In An Ionic Bond Electrons Are
Muz Play
Apr 01, 2025 · 6 min read
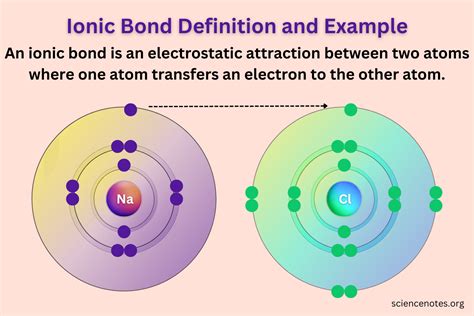
Table of Contents
In an Ionic Bond, Electrons Are…Transferred! Understanding the Fundamentals of Ionic Bonding
Ionic bonds, a cornerstone of chemistry, represent a fundamental type of chemical bonding characterized by the transfer of electrons between atoms. This transfer creates ions – charged particles – that are electrostatically attracted to each other, forming a stable ionic compound. Understanding this electron transfer is crucial to grasping the properties and behavior of a vast array of materials. This comprehensive guide will delve into the intricacies of ionic bonding, exploring the driving forces behind electron transfer, the formation of ions, and the properties of ionic compounds.
The Dance of Electrons: Why Transfer Happens
The fundamental principle governing ionic bond formation is the octet rule. This rule dictates that atoms tend to gain, lose, or share electrons to achieve a stable electron configuration with eight valence electrons in their outermost shell (except for hydrogen and helium, which aim for two). Atoms with a nearly full or nearly empty valence shell are particularly prone to participating in ionic bonding.
Electronegativity: The Tug-of-War
Electronegativity plays a critical role in determining whether an ionic bond will form. Electronegativity is a measure of an atom's ability to attract electrons towards itself in a chemical bond. A significant difference in electronegativity between two atoms is a prerequisite for ionic bonding. When a highly electronegative atom encounters a low electronegativity atom, the electronegative atom exerts a strong pull on the valence electrons of the less electronegative atom. If this pull is strong enough, the electron is completely transferred, resulting in the formation of ions.
Ionization Energy and Electron Affinity: The Energy Equation
Two key concepts underpin the energetics of electron transfer: ionization energy and electron affinity.
-
Ionization Energy: This is the energy required to remove an electron from a neutral atom in its gaseous state. Atoms with low ionization energies readily lose electrons, becoming positively charged cations. Alkaline metals, for instance, have low ionization energies and are readily oxidized (lose electrons).
-
Electron Affinity: This is the energy change that occurs when an electron is added to a neutral atom in its gaseous state. Atoms with high electron affinities readily gain electrons, becoming negatively charged anions. Halogens, for example, have high electron affinities and are easily reduced (gain electrons).
The transfer of electrons is energetically favorable when the energy released by the high electron affinity of the anion-forming atom outweighs the energy required to ionize the cation-forming atom. This energy balance drives the formation of the ionic bond.
Formation of Ions: A Closer Look
The process of ionic bond formation can be visualized as a two-step process:
-
Electron Transfer: A highly electronegative atom (usually a nonmetal) attracts and accepts one or more electrons from a less electronegative atom (usually a metal). This transfer creates ions: the atom that loses electrons becomes a positively charged cation, and the atom that gains electrons becomes a negatively charged anion.
-
Electrostatic Attraction: The oppositely charged ions are then held together by strong electrostatic forces of attraction, forming an ionic bond. This attraction is governed by Coulomb's Law, which states that the force of attraction is directly proportional to the product of the charges and inversely proportional to the square of the distance between them. The stronger the charges and the closer the ions, the stronger the ionic bond.
Examples of Ion Formation:
-
Sodium Chloride (NaCl): Sodium (Na), an alkali metal, has one valence electron. Chlorine (Cl), a halogen, has seven valence electrons. Sodium readily loses its valence electron to achieve a stable octet, forming a +1 cation (Na⁺). Chlorine gains this electron, completing its octet and forming a -1 anion (Cl⁻). The electrostatic attraction between Na⁺ and Cl⁻ forms the ionic bond in NaCl.
-
Magnesium Oxide (MgO): Magnesium (Mg) has two valence electrons, while oxygen (O) has six. Magnesium loses two electrons to become a +2 cation (Mg²⁺), and oxygen gains two electrons to become a -2 anion (O²⁻). The strong electrostatic attraction between Mg²⁺ and O²⁻ forms the ionic bond in MgO.
Properties of Ionic Compounds: A Consequence of the Bond
The unique properties of ionic compounds are a direct consequence of the strong electrostatic forces between the ions and their crystal lattice structure.
-
High Melting and Boiling Points: The strong electrostatic attractions between ions require significant energy to overcome, resulting in high melting and boiling points.
-
Brittleness: Ionic crystals have a rigid, ordered structure. Applying stress can cause like-charged ions to come into close proximity, leading to electrostatic repulsion and fracture of the crystal.
-
Solubility in Polar Solvents: Ionic compounds often dissolve readily in polar solvents like water. The polar solvent molecules can surround and separate the ions, weakening the electrostatic attractions and allowing the ions to disperse into the solution.
-
Electrical Conductivity: Ionic compounds are typically poor conductors of electricity in the solid state because the ions are fixed in the crystal lattice. However, when molten or dissolved in water, they become good conductors as the ions are free to move and carry an electric current.
Beyond the Basics: Factors Influencing Ionic Bonding
While the basic principles of ionic bonding are straightforward, several factors can influence the strength and characteristics of the ionic bond:
-
Charge of the ions: Higher charges on the ions lead to stronger electrostatic attractions and thus stronger ionic bonds.
-
Size of the ions: Smaller ions result in stronger ionic bonds because the electrostatic attraction is inversely proportional to the distance between the ions.
-
Polarizability: The ability of an ion's electron cloud to be distorted by an external electric field influences the strength of the ionic bond. Larger ions are more polarizable.
-
Coordination Number: This refers to the number of ions of opposite charge surrounding a given ion in the crystal lattice. It affects the overall stability and strength of the ionic structure.
Applications of Ionic Compounds: A World Shaped by Ions
Ionic compounds are ubiquitous, playing vital roles in various applications:
-
Medicine: Many medications are ionic compounds, including salts used to regulate electrolyte balance and drugs containing metal ions for therapeutic effects.
-
Industry: Ionic compounds are crucial in diverse industries, from fertilizers (e.g., potassium nitrate) to construction materials (e.g., gypsum).
-
Everyday Life: Table salt (NaCl), a quintessential ionic compound, is a staple in our kitchens. Other common examples include baking soda (sodium bicarbonate) and Epsom salts (magnesium sulfate).
Conclusion: A Powerful Bond with Wide-Reaching Implications
In essence, ionic bonding involves the complete transfer of electrons between atoms, resulting in the formation of positively and negatively charged ions. This electron transfer is driven by the desire of atoms to achieve a stable electron configuration (octet rule) and the interplay of electronegativity, ionization energy, and electron affinity. The resulting ionic compounds possess distinct properties, including high melting points, brittleness, solubility in polar solvents, and electrical conductivity when molten or dissolved. Understanding ionic bonding is not only essential for a fundamental grasp of chemistry but also crucial for appreciating the structure, properties, and applications of a vast array of materials that shape our world. From the salt on our tables to the complex compounds used in advanced technologies, the transfer of electrons in ionic bonds forms the foundation of much of the chemistry we encounter daily.
Latest Posts
Latest Posts
-
Conservative And Non Conservative Forces Examples
Apr 05, 2025
-
Sketch Each Line And Write The Equation
Apr 05, 2025
-
Identify The Oxidation Half Reaction Of Select One
Apr 05, 2025
-
Matter Is Conserved Physical Or Chemica Change
Apr 05, 2025
-
Difference Between Nuclear Reaction And Chemical Reaction
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about In An Ionic Bond Electrons Are . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
