If Delta S Is Positive Is It Spontaneous
Muz Play
Apr 01, 2025 · 5 min read
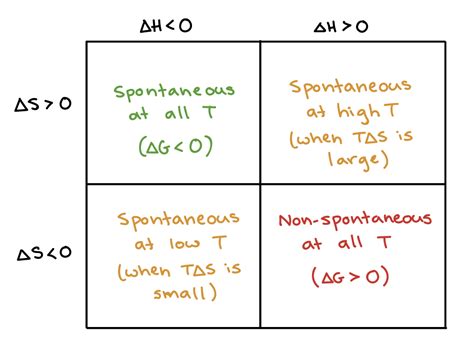
Table of Contents
Is a Positive Delta S Always Spontaneous? Understanding Entropy and Spontaneity
The question of whether a positive change in entropy (ΔS > 0) guarantees a spontaneous process is a fundamental concept in thermodynamics. While a positive ΔS strongly favors spontaneity, it's not the sole determinant. Spontaneity is a complex interplay of entropy and enthalpy, governed by the Gibbs Free Energy (ΔG). Let's delve deep into this crucial relationship.
Understanding Entropy (ΔS)
Entropy, often described as the measure of disorder or randomness in a system, is a crucial thermodynamic property. A positive change in entropy (ΔS > 0) signifies an increase in disorder. This could manifest in several ways:
Examples of Processes with Positive ΔS:
- Melting of a solid: A solid's ordered structure transitions to the more disordered liquid state.
- Boiling of a liquid: The transition from a liquid to a gas drastically increases disorder due to increased molecular freedom.
- Dissolution of a solid in a liquid: The solid's ordered structure breaks down, and its particles disperse randomly within the liquid solvent.
- Expansion of a gas: As a gas expands into a larger volume, its molecules become more dispersed, leading to higher entropy.
- Chemical reactions that increase the number of molecules: Reactions producing more gaseous products than reactants generally have a positive ΔS.
Key takeaway: A positive ΔS indicates a process that increases the randomness or disorder of the system. This inherent tendency towards disorder is a driving force in many natural processes.
Understanding Enthalpy (ΔH)
Enthalpy (ΔH) represents the heat exchanged between a system and its surroundings at constant pressure. It's a measure of the system's heat content.
Exothermic vs. Endothermic Reactions and their ΔH values:
- Exothermic reactions (ΔH < 0): Release heat to the surroundings. These reactions often feel warm or hot.
- Endothermic reactions (ΔH > 0): Absorb heat from the surroundings. These reactions often feel cold.
Key takeaway: Enthalpy change provides information about the heat transfer during a process. While a negative ΔH (exothermic) favors spontaneity, it's not the only factor.
The Gibbs Free Energy (ΔG) and Spontaneity
The true determinant of spontaneity at constant temperature and pressure is the Gibbs Free Energy (ΔG), defined as:
ΔG = ΔH - TΔS
where:
- ΔG = change in Gibbs Free Energy
- ΔH = change in enthalpy
- T = absolute temperature (in Kelvin)
- ΔS = change in entropy
Spontaneity is dictated by the sign of ΔG:
- ΔG < 0 (negative): The process is spontaneous under the given conditions. The reaction proceeds without external input.
- ΔG > 0 (positive): The process is non-spontaneous under the given conditions. It requires external input (like energy) to occur.
- ΔG = 0 (zero): The process is at equilibrium. The forward and reverse reactions occur at the same rate.
The Role of Temperature (T)
The temperature plays a critical role in determining spontaneity, especially when ΔH and ΔS have opposing signs. The TΔS term in the Gibbs Free Energy equation becomes increasingly significant at higher temperatures.
Scenarios Based on ΔH and ΔS:
-
ΔH < 0 and ΔS > 0: This scenario is always spontaneous (ΔG < 0) at all temperatures. Both enthalpy and entropy favor the process. Examples include many exothermic reactions that increase disorder (e.g., combustion).
-
ΔH > 0 and ΔS > 0: This scenario is spontaneous only at high temperatures. At low temperatures, the positive ΔH dominates, making ΔG positive. However, as temperature increases, the TΔS term becomes larger, eventually making ΔG negative. Examples include many endothermic phase transitions (e.g., melting ice).
-
ΔH < 0 and ΔS < 0: This scenario is spontaneous only at low temperatures. At high temperatures, the negative TΔS term dominates, making ΔG positive. Examples might include certain chemical reactions where order increases and heat is released.
-
ΔH > 0 and ΔS < 0: This scenario is never spontaneous (ΔG > 0) at any temperature. Both enthalpy and entropy oppose the process.
Positive ΔS: A Favoring Factor, Not a Guarantee
A positive ΔS increases the likelihood of spontaneity, but it doesn't guarantee it. The overall spontaneity is determined by the interplay between ΔH and TΔS as reflected in the ΔG value. Therefore, solely focusing on a positive ΔS is insufficient for predicting spontaneity.
Practical Applications and Examples:
Let's illustrate these concepts with some real-world examples:
1. The Dissolution of Potassium Nitrate (KNO3): When potassium nitrate dissolves in water, it's an endothermic process (ΔH > 0). However, the dissolution significantly increases the disorder of the system (ΔS > 0) as the ordered crystal structure breaks down and the ions become dispersed in the solution. At room temperature, the TΔS term overcomes the positive ΔH, leading to a negative ΔG and spontaneous dissolution.
2. The Combustion of Methane (CH4): The combustion of methane is a highly exothermic reaction (ΔH < 0), releasing a significant amount of heat. The reaction also produces more gas molecules than consumed, resulting in a positive ΔS. This combination (ΔH < 0 and ΔS > 0) makes the combustion of methane highly spontaneous at all temperatures.
3. The Formation of an Ice Crystal from Liquid Water: The freezing of water is an exothermic process (ΔH < 0) because heat is released. However, the process also decreases the disorder (ΔS < 0) as the water molecules transition from a relatively disordered liquid to a highly ordered crystalline solid. At temperatures below 0°C (273 K), the magnitude of ΔH is larger than TΔS, resulting in a negative ΔG and spontaneous freezing. Above 0°C, the reverse is true, leading to melting (a spontaneous process at that temperature).
Conclusion:
While a positive change in entropy (ΔS > 0) favors spontaneity by increasing disorder, it's not the only factor. The spontaneity of a process hinges on the Gibbs Free Energy (ΔG), which accounts for both enthalpy (ΔH) and entropy changes. Temperature plays a crucial role in determining the relative contributions of ΔH and TΔS. Only through careful analysis of both enthalpy and entropy changes, along with the temperature, can we accurately predict whether a process will be spontaneous. Remember, a positive ΔS is a significant contributing factor, but not a sufficient condition for predicting spontaneity.
Latest Posts
Latest Posts
-
What Is The Basic Idea Behind Disengagement Theory
Apr 03, 2025
-
Is Bromine A Good Leaving Group
Apr 03, 2025
-
Five Blind Men And The Elephant
Apr 03, 2025
-
Absorption Spectrum Chlorophyll A And B
Apr 03, 2025
-
Mechanical And Organic Solidarity By Emile Durkheim
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about If Delta S Is Positive Is It Spontaneous . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
