Is Bromine A Good Leaving Group
Muz Play
Apr 03, 2025 · 6 min read
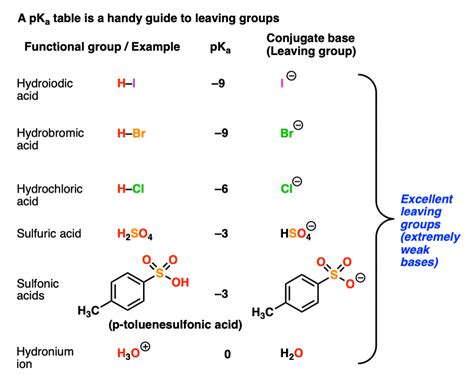
Table of Contents
Is Bromine a Good Leaving Group? A Deep Dive into Leaving Group Ability
Bromine's role as a leaving group in organic chemistry reactions is a crucial topic for students and researchers alike. Understanding its effectiveness is essential for predicting reaction outcomes and designing efficient synthetic strategies. This comprehensive article will delve into the factors determining leaving group ability, analyze bromine's performance in this regard, and explore its applications in various organic reactions.
Understanding Leaving Groups
Before we assess bromine's capabilities, let's establish a clear understanding of what constitutes a "good" leaving group. A good leaving group is an atom or group of atoms that readily departs from a molecule, taking a pair of electrons with it. This departure generates a more stable negative ion or neutral molecule. The stability of the departing species directly influences the reaction rate.
Several factors contribute to a group's effectiveness as a leaving group:
1. Stability of the Leaving Group Anion
The most significant factor is the stability of the anion formed when the group departs. Stable anions are weak bases, meaning they are less likely to react with other molecules to reform the original bond. Stronger bases are poor leaving groups, while weaker bases are good leaving groups. This is because strong bases readily accept protons, reversing the departure process.
2. Polarizability of the Leaving Group
A highly polarizable leaving group can better stabilize the negative charge through electron delocalization. This enhanced stability translates to a greater ability to depart. Larger atoms with more diffuse electron clouds tend to be more polarizable.
3. Resonance Stabilization
If the leaving group can benefit from resonance stabilization after departure, the negative charge is dispersed over multiple atoms. This significantly enhances the leaving group's stability and improves its ability to leave.
4. Size and Steric Effects
While not as dominant as stability, the size and steric bulk of a leaving group can influence its ability to leave. Bulky groups may experience steric hindrance, making departure more difficult.
Bromine as a Leaving Group: A Detailed Analysis
Bromine (Br), in the form of a bromide ion (Br⁻), is generally considered a good leaving group. Let's examine why:
1. Bromide Ion Stability
The bromide ion (Br⁻) is a relatively stable anion. It's a weak base and less likely to abstract a proton compared to stronger bases like hydroxide (OH⁻) or alkoxide (RO⁻). This inherent stability makes it a favorable leaving group.
2. Polarizability of Bromine
Bromine is a relatively large atom with a sizeable electron cloud. Its high polarizability effectively disperses the negative charge, further stabilizing the bromide ion after its departure. This contributes significantly to its effectiveness as a leaving group.
3. Absence of Resonance Stabilization in Simple Cases
In simple alkyl bromides, bromine doesn't experience resonance stabilization. However, in more complex molecules containing conjugated systems, the departing bromide ion might benefit from some degree of resonance stabilization, further enhancing its leaving group ability.
4. Comparing Bromine to Other Halogens
Comparing bromine to other halogens provides valuable insights. Iodine (I⁻) is generally considered an even better leaving group due to its larger size and even greater polarizability. However, fluorine (F⁻) is a very poor leaving group due to its small size, high electronegativity, and poor polarizability, resulting in a highly unstable and reactive fluoride ion. Chlorine (Cl⁻) sits between bromine and iodine in terms of leaving group ability, exhibiting intermediate properties.
Reactions Where Bromine Acts as a Leaving Group
Bromine's proficiency as a leaving group makes it a key component in a variety of organic reactions, including:
1. Nucleophilic Substitution Reactions (SN1 and SN2)
Bromine is a prominent leaving group in both SN1 and SN2 reactions. In SN2 reactions, the backside attack of a nucleophile simultaneously displaces the bromide ion. The reaction rate is highly sensitive to steric hindrance around the carbon atom. In SN1 reactions, the bromide ion leaves first, creating a carbocation intermediate, which is then attacked by the nucleophile. The rate-determining step is the formation of the carbocation, making the stability of the carbocation crucial. Tertiary alkyl bromides undergo SN1 reactions more readily than primary or secondary alkyl bromides due to the greater stability of the tertiary carbocation.
2. Elimination Reactions (E1 and E2)
Bromine also acts as a leaving group in elimination reactions, where a double bond is formed by removing a proton and the bromide ion from adjacent carbon atoms. E2 reactions are concerted, meaning the proton abstraction and bromide departure happen simultaneously. E1 reactions involve a two-step mechanism similar to SN1 reactions, with the formation of a carbocation intermediate followed by proton loss.
3. Grignard Reactions
While not directly acting as a leaving group in the initial step, alkyl bromides are crucial substrates in Grignard reagent formation. The reaction of an alkyl halide with magnesium metal generates a Grignard reagent, which then reacts with various electrophiles. The bromide leaves the alkyl group during the formation of the Grignard reagent.
Factors Affecting Bromine's Leaving Group Ability in Specific Cases
While bromine is generally a good leaving group, several factors can influence its effectiveness in specific contexts:
1. Steric Hindrance
Bulky groups surrounding the carbon-bromine bond can hinder the departure of the bromide ion, slowing down reaction rates, especially in SN2 reactions.
2. Solvent Effects
The solvent plays a significant role in reactions involving bromide as a leaving group. Polar protic solvents often favor SN1 and E1 reactions, while polar aprotic solvents generally favor SN2 reactions.
3. Nature of the Nucleophile/Base
A strong nucleophile or base will generally accelerate the reaction rate, especially in SN2 and E2 reactions.
4. Temperature
Higher temperatures generally increase reaction rates for both substitution and elimination reactions, facilitating the departure of the bromide ion.
Conclusion: Bromine – A Versatile Leaving Group
In conclusion, bromine consistently performs well as a leaving group in various organic reactions. Its stability as a bromide ion, coupled with its reasonable polarizability, makes it a reliable choice for facilitating nucleophilic substitution and elimination reactions. While factors like steric hindrance and solvent effects can modify its behavior, understanding these influencing factors allows for precise control over reaction outcomes. Bromine's versatility makes it an essential component in a wide range of organic syntheses, solidifying its status as a valuable tool for organic chemists. Its role in reactions highlights the interplay between leaving group ability, reaction mechanisms, and reaction conditions, enriching our understanding of organic reaction dynamics. Further research continues to explore the nuances of bromine's leaving group behavior in diverse reaction systems, expanding its applications in organic synthesis and beyond.
Latest Posts
Latest Posts
-
Do Double Bonds Increase Boiling Point
Apr 03, 2025
-
Staphylococcus Epidermidis Hemolysis On Blood Agar
Apr 03, 2025
-
Formula For Rate Of Natural Increase
Apr 03, 2025
-
Are All Bronsted Acids Lewis Acids
Apr 03, 2025
-
What Is The Purpose Of An Indicator
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Bromine A Good Leaving Group . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
