Identify What A Coffee Cup Calorimeter Measures
Muz Play
Apr 04, 2025 · 7 min read
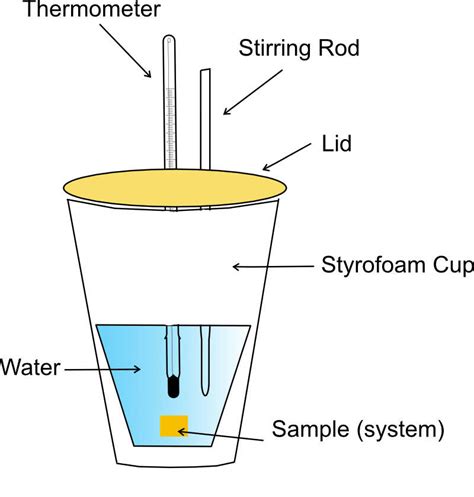
Table of Contents
Identify What a Coffee Cup Calorimeter Measures
A coffee cup calorimeter, also known as a constant-pressure calorimeter, is a simple yet effective device used in chemistry and physics experiments to measure the heat transfer associated with a reaction or process occurring in solution. It's crucial to understand that it doesn't directly measure temperature; rather, it's used to indirectly determine the heat exchanged based on observed temperature changes. This article will delve deep into the principles behind its operation, the specific quantities it measures, the limitations, and how to interpret the obtained data.
Understanding the Fundamentals: Heat Transfer and Specific Heat Capacity
Before diving into the specifics of a coffee cup calorimeter, let's review fundamental concepts. Heat transfer refers to the flow of thermal energy from a hotter object or system to a colder one. This transfer continues until thermal equilibrium is reached, meaning both systems are at the same temperature. The amount of heat transferred (q) is directly proportional to the mass (m) of the substance, its specific heat capacity (c), and the change in temperature (ΔT). This relationship is expressed by the following equation:
q = mcΔT
- q: heat transferred (in Joules, J)
- m: mass of the substance (in grams, g)
- c: specific heat capacity of the substance (in J/g°C or J/gK)
- ΔT: change in temperature (in °C or K)
Specific heat capacity (c) is a crucial property of a substance. It represents the amount of heat required to raise the temperature of 1 gram of that substance by 1 degree Celsius (or 1 Kelvin). Water, for instance, has a relatively high specific heat capacity (approximately 4.18 J/g°C), meaning it requires a significant amount of heat to change its temperature.
The Coffee Cup Calorimeter: Design and Operation
The coffee cup calorimeter is aptly named because it typically uses a Styrofoam cup (or a similar well-insulated container) as its core component. Styrofoam's low thermal conductivity minimizes heat exchange between the calorimeter's contents and the surrounding environment. This approximation of an adiabatic system is crucial for accurate measurements. The cup is usually nested within another cup to further reduce heat loss. A thermometer is inserted through the lid to monitor the temperature changes within the calorimeter. A stirrer is also often included to ensure even distribution of heat throughout the solution.
How it Works: Measuring Heat of Reaction
To use a coffee cup calorimeter, a reaction is carried out within the Styrofoam cup. The temperature of the solution is monitored before, during, and after the reaction. The difference in temperature, ΔT, represents the temperature change resulting from the heat absorbed or released by the reaction. Since the calorimeter is approximately adiabatic, the heat exchanged during the reaction is assumed to be entirely absorbed or released by the solution within the calorimeter.
Therefore, by knowing the mass (m) of the solution and its specific heat capacity (c), one can calculate the heat transferred (q) using the equation q = mcΔT. This calculated heat (q) represents the heat of reaction (ΔH), or enthalpy change, for the process under investigation, provided the reaction takes place at constant pressure.
What a Coffee Cup Calorimeter Actually Measures
It's important to reiterate that a coffee cup calorimeter directly measures temperature change (ΔT). It indirectly measures the heat transferred (q), specifically the heat of reaction at constant pressure (ΔH). This is because:
- It assumes constant pressure: The calorimeter is open to the atmosphere, so the pressure remains essentially constant. This is crucial, as enthalpy changes (ΔH) are defined under constant pressure conditions.
- It assumes negligible heat loss: The calorimeter is designed to minimize heat exchange with the surroundings. However, some heat loss inevitably occurs. Advanced calorimeters correct for these losses, but simple coffee cup calorimeters often rely on the assumption of negligible heat loss. This leads to some inherent error in the measurement.
- It relies on the specific heat capacity: Accurate measurement depends on knowing the specific heat capacity of the solution. The specific heat capacity of a mixture may differ from that of pure water.
Types of Reactions Studied with Coffee Cup Calorimeters
Coffee cup calorimeters find applications in studying a variety of reactions, including:
1. Acid-Base Neutralization Reactions:
Determining the enthalpy change of neutralization reactions, like the reaction between a strong acid (e.g., HCl) and a strong base (e.g., NaOH).
2. Dissolution Reactions:
Measuring the heat of solution (enthalpy of dissolution) when a solute dissolves in a solvent (e.g., dissolving NaCl in water).
3. Metal-Acid Reactions:
Investigating the heat released during a metal-acid reaction (e.g., the reaction between zinc and hydrochloric acid).
4. Specific Heat Capacity Determination:
A coffee cup calorimeter can be used to determine the specific heat capacity of an unknown substance by measuring the temperature change when a known amount of heat is added.
Limitations of Coffee Cup Calorimeters
While coffee cup calorimeters are simple and inexpensive, they have limitations:
- Heat loss: Despite the insulating properties of the Styrofoam cup, some heat loss to the surroundings is inevitable. This leads to inaccuracies in the measured heat of reaction.
- Incomplete reactions: The calorimeter doesn't guarantee complete reaction; some reactants might remain unreacted.
- Non-adiabatic conditions: The system is only approximately adiabatic. Improved insulation and experimental techniques can minimize heat exchange but cannot entirely eliminate it.
- Limited accuracy: Coffee cup calorimeters provide relatively low-precision measurements compared to more sophisticated calorimeters. They are best suited for educational demonstrations and introductory experiments, not high-precision scientific work.
- Assumption of constant specific heat: The specific heat of a solution can change with temperature.
Improving Accuracy and Addressing Limitations
While the inherent simplicity of a coffee cup calorimeter contributes to its limitations, certain strategies can improve accuracy and minimize errors:
- Improved insulation: Using multiple nested Styrofoam cups or employing other forms of thermal insulation can significantly reduce heat loss.
- Rapid measurements: Performing the experiment quickly minimizes the time available for heat exchange with the surroundings.
- Stirring: Thorough and consistent stirring helps ensure uniform temperature throughout the solution.
- Calibration: Calibrating the calorimeter with a known reaction can help account for heat loss and improve the accuracy of the measurements.
- Use of a lid: Using a lid with a small hole for the thermometer reduces heat loss through evaporation.
Interpreting Results and Data Analysis
Data obtained from a coffee cup calorimeter experiment typically involves plotting temperature against time. This graph reveals the temperature change during the reaction. The ΔT is usually calculated from the difference between the initial and final temperatures after the reaction has reached equilibrium. This ΔT is then used in the equation q = mcΔT to calculate the heat exchanged. Remember, a positive q indicates an exothermic reaction (heat released), while a negative q indicates an endothermic reaction (heat absorbed).
The results should always be presented alongside any assumptions made (e.g., negligible heat loss) and any sources of error. A thorough error analysis is crucial for evaluating the reliability of the obtained results.
Beyond the Coffee Cup: Advanced Calorimetry Techniques
While coffee cup calorimeters serve as a valuable introductory tool, more sophisticated calorimeters are necessary for precise measurements and complex reactions. Bomb calorimeters, for example, are used to measure the heat of combustion under constant volume conditions. These advanced calorimeters often incorporate features to minimize heat loss and precisely measure temperature changes, leading to significantly higher accuracy compared to a coffee cup calorimeter.
In conclusion, a coffee cup calorimeter is a simple and effective tool for measuring the heat of reaction at constant pressure. It indirectly measures the heat exchanged (q) by directly measuring the temperature change (ΔT) in a well-insulated system. While its simplicity introduces some limitations, understanding these limitations and applying appropriate experimental techniques can lead to reasonably accurate results for educational and introductory experiments. Remember, always consider the assumptions and limitations inherent in the method when interpreting the data and drawing conclusions.
Latest Posts
Latest Posts
-
When Does Dna Replication Occur In A Eukaryotic Cell
Apr 04, 2025
-
What Was Shown By Both Redis And Pasteurs Experiments
Apr 04, 2025
-
The Elements In Group 1 Are Called The
Apr 04, 2025
-
Proof Of The Inverse Function Theorem
Apr 04, 2025
-
Nonmetal Halogen Family Atomic Mass 35
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Identify What A Coffee Cup Calorimeter Measures . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
