Is Hbr An Acid Or Base
Muz Play
Apr 05, 2025 · 5 min read
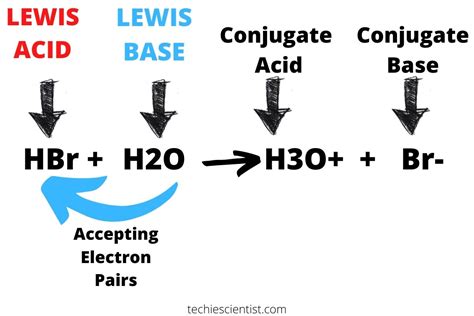
Table of Contents
Is HBr an Acid or Base? Understanding the Properties of Hydrobromic Acid
Hydrogen bromide (HBr), in its aqueous solution form, hydrobromic acid, is unequivocally an acid. This understanding stems from its behavior according to various definitions of acids and bases, most notably the Arrhenius, Brønsted-Lowry, and Lewis theories. This article delves deep into the properties of HBr, explaining why it's classified as a strong acid and exploring its reactions and applications.
Understanding Acid-Base Theories
Before definitively classifying HBr, it's crucial to understand the different acid-base theories that help us categorize substances.
Arrhenius Theory
The Arrhenius theory, one of the earliest acid-base theories, defines an acid as a substance that increases the concentration of hydrogen ions (H⁺) in an aqueous solution, while a base increases the hydroxide ion (OH⁻) concentration. HBr, when dissolved in water, readily dissociates into H⁺ and Br⁻ ions:
HBr(aq) → H⁺(aq) + Br⁻(aq)
This clear increase in H⁺ ion concentration firmly places HBr within the Arrhenius definition of an acid.
Brønsted-Lowry Theory
The Brønsted-Lowry theory offers a broader perspective. It defines an acid as a proton (H⁺) donor and a base as a proton acceptor. In the reaction of HBr with water:
HBr(aq) + H₂O(l) → H₃O⁺(aq) + Br⁻(aq)
HBr donates a proton (H⁺) to a water molecule (H₂O), forming a hydronium ion (H₃O⁺) and a bromide ion (Br⁻). This proton donation clearly showcases HBr's behavior as a Brønsted-Lowry acid.
Lewis Theory
The Lewis theory provides the most encompassing definition. It defines an acid as an electron-pair acceptor and a base as an electron-pair donor. While less directly applicable to HBr's acidic behavior than the Arrhenius and Brønsted-Lowry theories, the Lewis theory also supports its classification as an acid. The hydrogen ion (H⁺), formed upon dissociation, lacks an electron pair and readily accepts a pair of electrons from a base, making it a Lewis acid.
The Strength of Hydrobromic Acid
HBr is not merely an acid; it's a strong acid. This distinction highlights its complete dissociation in water. Unlike weak acids, which only partially dissociate, strong acids, such as HBr, virtually ionize completely. This complete dissociation means that in an aqueous solution of HBr, almost all HBr molecules break down into H⁺ and Br⁻ ions. This leads to a significantly higher concentration of H⁺ ions compared to weak acids at the same concentration.
Chemical Properties and Reactions
The highly acidic nature of HBr leads to several characteristic reactions:
Reaction with Metals
HBr reacts vigorously with many metals, producing hydrogen gas (H₂) and a metal bromide salt. For example, the reaction with zinc:
2HBr(aq) + Zn(s) → ZnBr₂(aq) + H₂(g)
This reaction is highly exothermic, releasing considerable heat.
Reaction with Metal Oxides and Hydroxides
HBr readily reacts with metal oxides and hydroxides, forming metal bromide salts and water. The reaction with magnesium oxide:
MgO(s) + 2HBr(aq) → MgBr₂(aq) + H₂O(l)
And the reaction with sodium hydroxide:
NaOH(aq) + HBr(aq) → NaBr(aq) + H₂O(l)
Reaction with Carbonates and Bicarbonates
HBr reacts with carbonates and bicarbonates, producing carbon dioxide gas (CO₂), water, and a metal bromide salt. For example, the reaction with calcium carbonate:
CaCO₃(s) + 2HBr(aq) → CaBr₂(aq) + CO₂(g) + H₂O(l)
Applications of Hydrobromic Acid
The strong acidic properties and high reactivity of HBr make it useful in various applications:
Industrial Applications
-
Production of Alkyl Bromides: HBr is a crucial reagent in the synthesis of alkyl bromides, important intermediates in the pharmaceutical and chemical industries.
-
Petroleum Refining: It's used in petroleum refining processes, primarily for the alkylation of isoparaffins.
-
Metal Cleaning: Its strong acidity makes it useful for cleaning metals and removing oxides.
Laboratory Applications
-
Analytical Chemistry: HBr is used in various analytical procedures, for example, to dissolve certain minerals and metals for analysis.
-
Organic Synthesis: As mentioned, its extensive use in organic chemistry spans a wide array of reactions, including bromination and the formation of various organic bromide compounds.
Safety Precautions
Because of its strong corrosive nature and potential for reactions with various substances, handling HBr requires strict safety precautions:
-
Protective Equipment: Always use appropriate protective equipment such as gloves, eye protection, and lab coats.
-
Ventilation: Adequate ventilation is essential to prevent inhalation of HBr fumes, which are extremely irritating and corrosive.
-
Storage: HBr should be stored in a cool, dry place, away from incompatible materials.
-
Spill Response: Have a spill response plan in place to safely handle any spills of hydrobromic acid.
Comparing HBr with other Acids
To further solidify the understanding of HBr's acidic properties, comparing it with other acids proves insightful:
HBr vs. HCl
Both HBr and HCl are strong acids, both exhibiting complete dissociation in water. However, HBr is generally considered a slightly stronger acid than HCl. This difference in strength stems from the slightly weaker H-Br bond compared to the H-Cl bond.
HBr vs. HF
While both are hydrohalic acids, HBr is a strong acid, whereas HF is a weak acid. This significant difference arises from the significantly stronger H-F bond, which reduces the extent of HF dissociation in water.
HBr vs. HI
Both HBr and HI are strong acids. HI is actually slightly stronger than HBr. This is again attributable to bond strength: the H-I bond is weaker than the H-Br bond.
Conclusion
In conclusion, HBr, in its aqueous form as hydrobromic acid, is unequivocally a strong acid. This classification is strongly supported by all major acid-base theories and is evident in its high reactivity and complete dissociation in water. Understanding its properties and safety precautions is paramount for its safe and effective use in various industrial and laboratory applications. The comparisons with other acids further highlight its unique position within the spectrum of acidic substances, emphasizing the importance of understanding the nuances of acid strength and reactivity. Always remember to prioritize safety when handling this corrosive and potent chemical.
Latest Posts
Latest Posts
-
Is Orange Juice An Acid Or Base
Apr 05, 2025
-
Which Is Smaller An Atom Or A Molecule
Apr 05, 2025
-
Stopping A Filibuster Requires That
Apr 05, 2025
-
What Are The Two Major Divisions Of The Skeletal System
Apr 05, 2025
-
Delta S Delta H Delta G Chart
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Is Hbr An Acid Or Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
