Is As A Metal Nonmetal Or Metalloid
Muz Play
Apr 02, 2025 · 6 min read
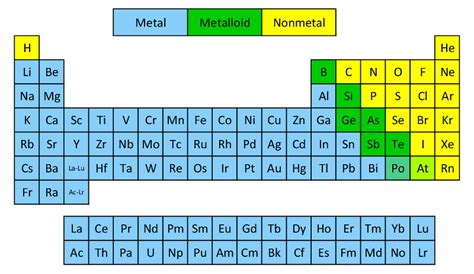
Table of Contents
Is Arsenic a Metal, Nonmetal, or Metalloid? Unveiling the Mysteries of a Semimetal
Arsenic, with its intriguing properties and complex chemistry, often sparks debates about its classification. Is it a metal, a nonmetal, or something in between? The answer, as we will explore in depth, lies in understanding its unique characteristics and positioning on the periodic table. This comprehensive guide will delve into the physical and chemical properties of arsenic, examine its position within the periodic table's metalloid family, and clarify why it's definitively classified as a metalloid.
Understanding the Periodic Table's Organization: Metals, Nonmetals, and Metalloids
The periodic table is organized to reflect the recurring trends in the properties of elements. These trends allow for the broad categorization of elements into three main groups: metals, nonmetals, and metalloids. Understanding these categories is crucial to grasping arsenic's classification.
Metals: The Characteristics of Metallic Elements
Metals are typically characterized by their:
- High electrical conductivity: They readily conduct electricity.
- High thermal conductivity: They efficiently transfer heat.
- Malleability: They can be hammered into thin sheets.
- Ductility: They can be drawn into wires.
- Luster: They possess a shiny appearance.
- High tensile strength: They are strong and resistant to breakage.
These properties stem from the structure of metallic bonding, where electrons are delocalized, allowing for easy movement and resulting in excellent conductivity. Examples of classic metals include iron, copper, gold, and silver.
Nonmetals: Opposites of Metallic Characteristics
Nonmetals, in contrast to metals, exhibit:
- Poor electrical conductivity: They generally do not conduct electricity.
- Poor thermal conductivity: They are poor conductors of heat.
- Brittleness: They tend to be brittle and easily shatter.
- Lack of luster: They typically lack a shiny appearance.
- Low tensile strength: They are generally weaker than metals.
These properties result from their strong covalent bonding, where electrons are tightly held within individual molecules, hindering their movement and conductivity. Common nonmetals include oxygen, nitrogen, chlorine, and sulfur.
Metalloids: Bridging the Gap Between Metals and Nonmetals
Metalloids, also known as semimetals, represent a fascinating middle ground. They possess properties that are intermediate between those of metals and nonmetals. This means their characteristics can vary significantly, depending on the specific element and the conditions under which they are examined. They are often semiconductors, meaning their electrical conductivity is somewhere between that of a conductor (metal) and an insulator (nonmetal). This conductivity is often temperature-dependent, increasing with increasing temperature. This unique characteristic makes them essential components in electronic devices.
Arsenic: A Detailed Examination of its Properties
Arsenic, situated in Group 15 (VA) of the periodic table, exhibits many of the characteristics of a metalloid. Let's delve into its key properties to understand its classification:
Physical Properties of Arsenic
- Appearance: Arsenic exists in several allotropic forms, but the most common is a greyish-white, crystalline solid. It's brittle and easily powdered.
- Conductivity: Arsenic is a semiconductor, meaning its electrical conductivity is significantly lower than that of metals like copper but higher than that of nonmetals like sulfur. Its conductivity is also temperature-dependent, increasing with higher temperatures.
- Density: Arsenic has a relatively high density compared to many nonmetals.
- Melting Point: Arsenic has a relatively low melting point compared to many metals.
- Boiling Point: Arsenic also has a relatively low boiling point.
Chemical Properties of Arsenic
- Reactivity: Arsenic is relatively unreactive at room temperature, but it becomes more reactive at higher temperatures.
- Oxidation States: Arsenic can exhibit several oxidation states, most commonly +3 and +5.
- Allotropes: Arsenic can exist in different forms or allotropes, each with slightly different properties. Grey arsenic is the most stable and common form.
- Compound Formation: Arsenic readily forms compounds with many other elements, particularly with halogens, oxygen, and sulfur.
- Toxicity: It's crucial to note the high toxicity of arsenic and its compounds, making careful handling absolutely necessary.
Why Arsenic is Classified as a Metalloid
Based on the properties described above, arsenic's classification as a metalloid is clear:
- Intermediate Conductivity: Arsenic's semiconducting nature, sitting between the excellent conductivity of metals and the poor conductivity of nonmetals, is a hallmark of metalloids.
- Brittleness: Its brittleness aligns more with nonmetals than with the malleability and ductility of metals.
- Appearance: Its greyish-white appearance, while not possessing the characteristic luster of many metals, is also not indicative of the typical appearance of nonmetals.
- Chemical Behavior: Its varied oxidation states and ability to form compounds with different elements reflect the diverse chemical reactivity characteristic of metalloids.
Arsenic's position in the periodic table, nestled between the metals and nonmetals, further supports its metalloid classification. It shares the periodic table's space with other well-established metalloids such as silicon, germanium, antimony, and tellurium, all exhibiting similar intermediate properties.
Applications of Arsenic and its Compounds
Despite its toxicity, arsenic and its compounds find applications in various fields, often leveraging its semiconducting properties or unique chemical reactivity:
- Semiconductors: Arsenic is used in the production of semiconductors, specifically in gallium arsenide (GaAs), which is used in high-speed electronics and optoelectronic devices such as lasers and LEDs.
- Alloys: Small quantities of arsenic are sometimes added to lead alloys to enhance their hardness and improve their resistance to corrosion.
- Agriculture (Historically): Arsenic compounds were formerly used as pesticides and herbicides, but their toxicity led to their widespread replacement by safer alternatives.
- Wood Preservatives (Historically): Arsenic-based wood preservatives were once common but have largely been phased out due to environmental concerns.
- Medicinal Uses (Historically): Arsenic compounds have been used in medicine throughout history, although their toxicity and side effects limit modern usage to very specific and carefully monitored cases.
Safety Precautions When Handling Arsenic
It is crucial to emphasize the inherent toxicity of arsenic. Its compounds are highly poisonous, and direct contact or ingestion can be lethal. Anyone handling arsenic or its compounds must take stringent safety precautions, including:
- Personal Protective Equipment (PPE): Always wear appropriate PPE, including gloves, eye protection, and respirators, when working with arsenic.
- Proper Ventilation: Ensure adequate ventilation to minimize inhalation risks.
- Waste Disposal: Dispose of arsenic waste according to regulations.
- Emergency Procedures: Have emergency response plans in place in case of spills or exposure.
Conclusion: Arsenic – A Definitive Metalloid
In conclusion, the evidence overwhelmingly supports the classification of arsenic as a metalloid. Its intermediate conductivity, brittle nature, chemical versatility, and position on the periodic table all align with the defining characteristics of metalloids. While its toxicity necessitates careful handling, understanding its properties and applications is vital in various scientific and technological fields. This comprehensive overview aimed to clear any ambiguity surrounding arsenic's classification and emphasize the importance of safe handling practices when dealing with this element.
Latest Posts
Latest Posts
-
How Many A Columns Are On The Periodic Table
Apr 03, 2025
-
Kelvin Celsius And Fahrenheit Are Three Types Of Scales
Apr 03, 2025
-
Difference Between Applied And Basic Science
Apr 03, 2025
-
Identify The Characteristics Of A Spontaneous Reaction
Apr 03, 2025
-
How To Find Average Acceleration From Acceleration Time Graph
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is As A Metal Nonmetal Or Metalloid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
