Liquid To Gas Endothermic Or Exothermic
Muz Play
Apr 03, 2025 · 5 min read
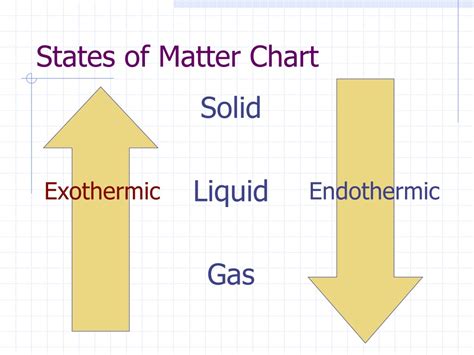
Table of Contents
Liquid to Gas: Endothermic or Exothermic? Understanding Phase Transitions
The transition of a substance from a liquid state to a gaseous state, a process commonly known as vaporization, is a fundamental concept in chemistry and physics. Understanding whether this transition is endothermic or exothermic is crucial for grasping many everyday phenomena, from boiling water to the evaporation of sweat. This comprehensive article will delve into the intricacies of liquid-to-gas phase transitions, clarifying the energy dynamics involved and exploring various real-world applications.
Understanding Endothermic and Exothermic Reactions
Before diving into the specifics of vaporization, let's briefly review the definitions of endothermic and exothermic processes.
-
Endothermic processes: These processes absorb heat from their surroundings. The system's energy increases, resulting in a decrease in the temperature of the surroundings. Think of it like a sponge soaking up water – the sponge (system) gains energy, and the water (surroundings) loses energy.
-
Exothermic processes: These processes release heat into their surroundings. The system's energy decreases, causing an increase in the temperature of the surroundings. Consider a burning candle – the candle (system) releases energy as heat and light, warming the surrounding air.
Liquid to Gas: The Endothermic Nature of Vaporization
The process of a liquid turning into a gas, whether through boiling, evaporation, or sublimation (solid directly to gas), is invariably endothermic. This means that the process absorbs heat from its surroundings.
Why is Vaporization Endothermic?
At the molecular level, the molecules in a liquid are relatively close together, held by intermolecular forces (like van der Waals forces, hydrogen bonds, or dipole-dipole interactions). To transition to the gaseous state, these molecules need to overcome these attractive forces and move further apart, requiring an input of energy. This energy is absorbed from the surroundings in the form of heat.
Imagine the molecules in a liquid as magnets clinging together. To separate them, you need to apply energy (heat) to overcome the magnetic attraction. Once separated, they become gas molecules moving freely and independently.
Boiling Point and Vaporization
The boiling point of a liquid is the temperature at which its vapor pressure equals the external pressure. At this point, the liquid rapidly transitions to a gas. The energy required to reach the boiling point and subsequently vaporize the liquid is directly related to the strength of the intermolecular forces. Liquids with stronger intermolecular forces require more energy to vaporize and thus have higher boiling points.
Evaporation: A Gradual Process
Evaporation is a form of vaporization that occurs at temperatures below the boiling point. Even at room temperature, some liquid molecules possess enough kinetic energy to overcome the intermolecular forces and escape into the gaseous phase. This process is also endothermic, as it still requires energy input to break the intermolecular bonds.
Factors Affecting Vaporization
Several factors can influence the rate of vaporization:
-
Temperature: Higher temperatures lead to faster vaporization rates because more molecules have sufficient kinetic energy to escape.
-
Surface area: A larger surface area allows more molecules to escape simultaneously, accelerating the process.
-
Intermolecular forces: Stronger intermolecular forces result in slower vaporization rates.
-
Air pressure: Lower air pressure reduces the resistance to escaping molecules, leading to faster vaporization.
Real-World Examples of Endothermic Vaporization
The endothermic nature of vaporization is evident in various everyday occurrences:
-
Sweating: Our bodies utilize evaporation as a cooling mechanism. Sweat, primarily water, absorbs heat from the skin as it evaporates, leaving the skin cooler.
-
Boiling water: Heating water to its boiling point requires energy input, and the subsequent vaporization (boiling) further absorbs heat from the heat source.
-
Refrigeration: Refrigerants absorb heat as they vaporize, allowing them to cool the interior of a refrigerator or air conditioner.
-
Drying clothes: Clothes dry because water molecules in the fabric absorb heat and evaporate, leaving behind dry clothes.
-
Formation of clouds: Water vapor in the atmosphere condenses to form clouds, a process that releases heat. The initial evaporation of water from bodies of water is an endothermic process.
Applications and Implications
The understanding of endothermic vaporization has crucial implications in various fields:
-
Chemical engineering: Designing efficient distillation columns and other separation processes requires a thorough understanding of the energy requirements for vaporization.
-
Meteorology: Predicting weather patterns and understanding climate change necessitates accurately modeling the evaporation rates from various water bodies.
-
Material science: Understanding vaporization helps in designing materials with specific properties, such as those resistant to evaporation or with controlled vaporization rates.
-
Biological systems: Many biological processes, including photosynthesis and respiration, involve phase transitions and require the consideration of heat transfer.
Distinguishing Vaporization from Condensation
It's important to distinguish between vaporization (liquid to gas) and condensation (gas to liquid). Condensation is an exothermic process, releasing heat as gas molecules lose kinetic energy and come together to form a liquid. The energy released during condensation is equivalent to the energy absorbed during vaporization. This highlights the reversible nature of phase transitions and the conservation of energy.
Conclusion
The transition of a liquid to a gas is an endothermic process, requiring an input of energy to overcome the intermolecular forces holding the liquid molecules together. This fundamental principle governs numerous natural phenomena and industrial processes. Understanding the endothermic nature of vaporization is critical for grasping various aspects of chemistry, physics, biology, and engineering. From the cooling effect of sweat to the operation of refrigerators, the implications of endothermic vaporization are far-reaching and essential to our daily lives. This knowledge serves as a cornerstone for further exploration into the complexities of phase transitions and thermodynamics. The ability to accurately predict and control vaporization rates has profound implications for various industries and scientific advancements, emphasizing the significance of this seemingly simple process. Future research in this area promises to unravel even more intricacies of this crucial phase transition, leading to further technological advancements and a deeper understanding of the natural world.
Latest Posts
Latest Posts
-
An Unsaturated Fatty Acid Resulting From Hydrogenation Is Known As
Apr 04, 2025
-
What Are The Elements That Make Up Salt
Apr 04, 2025
-
Ionic Compound For Sodium And Sulfur
Apr 04, 2025
-
Cells Are Basic Unit Of Life
Apr 04, 2025
-
Two Bones That Form The Nasal Septum
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Liquid To Gas Endothermic Or Exothermic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
