Number Of Valence Electrons In Alkali Metals
Muz Play
Apr 05, 2025 · 6 min read
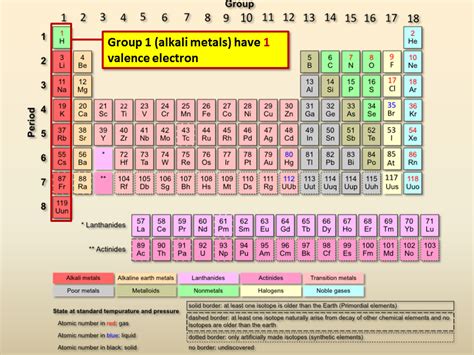
Table of Contents
The Number of Valence Electrons in Alkali Metals: A Deep Dive
Alkali metals, the stars of Group 1 on the periodic table, are known for their remarkable reactivity and distinctive properties. This reactivity is fundamentally linked to their electronic structure, specifically the number of valence electrons they possess. Understanding this crucial aspect unlocks the key to comprehending their behavior in chemical reactions, their physical properties, and their overall importance in various applications. This article will delve deep into the topic of valence electrons in alkali metals, exploring their significance and providing a comprehensive overview.
What are Valence Electrons?
Before we dive into the specifics of alkali metals, let's establish a clear understanding of valence electrons. Valence electrons are the electrons located in the outermost shell (also known as the valence shell) of an atom. These electrons are the most loosely bound to the nucleus and, therefore, play the most significant role in chemical bonding and determining an element's chemical properties. They are the primary participants in chemical reactions, readily gaining, losing, or sharing electrons to achieve a stable electron configuration, typically a full outermost shell. This stable configuration, often mirroring the electron arrangement of a noble gas, is a cornerstone of the octet rule, a fundamental principle in chemistry.
The Electronic Configuration of Alkali Metals
Alkali metals, including lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr), are characterized by a single valence electron. Their electronic configurations consistently follow a pattern: they have a filled inner electron shell(s) followed by a single electron in their outermost s orbital. For example:
- Lithium (Li): 1s²2s¹ (One valence electron in the 2s orbital)
- Sodium (Na): 1s²2s²2p⁶3s¹ (One valence electron in the 3s orbital)
- Potassium (K): 1s²2s²2p⁶3s²3p⁶4s¹ (One valence electron in the 4s orbital)
- Rubidium (Rb): 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶5s¹ (One valence electron in the 5s orbital)
- Cesium (Cs): 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶5s²4d¹⁰5p⁶6s¹ (One valence electron in the 6s orbital)
- Francium (Fr): 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶5s²4d¹⁰5p⁶6s²4f¹⁴5d¹⁰6p⁶7s¹ (One valence electron in the 7s orbital)
This consistent pattern of one valence electron is the defining characteristic of this group and the primary reason for their similar chemical behavior.
Significance of the Single Valence Electron
The presence of just one valence electron has profound consequences on the properties of alkali metals:
-
Low Ionization Energy: Because the valence electron is relatively far from the nucleus and shielded by the inner electrons, it requires only a small amount of energy to remove it. This results in low ionization energies, making them readily form +1 cations.
-
High Reactivity: Their eagerness to lose this single electron to achieve a stable electron configuration makes them highly reactive. They readily react with non-metals, especially halogens (Group 17), forming ionic compounds. This reactivity increases as you move down the group due to increasing atomic size and decreasing ionization energy.
-
Low Electronegativity: Alkali metals have low electronegativity, meaning they have a low tendency to attract electrons. This further explains their tendency to lose electrons rather than gain them.
-
Metallic Bonding: The single valence electron contributes to the metallic bonding within the metal lattice. These electrons are delocalized, forming a "sea" of electrons that holds the positively charged metal ions together. This explains their characteristic metallic properties like excellent electrical and thermal conductivity, malleability, and ductility.
Chemical Reactions and the Valence Electron
The single valence electron plays a central role in the chemical reactions of alkali metals:
-
Reactions with Halogens: Alkali metals readily react with halogens to form ionic compounds, like sodium chloride (NaCl). The alkali metal loses its valence electron to the halogen, forming a +1 cation and a -1 anion, respectively. The electrostatic attraction between these oppositely charged ions results in the formation of the ionic bond.
-
Reactions with Water: The reaction of alkali metals with water is highly exothermic (releases significant heat) and often violent. The alkali metal loses its valence electron to a hydrogen ion from the water molecule, producing hydrogen gas and a hydroxide ion. The heat generated can even ignite the hydrogen gas.
-
Reactions with Oxygen: Alkali metals react with oxygen to form oxides. The nature of the oxide depends on the specific alkali metal. For example, lithium forms lithium oxide (Li₂O), while sodium forms sodium peroxide (Na₂O₂), and potassium forms potassium superoxide (KO₂).
Impact of Atomic Size on Reactivity
As you move down Group 1, the atomic radius increases. This increase in atomic size impacts the reactivity of alkali metals:
-
Shielding Effect: The increasing number of inner electron shells shields the outermost valence electron from the positive charge of the nucleus more effectively. This reduces the electrostatic attraction between the nucleus and the valence electron.
-
Decreased Ionization Energy: The weaker attraction between the nucleus and the valence electron leads to a decrease in ionization energy. Consequently, the valence electron is even more easily lost, resulting in an increase in reactivity. This is why cesium and francium are among the most reactive elements.
Applications of Alkali Metals and their Valence Electrons
The unique properties of alkali metals stemming from their single valence electron make them invaluable in various applications:
-
Sodium Lamps: Sodium vapor lamps utilize sodium's characteristic yellow emission spectrum when its valence electrons transition between energy levels, providing efficient and bright illumination.
-
Lithium-ion Batteries: Lithium's high electrochemical potential makes it a crucial component in lithium-ion batteries, which are widely used in portable electronic devices and electric vehicles. The ability of lithium to readily lose and gain its valence electron is central to the battery's function.
-
Potassium in Fertilizers: Potassium, an essential nutrient for plant growth, plays a crucial role in regulating water balance and enzyme activation within plants. Its readily available single valence electron contributes to its ease of absorption by plants.
-
Cesium in Atomic Clocks: Cesium's precise atomic transitions are harnessed in atomic clocks for ultra-high accuracy timekeeping. This precise behavior is directly related to its electronic structure and the energy levels of its valence electron.
Conclusion: The Central Role of the Single Valence Electron
The single valence electron in alkali metals is the cornerstone of their unique chemical and physical properties. Its ease of removal, coupled with the increasing atomic size down the group, directly affects their reactivity, ionization energy, and electronegativity. This single electron is not just a numerical detail but the driving force behind their significant applications in various fields, showcasing the profound impact of electronic structure on an element's overall behavior. Understanding this simple yet powerful concept unlocks a deeper appreciation for the chemistry and applications of this fascinating group of elements. Further exploration into specific reactions and applications of each individual alkali metal can provide an even richer understanding of their importance in the scientific and technological landscape.
Latest Posts
Latest Posts
-
Charge In A Velocity Over Change In Time
Apr 05, 2025
-
Are Strawberries Considered A Citrus Fruit
Apr 05, 2025
-
Electronegativity Increases From Left To Right
Apr 05, 2025
-
Microbes Known As Transient Microbiota Are
Apr 05, 2025
-
Average Kinetic Energy Of The Particles Of A Substance
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Number Of Valence Electrons In Alkali Metals . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
