Induced Fit Model Vs Lock And Key
Muz Play
Apr 02, 2025 · 6 min read
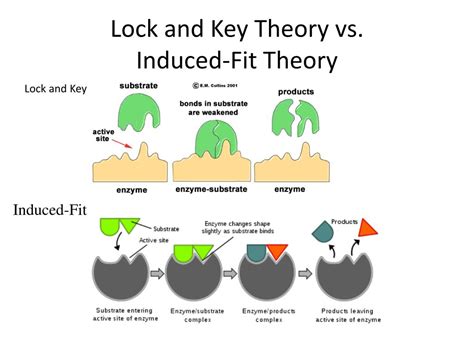
Table of Contents
Induced Fit vs. Lock and Key: A Deep Dive into Enzyme-Substrate Interactions
The interaction between enzymes and their substrates is a cornerstone of biochemistry, governing countless biological processes. For decades, the prevailing model explaining this interaction was the lock and key model. However, this model has since been refined and, in many cases, superseded by the induced fit model. While seemingly simple at first glance, the differences between these models are crucial to understanding enzyme specificity, catalytic efficiency, and regulation. This article will delve deep into both models, comparing and contrasting their mechanisms and highlighting their respective strengths and limitations.
The Lock and Key Model: A Rigid Approach
Proposed by Emil Fischer in 1894, the lock and key model presents a simple and intuitive analogy. It postulates that the enzyme's active site possesses a rigid, pre-formed structure perfectly complementary to the shape of its substrate. The substrate, like a key fitting into a lock, binds precisely to the active site, initiating the catalytic process.
Strengths of the Lock and Key Model:
- Simplicity and Intuitiveness: The model's clear analogy makes it easy to understand and visualize, serving as a useful introductory concept for students.
- Explains Specificity: The model effectively explains the high degree of specificity observed in many enzyme-substrate interactions. Only the correctly shaped substrate can "fit" into the enzyme's active site.
Limitations of the Lock and Key Model:
- Rigidity Assumption: The model's crucial flaw lies in its assumption of a rigid, unchanging enzyme structure. Numerous studies have demonstrated that enzyme active sites are highly flexible and dynamic, undergoing conformational changes upon substrate binding.
- Fails to Explain Transition State Stabilization: The lock and key model doesn't adequately explain how enzymes achieve such remarkable rate enhancements. It fails to account for the enzyme's role in stabilizing the high-energy transition state, a crucial step in catalysis.
- Insufficient Explanation for Enzyme Regulation: The model doesn't explain how enzyme activity can be regulated through allosteric mechanisms or covalent modifications, both of which involve changes in enzyme conformation.
The Induced Fit Model: A Dynamic Interaction
Proposed by Daniel Koshland in 1958, the induced fit model offers a more dynamic and accurate depiction of enzyme-substrate interactions. This model suggests that the enzyme's active site is flexible and undergoes conformational changes upon substrate binding. The substrate's binding induces a change in the enzyme's shape, creating a more complementary binding site that optimizes interactions and facilitates catalysis. This "induced fit" enhances the enzyme's ability to bind the substrate tightly and catalyze the reaction efficiently.
The Mechanism of Induced Fit:
- Initial Interaction: The substrate initially interacts with the enzyme's active site, which is not perfectly complementary in its unbound state.
- Conformational Change: This interaction triggers a conformational change in the enzyme's active site, adapting its shape to better accommodate the substrate.
- Tight Binding: The induced fit results in tighter binding of the substrate, maximizing interactions and positioning catalytic residues optimally.
- Catalysis: The enzyme catalyzes the reaction, converting the substrate into products.
- Product Release: The products are released, and the enzyme reverts to its original conformation, ready to bind another substrate.
Strengths of the Induced Fit Model:
- Explains Enzyme Flexibility: The model accounts for the inherent flexibility of enzymes, acknowledging that their active sites are not static structures.
- Transition State Stabilization: The induced fit process allows the enzyme to precisely stabilize the high-energy transition state of the reaction, significantly lowering the activation energy and accelerating the reaction rate.
- Accounts for Specificity and Regulation: The model elegantly explains how enzymes achieve substrate specificity and how their activity can be regulated through conformational changes influenced by allosteric effectors or covalent modifications.
- Better Representation of Reality: Experimental data, such as X-ray crystallography and molecular dynamics simulations, consistently support the induced fit model, demonstrating the dynamic nature of enzyme-substrate interactions.
Examples of Induced Fit in Action:
Many enzymes exhibit classic examples of induced fit. Hexokinase, for instance, undergoes a significant conformational change upon glucose binding, closing around the substrate to exclude water and create an optimal catalytic environment. Other examples include lysozyme, which changes its shape to encapsulate its bacterial cell wall substrate, and chymotrypsin, whose active site undergoes a dramatic reorganization upon substrate binding.
Comparing and Contrasting the Models:
| Feature | Lock and Key Model | Induced Fit Model |
|---|---|---|
| Enzyme Active Site | Rigid, pre-formed | Flexible, dynamic |
| Substrate Binding | Precise fit, like a key in a lock | Initial interaction triggers conformational change |
| Catalysis | Simple binding and reaction | Conformational change optimizes catalysis |
| Transition State | Does not adequately explain stabilization | Explains stabilization through induced fit |
| Enzyme Regulation | Does not readily explain regulation | Explains allosteric and covalent regulation |
| Accuracy | Limited accuracy | More accurate representation of reality |
The Significance of Understanding Enzyme-Substrate Interactions:
Understanding the intricacies of enzyme-substrate interactions is paramount for several reasons:
- Drug Development: The induced fit model provides a crucial framework for designing drugs that specifically target enzyme active sites. By understanding the dynamic nature of these interactions, scientists can develop more effective inhibitors that block enzyme activity and treat various diseases.
- Biotechnology: The principles of enzyme-substrate interactions are fundamental to various biotechnological applications, including enzyme engineering, biosensors, and biocatalysis.
- Fundamental Biological Understanding: Understanding how enzymes function at a molecular level is crucial to understanding fundamental biological processes, from metabolism to signal transduction.
Beyond the Binary: A Spectrum of Interactions
It's important to note that the induced fit and lock and key models are not mutually exclusive. Instead, they represent two ends of a spectrum. Some enzyme-substrate interactions may more closely resemble the lock and key model, exhibiting a high degree of pre-formed complementarity. However, most enzyme-substrate interactions show significant features of induced fit, demonstrating the dynamic nature of enzyme catalysis. The degree of conformational change upon binding varies among enzymes, reflecting the diverse mechanisms and functions of these biological catalysts.
Future Directions and Research:
Ongoing research continues to refine our understanding of enzyme-substrate interactions. Advanced techniques like molecular dynamics simulations, cryo-electron microscopy, and NMR spectroscopy provide increasingly detailed insights into the dynamic nature of these processes. This research is crucial for developing more effective therapeutic strategies and for advancing our fundamental understanding of biological processes. The focus is shifting toward understanding the detailed conformational changes and energetic landscapes involved in enzyme catalysis, moving beyond simple static models to a comprehensive understanding of the dynamic interplay between enzymes and substrates. The integration of computational methods with experimental approaches promises to unveil even more nuanced details about this crucial biological phenomenon.
Conclusion:
While the lock and key model provided a foundational understanding of enzyme specificity, the induced fit model offers a far more comprehensive and accurate description of enzyme-substrate interactions. This model, emphasizing the dynamic nature of enzyme active sites and the importance of conformational changes in catalysis, provides a powerful framework for understanding enzyme function, regulation, and its implication in various biological processes and biotechnological applications. The ongoing research utilizing cutting-edge technologies promises to further refine our understanding of this fascinating interplay and will undoubtedly revolutionize our ability to manipulate and utilize enzymes for various applications.
Latest Posts
Latest Posts
-
Cis 1 3 Dimethylcyclohexane Chair Conformation
Apr 03, 2025
-
What Does The Bacterial Chromosome Do
Apr 03, 2025
-
What Is The Difference Between Dehydration And Hydrolysis
Apr 03, 2025
-
What Is The Most Complex Level Of Organization
Apr 03, 2025
-
What Determines The Volume Of Gas
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Induced Fit Model Vs Lock And Key . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
