Monomers Are Connected In What Type Of Reaction
Muz Play
Apr 02, 2025 · 6 min read
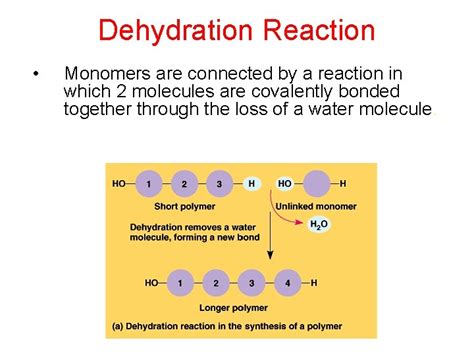
Table of Contents
Monomers are Connected in What Type of Reaction? Understanding Polymerization
Polymerization, the process of connecting monomers to form polymers, is fundamental to materials science and biology. Understanding the types of reactions involved is crucial for manipulating material properties and comprehending biological processes. This article delves deep into the various types of polymerization reactions, exploring their mechanisms, characteristics, and applications.
What are Monomers and Polymers?
Before we dive into the reactions, let's define our key players:
-
Monomers: These are small, repeating molecular units that act as the building blocks of polymers. They can be simple molecules, like ethylene (CH₂=CH₂), or complex ones, like amino acids. The crucial characteristic of a monomer is its ability to react with other monomers to form a chain.
-
Polymers: These are large molecules composed of many repeating monomer units linked together. These chains can be linear, branched, or cross-linked, leading to a wide array of material properties. Examples of polymers include plastics (polyethylene, polypropylene), natural rubber, proteins, and DNA.
The Key Reaction: Polymerization
The process of joining monomers to form polymers is called polymerization. This isn't a single reaction type but rather a broad category encompassing several distinct mechanisms. We can broadly classify polymerization reactions into two major groups:
1. Addition Polymerization (Chain-Growth Polymerization):
This type of polymerization involves the sequential addition of monomers to a growing chain without the loss of any atoms. It's characterized by a fast reaction rate and often requires an initiator to start the chain reaction. Several key steps are involved:
-
Initiation: An initiator, typically a free radical, cation, or anion, reacts with a monomer, creating an active center (a radical, cation, or anion) on one end of the monomer. This activated monomer is now ready to react with another monomer. Examples of initiators include peroxides (free radical), Lewis acids (cationic), and organometallic compounds (anionic).
-
Propagation: The active center on the initiated monomer attacks another monomer, adding it to the growing chain and regenerating the active center at the end of the chain. This step repeats many times, rapidly increasing the chain length.
-
Termination: The propagation step continues until the chain reaction is terminated. This can occur through several mechanisms, depending on the type of polymerization:
- Radical Termination: Two growing chains can combine, or a radical can react with an inhibitor or transfer agent, ending the propagation.
- Ionic Termination: Ionic termination often involves reaction with impurities or counterions.
Characteristics of Addition Polymerization:
- High molecular weight polymers: This process typically leads to polymers with high molecular weights.
- Fast reaction rates: The chain reaction nature leads to rapid polymer formation.
- Requires an initiator: An initiator is necessary to start the chain reaction.
- Monomers usually have double or triple bonds: The unsaturated nature of the monomers allows for the addition reaction to occur.
Examples of Addition Polymers:
- Polyethylene (PE): Made from ethylene monomers. Used in plastic bags, bottles, and films.
- Polypropylene (PP): Made from propylene monomers. Used in packaging, fibers, and containers.
- Polyvinyl chloride (PVC): Made from vinyl chloride monomers. Used in pipes, flooring, and window frames.
- Polystyrene (PS): Made from styrene monomers. Used in packaging, insulation, and disposable tableware.
- Polytetrafluoroethylene (PTFE) (Teflon): Made from tetrafluoroethylene monomers. Known for its non-stick properties and high chemical resistance.
2. Condensation Polymerization (Step-Growth Polymerization):
In contrast to addition polymerization, condensation polymerization involves the joining of monomers with the elimination of a small molecule, typically water or methanol. The reaction occurs between functional groups on the monomers, and the molecular weight increases gradually.
Mechanism:
Unlike the chain reaction in addition polymerization, condensation polymerization proceeds through a stepwise mechanism. Each step involves a reaction between two monomers or growing chains, forming a new bond and releasing a small molecule. This process continues until the desired molecular weight is reached.
Characteristics of Condensation Polymerization:
- Stepwise growth: The molecular weight increases gradually.
- No initiator required: The reaction proceeds without an initiator.
- Functional groups are essential: Monomers must possess reactive functional groups (e.g., -OH, -COOH, -NH₂) capable of reacting with each other.
- Lower reaction rates compared to addition polymerization: The reaction is slower than addition polymerization.
- By-product formation: A small molecule, such as water or methanol, is eliminated during each step.
Examples of Condensation Polymers:
- Polyesters: Formed from the reaction of diols and diacids. Used in clothing fibers (polyester), bottles (PET), and films.
- Polyamides (Nylons): Formed from the reaction of diamines and diacids. Used in textiles, fibers, and engineering plastics.
- Polyurethanes: Formed from the reaction of diisocyanates and diols. Used in foams, coatings, and elastomers.
- Polycarbonates: Formed from the reaction of bisphenols and phosgene. Used in safety glasses, compact discs, and automotive parts.
Comparing Addition and Condensation Polymerization:
| Feature | Addition Polymerization | Condensation Polymerization |
|---|---|---|
| Mechanism | Chain-growth | Step-growth |
| Monomer type | Usually unsaturated monomers | Monomers with functional groups |
| Initiator | Required | Not required |
| By-product | None | Small molecule (e.g., water, methanol) |
| Reaction rate | Fast | Slow |
| Molecular weight | High molecular weight polymers | Gradual increase of molecular weight |
Other Types of Polymerization:
Besides addition and condensation polymerization, other types exist, each with unique characteristics and applications:
-
Ring-opening polymerization: This involves the opening of cyclic monomers to form linear polymers. Examples include the polymerization of caprolactone to form polycaprolactone, a biodegradable polymer.
-
Living polymerization: This allows for precise control over polymer chain length and architecture. Living polymerizations are characterized by the absence of termination steps, enabling the synthesis of well-defined polymers with narrow molecular weight distributions. Anionic and cationic polymerizations can be controlled to create living polymerization conditions.
-
Solid-state polymerization: This occurs in the solid state without a solvent, offering advantages in terms of reduced waste and energy consumption.
Applications of Polymerization:
The ability to create polymers with tailored properties through polymerization has revolutionized various industries:
-
Plastics Industry: A vast range of plastics, from flexible films to rigid containers, are produced through polymerization.
-
Textiles: Polymers such as nylon, polyester, and acrylic are used to create synthetic fibers for clothing and other applications.
-
Biomedical Engineering: Biocompatible and biodegradable polymers are used in drug delivery systems, implants, and tissue engineering.
-
Coatings and Adhesives: Polymers are used in various coatings, adhesives, and sealants, providing protection, bonding, and sealing properties.
Conclusion:
Polymerization, encompassing addition and condensation reactions, is a cornerstone of materials science and biotechnology. Understanding the different types of polymerization reactions and their mechanisms is crucial for developing new materials with tailored properties and for advancing our knowledge of biological systems. The versatility and widespread applications of polymers highlight the importance of this fundamental chemical process. Further research and development in polymerization techniques continue to open up new possibilities for innovation across various fields.
Latest Posts
Latest Posts
-
Does Higher Bond Order Mean Stronger Bond
Apr 03, 2025
-
Does Sohcahtoa Work On Non Right Triangles
Apr 03, 2025
-
What Are The Four Main Types Of Context
Apr 03, 2025
-
How To Know If A Graph Is Symmetric
Apr 03, 2025
-
Is Blood Clotting Negative Or Positive Feedback
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Monomers Are Connected In What Type Of Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
