Most Likely Cations And Anions On The Periodic Table
Muz Play
Apr 04, 2025 · 6 min read
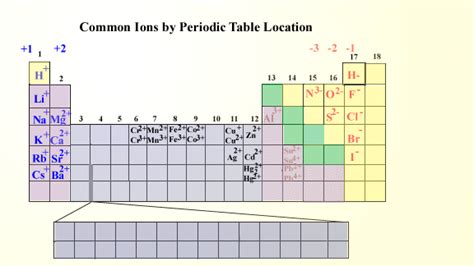
Table of Contents
Most Likely Cations and Anions on the Periodic Table: A Comprehensive Guide
Understanding cations and anions is fundamental to grasping the behavior of elements and compounds in chemistry. This comprehensive guide delves into the periodic trends that determine which elements are most likely to form positive ions (cations) and negative ions (anions). We'll explore the relationship between electron configuration, electronegativity, and the formation of ionic compounds, providing you with a robust understanding of this crucial chemical concept.
Understanding Ions: Cations and Anions
Before diving into specific elements, let's clarify the core concepts. An ion is an atom or molecule that carries a net electrical charge. This charge arises from a gain or loss of electrons.
-
Cations: These are positively charged ions, formed when an atom loses one or more electrons. The loss of negatively charged electrons leaves the atom with a net positive charge.
-
Anions: These are negatively charged ions, formed when an atom gains one or more electrons. The addition of negatively charged electrons gives the atom a net negative charge.
Periodic Trends Predicting Ion Formation
The periodic table is our invaluable tool for predicting the likelihood of an element forming a cation or an anion. Several key periodic trends guide this prediction:
1. Electronegativity
Electronegativity measures an atom's ability to attract electrons within a chemical bond. Elements with high electronegativity tend to attract electrons strongly, making them more likely to form anions. Conversely, elements with low electronegativity are more likely to lose electrons and form cations. Electronegativity generally increases across a period (from left to right) and decreases down a group (from top to bottom).
2. Ionization Energy
Ionization energy is the energy required to remove an electron from a neutral atom. Elements with low ionization energies readily lose electrons, favoring cation formation. Ionization energy generally increases across a period and decreases down a group, mirroring the electronegativity trend.
3. Electron Affinity
Electron affinity represents the energy change when an atom gains an electron. Elements with high electron affinities readily accept electrons, favoring anion formation. Electron affinity trends are more complex than electronegativity and ionization energy, but generally show an increase across periods and a less consistent trend down groups.
Most Likely Cations: A Group-by-Group Analysis
Let's examine the groups most likely to form cations:
Group 1 (Alkali Metals): The Eager Electron Donors
The alkali metals (Li, Na, K, Rb, Cs, Fr) are renowned for their extreme reactivity. They possess only one electron in their outermost shell, which they readily lose to achieve a stable noble gas configuration. This makes them highly likely to form +1 cations. For example:
- Sodium (Na): Loses one electron to become Na⁺.
- Potassium (K): Loses one electron to become K⁺.
Group 2 (Alkaline Earth Metals): Reluctant, but Still Willing
Alkaline earth metals (Be, Mg, Ca, Sr, Ba, Ra) have two electrons in their outermost shell. While slightly less reactive than alkali metals, they still readily lose these two electrons to achieve a stable noble gas configuration, forming +2 cations. Examples include:
- Magnesium (Mg): Loses two electrons to become Mg²⁺.
- Calcium (Ca): Loses two electrons to become Ca²⁺.
Transition Metals: A Variety of Charges
Transition metals (located in the d-block) exhibit a much wider range of possible charges. Their variable oxidation states arise from the involvement of both (n-1)d and ns electrons in bonding. This leads to the formation of cations with various charges, depending on the specific element and its chemical environment. Common examples include:
- Iron (Fe): Can form Fe²⁺ and Fe³⁺ ions.
- Copper (Cu): Can form Cu⁺ and Cu²⁺ ions.
- Zinc (Zn): Typically forms Zn²⁺ ions.
Post-Transition Metals: A More Limited Range
Post-transition metals (like aluminum, tin, and lead) usually form cations, but their charge is often more predictable than transition metals. Aluminum typically forms Al³⁺, while tin and lead can form both +2 and +4 cations.
Most Likely Anions: Focusing on Nonmetals
Now let's focus on the elements that most readily form anions:
Group 17 (Halogens): The Electron Grabbers
Halogens (F, Cl, Br, I, At) are highly electronegative and possess seven electrons in their outermost shell. They readily gain one electron to achieve a stable noble gas configuration, forming -1 anions (halide ions). For example:
- Chlorine (Cl): Gains one electron to become Cl⁻.
- Fluorine (F): Gains one electron to become F⁻.
Group 16 (Chalcogens): Often Gaining Two Electrons
Chalcogens (O, S, Se, Te, Po) have six electrons in their outermost shell. They typically gain two electrons to achieve a stable noble gas configuration, forming -2 anions. Key examples include:
- Oxygen (O): Gains two electrons to become O²⁻.
- Sulfur (S): Gains two electrons to become S²⁻.
Group 15 (Pnictogens): Less Common Anion Formation
Elements in group 15 (N, P, As, Sb, Bi) have five valence electrons. While they can form anions, it's less common than for group 16 and 17 elements. Nitrogen commonly forms nitride (N³⁻) ions, particularly in compounds with highly electropositive metals. Phosphorus can also form phosphide (P³⁻) ions, but less readily than nitrogen.
Polyatomic Ions: Beyond Single Atoms
Many ions are not single atoms but rather groups of atoms covalently bonded together carrying a net charge. These are called polyatomic ions. Examples include:
- Nitrate (NO₃⁻): A common polyatomic anion found in many fertilizers.
- Sulfate (SO₄²⁻): Another frequently encountered polyatomic anion.
- Phosphate (PO₄³⁻): Crucial for biological systems and found in many minerals.
- Ammonium (NH₄⁺): A relatively uncommon polyatomic cation.
Predicting Ionic Compound Formation
The principles discussed above allow us to predict the formation of ionic compounds. Ionic compounds are formed through the electrostatic attraction between cations and anions. The charges of the ions dictate the formula of the compound. For instance:
- Sodium chloride (NaCl): Na⁺ + Cl⁻ → NaCl
- Magnesium oxide (MgO): Mg²⁺ + O²⁻ → MgO
- Aluminum oxide (Al₂O₃): 2Al³⁺ + 3O²⁻ → Al₂O₃ (Balancing the charges is crucial)
Exceptions and Complexities
While periodic trends provide excellent predictive power, exceptions exist. Factors like the size of the ions, their polarizability, and the surrounding chemical environment can influence ion formation. Some transition metals exhibit a wide range of oxidation states, making their behavior less predictable.
Conclusion: Mastering Cations and Anions
Understanding the likelihood of different elements forming cations or anions is critical for comprehending chemical reactions and predicting the properties of compounds. By leveraging the periodic trends of electronegativity, ionization energy, and electron affinity, we can make informed predictions about ionic compound formation. While exceptions exist, this foundational knowledge provides a robust framework for navigating the fascinating world of ions and their role in chemistry. Remember to always consider the specific chemical context when analyzing the behavior of elements and predicting ion formation. Further research into specific elements and their unique properties will further enhance your understanding of this fundamental chemical concept.
Latest Posts
Latest Posts
-
What Does The Nature Of A Business Mean
Apr 04, 2025
-
Most Of The Oxygen In The Blood Is Transported By
Apr 04, 2025
-
When Does Dna Replication Occur In A Eukaryotic Cell
Apr 04, 2025
-
What Was Shown By Both Redis And Pasteurs Experiments
Apr 04, 2025
-
The Elements In Group 1 Are Called The
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Most Likely Cations And Anions On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
