Is Osmotic Pressure A Colligative Property
Muz Play
Apr 05, 2025 · 6 min read
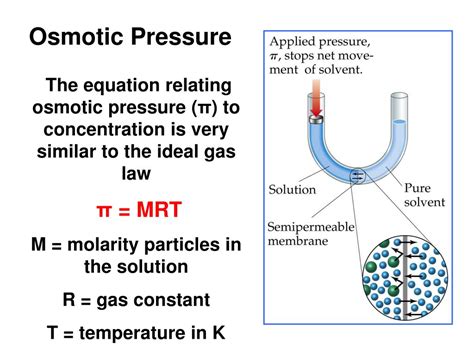
Table of Contents
Is Osmotic Pressure a Colligative Property? A Deep Dive
Osmotic pressure, a fundamental concept in chemistry and biology, plays a crucial role in various natural processes, from water transport in plants to maintaining cell integrity. But what exactly is osmotic pressure, and why is it considered a colligative property? This comprehensive article will delve into the intricacies of osmotic pressure, exploring its definition, its relationship to colligative properties, and its significance in different scientific fields.
Understanding Osmotic Pressure
Osmotic pressure is the minimum pressure needed to prevent the inward flow of water across a selectively permeable membrane. Imagine a membrane separating two solutions: one with a higher solute concentration (hypertonic) and another with a lower solute concentration (hypotonic). Water molecules naturally tend to move from a region of high water concentration (low solute concentration) to a region of low water concentration (high solute concentration) to equalize the concentration across the membrane. This movement of water is called osmosis.
Osmotic pressure is the force that opposes this osmotic flow. It's the pressure that must be applied to the hypertonic solution to prevent the net movement of water into it. The greater the difference in solute concentration across the membrane, the greater the osmotic pressure required to prevent osmosis.
Factors Affecting Osmotic Pressure
Several factors influence the magnitude of osmotic pressure:
-
Solute Concentration: The primary determinant of osmotic pressure is the concentration of solute particles in the solution. A higher concentration of solute particles leads to a higher osmotic pressure. This is directly proportional, meaning if you double the concentration, you double the osmotic pressure (at a constant temperature).
-
Temperature: Temperature also plays a role. Higher temperatures generally lead to increased kinetic energy of the solute particles, resulting in a higher osmotic pressure. The relationship is directly proportional as well.
-
Nature of the Solute: While the concentration of solute particles is the key, the nature of the solute (whether it dissociates into ions or remains as molecules) influences the effective number of particles. For example, 1 mole of NaCl dissociates into 2 moles of ions (Na+ and Cl-), exerting a greater osmotic pressure than 1 mole of glucose, which remains as a single molecule. This is why the term "osmolarity" is used – it accounts for both the molarity and the number of particles each molecule produces.
The Colligative Nature of Osmotic Pressure
The defining characteristic of a colligative property is its dependence solely on the number of solute particles in a solution, not on the identity or nature of those particles. The key is the concentration of solute particles, irrespective of what those particles are. Osmotic pressure perfectly fits this definition.
Here's why osmotic pressure qualifies as a colligative property:
-
Independent of Solute Identity: The osmotic pressure exerted by a 1 molar solution of glucose is different from that exerted by a 1 molar solution of sucrose, but if we consider a 1 molal solution of both glucose and sucrose, their osmotic pressure will be similar. This is because the osmotic pressure depends on the number of particles, not on their chemical nature.
-
Directly Proportional to Concentration: As mentioned earlier, increasing the concentration of solute particles directly increases the osmotic pressure. This proportionality is a hallmark of colligative properties. The Van't Hoff equation, Π = iMRT, mathematically demonstrates this relationship, where:
- Π = osmotic pressure
- i = Van't Hoff factor (accounts for dissociation)
- M = molar concentration
- R = ideal gas constant
- T = absolute temperature
-
Dependence on Particle Number: The Van't Hoff factor (i) in the equation above elegantly highlights this point. For non-electrolytes like glucose, i = 1, while for strong electrolytes like NaCl, i = 2. This reflects the fact that the osmotic pressure depends on the total number of particles in solution, accounting for dissociation.
Distinguishing Osmotic Pressure from Other Colligative Properties
Osmotic pressure is one of four major colligative properties. The others are:
-
Vapor Pressure Lowering: The presence of a non-volatile solute reduces the vapor pressure of the solvent.
-
Boiling Point Elevation: Adding a non-volatile solute raises the boiling point of the solvent.
-
Freezing Point Depression: Adding a non-volatile solute lowers the freezing point of the solvent.
While all four properties depend on the number of solute particles, they manifest differently and are measured differently. Osmotic pressure focuses on the pressure required to prevent osmosis across a semi-permeable membrane, whereas the others deal with changes in the physical properties of the solvent (vapor pressure, boiling point, freezing point). The underlying principle, however, remains the same – the influence of the number of solute particles.
Applications of Osmotic Pressure
The principle of osmotic pressure has numerous applications across various fields:
Biology and Medicine:
-
Water Regulation in Cells: Osmotic pressure maintains the proper balance of water and electrolytes within cells. Changes in osmotic pressure can lead to cell shrinkage (crenation) or swelling (lysis).
-
Kidney Function: The kidneys play a crucial role in regulating osmotic pressure by controlling the excretion of water and solutes in urine.
-
Intravenous Solutions: Intravenous fluids are carefully formulated to match the osmotic pressure of blood plasma to prevent adverse effects on blood cells. Isotonic solutions have the same osmotic pressure as blood, hypotonic solutions have lower osmotic pressure, and hypertonic solutions have higher osmotic pressure.
-
Drug Delivery: Osmotic pressure is utilized in certain drug delivery systems to control the release of medication.
Chemistry and Engineering:
-
Desalination: Reverse osmosis, a process that uses high pressure to force water across a semi-permeable membrane, is used for desalination of seawater.
-
Water Purification: Osmotic pressure principles are involved in various water purification technologies.
-
Food Preservation: Osmotic pressure plays a role in food preservation techniques like pickling and drying.
Measuring Osmotic Pressure
Precise measurement of osmotic pressure can be challenging. Several methods exist, each with its advantages and limitations:
-
Direct Measurement: Involves applying pressure to a solution until there is no net movement of water across a semi-permeable membrane. This is often difficult to achieve in practice due to the high pressures often involved.
-
Indirect Measurement: Based on measuring colligative properties like freezing point depression or boiling point elevation, which are related to osmotic pressure through thermodynamic relationships. This is a common method due to its relative simplicity.
-
Specialized Instruments: Instrumentation like osmometers allows for accurate determination of osmotic pressure, using techniques that measure changes in physical properties related to the osmotic pressure.
The Importance of the Van't Hoff Equation
The Van't Hoff equation, Π = iMRT, is crucial for understanding and calculating osmotic pressure. It shows the direct proportionality between osmotic pressure and the molar concentration of solute particles (M), temperature (T), and the Van't Hoff factor (i). The ideal gas constant (R) is a fundamental constant. This equation allows us to predict and understand the behavior of solutions in relation to osmotic pressure.
Conclusion
Osmotic pressure is undeniably a colligative property. Its dependence solely on the number of solute particles, independent of their identity, is fundamental to its classification. The Van't Hoff equation perfectly encapsulates this relationship and allows for quantitative analysis. Understanding osmotic pressure is critical in various fields, from biology and medicine to chemistry and engineering, highlighting its importance in diverse applications and scientific processes. Its significance underscores the profound impact of colligative properties on our understanding of solutions and their behavior. Further research continues to explore the nuances of osmotic pressure and its implications in various contexts, solidifying its role as a cornerstone concept in many scientific disciplines.
Latest Posts
Latest Posts
-
How To Calculate Break Even Sales Dollars
Apr 05, 2025
-
Volume Of A Solid With A Known Cross Section
Apr 05, 2025
-
Bacterial Growth Curves Experiment With Bacterial Growth
Apr 05, 2025
-
What Is A Secondary Alkyl Halide
Apr 05, 2025
-
Is Oil And Water A Heterogeneous Mixture
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Is Osmotic Pressure A Colligative Property . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
