. You can also label the bond angles (approximately 104.5°).
Example:
H
/ \
/ \
O H
Method 2: The Ball-and-Stick Model with Bond Angles
This method improves upon the simple stick-and-ball model by visually emphasizing the bond angle.
Steps:
-
Draw the Atoms: As before, draw a larger circle for oxygen and two smaller circles for hydrogen.
-
Accurate Placement: Use a protractor to ensure the angle between the two hydrogen-oxygen bonds is approximately 104.5 degrees. This requires careful positioning of the hydrogen atoms.
-
Connect with Sticks: Draw slightly thicker lines to represent the covalent bonds.
-
Labeling: Label the atoms (O and H) and the bond angle (104.5°).
Method 3: The Space-Filling Model
This model provides a more realistic representation of the molecule's size and shape, showing the relative sizes of the atoms and how they occupy space.
Steps:
-
Draw Overlapping Circles: Draw a larger circle for the oxygen atom. Then, draw two smaller circles for the hydrogen atoms, overlapping slightly with the oxygen atom. The overlap represents the electron clouds.
-
Shape and Size: Adjust the size and overlap to reflect the relative sizes of the atoms: oxygen is significantly larger than hydrogen.
-
No Explicit Bonds: Unlike the previous models, you don't draw explicit lines representing the bonds. The overlap of the atoms suggests the bonding.
-
Labeling: Label the atoms (O and H).
Method 4: Advanced Representations: Illustrating Polarity
Water's polarity is a critical property, impacting its behavior and interactions. More advanced drawings can illustrate this.
Steps:
-
Basic Structure: Begin with any of the previous models.
-
Partial Charges: Indicate the partial charges using the Greek letter delta (δ). Oxygen has a partial negative charge (δ-), while each hydrogen has a partial positive charge (δ+).
-
Dipole Moment: Draw a dipole moment arrow (an arrow with a plus sign at the tail and an arrowhead at the head), pointing from the positive end (hydrogen side) to the negative end (oxygen side). This visually represents the molecule's dipole moment.
-
Electron Density: You can subtly shade the oxygen atom darker to visually represent the higher electron density around it.
Software and Tools for Drawing Water Molecules
For more complex and visually appealing representations, consider using specialized software:
-
ChemDraw: A widely used software for drawing chemical structures. It allows for precise bond angles, atom sizes, and advanced features.
-
MolView: An online tool that allows you to visualize molecules in 3D and create various representations, including ball-and-stick, space-filling, and more.
-
Avogadro: A free, open-source molecular editor that offers a range of visualization and manipulation capabilities.
Beyond the Basics: Illustrating Hydrogen Bonding
Water molecules are unique in their ability to form hydrogen bonds with each other. This is a crucial aspect that can be added to more sophisticated drawings:
-
Multiple Molecules: Draw several water molecules.
-
Hydrogen Bonds: Use dotted or dashed lines to represent the hydrogen bonds. These bonds occur between the slightly positive hydrogen atom of one molecule and the slightly negative oxygen atom of another molecule.
-
Labeling: Label both the covalent bonds and the hydrogen bonds to distinguish between them.
This detailed representation accurately captures the interactions responsible for many of water's properties, such as its high boiling point and surface tension.
Tips for Effective Drawing
-
Use the Right Tools: A ruler, protractor, and colored pencils or markers will significantly enhance accuracy and visual appeal.
-
Practice Makes Perfect: Drawing water molecules repeatedly will improve your precision and speed.
-
Maintain Consistency: Ensure your representation is consistent with the chosen model; don't mix styles within a single drawing.
-
Clarity and Labeling: Clear labels and annotations are crucial for understanding.
-
Consider your Audience: The complexity of your drawing should match the audience's understanding of chemistry.
Conclusion: Mastering the Art of Drawing Water Molecules
Drawing a water molecule effectively goes beyond simply putting atoms together. It's about understanding the underlying chemistry and representing it visually. This guide has provided a range of methods, from simple to sophisticated, enabling you to choose the approach most suitable for your needs. By mastering these techniques, you can communicate complex scientific concepts clearly and accurately, whether for educational purposes, scientific research, or simply for the love of scientific illustration. Remember, practice and attention to detail are key to achieving accurate and visually appealing representations of this fundamental molecule.
Latest Posts
Latest Posts
-
When To Use Brackets Or Parentheses In Domain And Range
Apr 05, 2025
-
Which Step In Cellular Respiration Produces The Most Atp
Apr 05, 2025
-
The Highness Or Lowness Of A Sound Is Called
Apr 05, 2025
-
Is Oxidation Gaining Or Losing Electrons
Apr 05, 2025
-
What Is The Difference Between Bacteriostatic And Bactericidal
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How To Draw A Water Molecule . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
