Molal Boiling Point Elevation Constant Table
Muz Play
Apr 02, 2025 · 6 min read
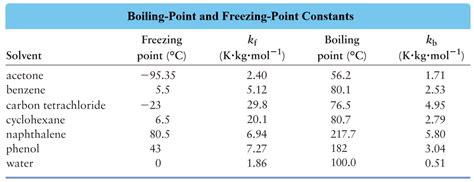
Table of Contents
Molal Boiling Point Elevation Constant Table: A Comprehensive Guide
The molal boiling point elevation constant, often denoted as K<sub>b</sub>, is a crucial colligative property in chemistry. It quantifies the extent to which the boiling point of a solvent is elevated when a non-volatile solute is added. Understanding K<sub>b</sub> is essential for various applications, from determining molar mass to predicting boiling points of solutions. This comprehensive guide delves into the intricacies of K<sub>b</sub>, exploring its definition, calculation, applications, and providing a detailed table of values for common solvents.
Understanding Colligative Properties and Boiling Point Elevation
Colligative properties are properties of solutions that depend on the concentration of solute particles, not their identity. These properties include boiling point elevation, freezing point depression, osmotic pressure, and vapor pressure lowering. Boiling point elevation occurs because the addition of a non-volatile solute reduces the escaping tendency of solvent molecules, thereby requiring a higher temperature to achieve the vapor pressure equal to atmospheric pressure – the boiling point.
The key factors influencing boiling point elevation are:
- The nature of the solvent: Different solvents possess different intermolecular forces, affecting their boiling points and consequently their K<sub>b</sub> values. A solvent with stronger intermolecular forces will generally have a lower K<sub>b</sub>.
- The concentration of the solute: The elevation in boiling point is directly proportional to the molality (moles of solute per kilogram of solvent) of the solution. This relationship is described by the equation:
ΔT<sub>b</sub> = K<sub>b</sub> * m
Where:
- ΔT<sub>b</sub> is the boiling point elevation (in °C or K)
- K<sub>b</sub> is the molal boiling point elevation constant (in °C kg/mol or K kg/mol)
- m is the molality of the solution (in mol/kg)
This equation highlights the linear relationship between molality and boiling point elevation. Doubling the molality will double the boiling point elevation.
Calculating the Molal Boiling Point Elevation Constant (K<sub>b</sub>)
While experimental determination is often necessary, K<sub>b</sub> can be theoretically estimated using thermodynamic principles. However, precise calculations require advanced knowledge of thermodynamic properties and are beyond the scope of this basic guide. Instead, we will focus on the practical application of already determined K<sub>b</sub> values.
Experimental Determination: K<sub>b</sub> is typically determined experimentally through the following steps:
- Precise measurement of the boiling point of the pure solvent.
- Preparation of solutions with known molalities of a non-volatile solute.
- Measurement of the boiling points of these solutions.
- Plotting a graph of ΔT<sub>b</sub> (change in boiling point) versus molality (m). The slope of this graph will be equal to K<sub>b</sub>.
Applications of the Molal Boiling Point Elevation Constant
The K<sub>b</sub> value finds applications in diverse areas of chemistry and related fields:
-
Determining the molar mass of an unknown solute: By measuring the boiling point elevation of a solution with a known mass of solute, one can calculate the molar mass of the solute using the boiling point elevation equation and the known K<sub>b</sub> value of the solvent. This technique is particularly useful for determining the molar mass of non-volatile, non-electrolyte solutes.
-
Predicting boiling points of solutions: Knowing the K<sub>b</sub> of a solvent and the molality of a solute, one can accurately predict the boiling point of the resulting solution. This is invaluable in various industrial processes and laboratory settings.
-
Understanding solute-solvent interactions: While primarily focusing on concentration, K<sub>b</sub> can indirectly provide insights into solute-solvent interactions. Significant deviations from the expected boiling point elevation might suggest strong interactions between the solute and solvent.
-
Solvent selection in chemical processes: The choice of solvent for a particular application often considers its boiling point and K<sub>b</sub> value. A solvent with a suitable K<sub>b</sub> can optimize the efficiency and outcome of the process.
Table of Molal Boiling Point Elevation Constants (K<sub>b</sub>) for Common Solvents
The following table lists the molal boiling point elevation constants for several common solvents. Remember that these values are approximate and can vary slightly depending on the experimental conditions and the source of the data. It's crucial to use the most accurate value available for your specific application.
| Solvent | Chemical Formula | K<sub>b</sub> (°C kg/mol) | Boiling Point (°C) |
|---|---|---|---|
| Water | H₂O | 0.512 | 100 |
| Benzene | C₆H₆ | 2.53 | 80.1 |
| Ethanol | C₂H₅OH | 1.22 | 78.4 |
| Chloroform | CHCl₃ | 3.63 | 61.2 |
| Carbon Tetrachloride | CCl₄ | 5.03 | 76.7 |
| Acetic Acid | CH₃COOH | 3.07 | 118.1 |
| Acetone | (CH₃)₂CO | 1.71 | 56.1 |
| Methanol | CH₃OH | 0.83 | 64.7 |
| Diethyl Ether | (C₂H₅)₂O | 2.02 | 34.6 |
| Cyclohexane | C₆H₁₂ | 2.79 | 80.7 |
| Toluene | C₇H₈ | 3.33 | 110.6 |
Important Considerations:
- Units: Ensure consistent units throughout your calculations. Molality should be in mol/kg, and K<sub>b</sub> should have the corresponding units (°C kg/mol or K kg/mol).
- Ideal Solutions: The equations for boiling point elevation assume ideal solutions, meaning there are minimal interactions between solute and solvent molecules. In real-world scenarios, deviations from ideality might occur.
- Electrolytes: The equations discussed here apply primarily to non-electrolyte solutes. For electrolyte solutes, the van't Hoff factor (i) needs to be incorporated into the equation to account for the dissociation of the solute into ions, impacting the effective concentration of particles. The modified equation becomes: ΔT<sub>b</sub> = i * K<sub>b</sub> * m
Advanced Topics and Further Exploration
For a more in-depth understanding of boiling point elevation and molal boiling point elevation constants, further exploration into the following topics is recommended:
- Thermodynamics of Solutions: Understanding the thermodynamic principles underlying colligative properties provides a more rigorous foundation for understanding boiling point elevation.
- Activity Coefficients: For non-ideal solutions, activity coefficients need to be considered to accurately predict boiling point elevation.
- Van't Hoff Factor and Electrolytes: A thorough understanding of the van't Hoff factor is critical for accurate calculations involving electrolyte solutions.
- Experimental Techniques: Learning about various experimental methods for determining K<sub>b</sub> and boiling points expands practical knowledge.
This comprehensive guide provides a solid foundation for understanding the molal boiling point elevation constant. By mastering this concept and utilizing the provided table of values, you can effectively solve problems related to boiling point elevation, determine molar masses, and predict the behavior of solutions in various applications. Remember always to consult reliable sources and use the most accurate K<sub>b</sub> values available for your specific solvent and conditions. Further exploration of the advanced topics mentioned will enhance your understanding and allow for more complex calculations and analysis.
Latest Posts
Latest Posts
-
Finding Domain Of A Composite Function
Apr 03, 2025
-
Image Formation In A Plane Mirror
Apr 03, 2025
-
Us Air Force Tongue And Quill
Apr 03, 2025
-
The Coefficients In A Chemical Equation Represent The
Apr 03, 2025
-
How To Calculate The Gradient Of A Stream
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Molal Boiling Point Elevation Constant Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
