Average Kinetic Energy Of The Particles Of A Substance
Muz Play
Apr 05, 2025 · 6 min read
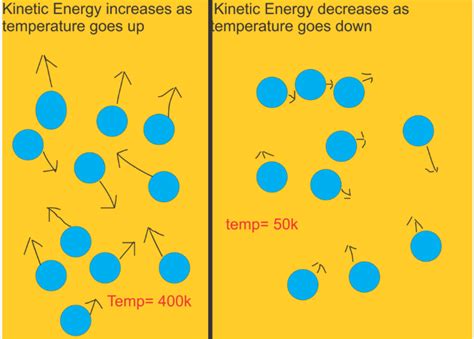
Table of Contents
The Average Kinetic Energy of Particles: A Deep Dive
The world around us is a whirlwind of activity at the microscopic level. Everything, from the air we breathe to the solid ground beneath our feet, is composed of countless particles—atoms and molecules—in constant, chaotic motion. Understanding the average kinetic energy of these particles is fundamental to comprehending the properties of matter, from its temperature and phase to its behavior in chemical reactions. This article delves into the concept of average kinetic energy, exploring its relationship with temperature, different states of matter, and its implications in various scientific fields.
What is Kinetic Energy?
Before we delve into the average kinetic energy of particles, it's crucial to understand the concept of kinetic energy itself. Kinetic energy is the energy possessed by an object due to its motion. The faster an object moves, the greater its kinetic energy. Quantitatively, it's expressed as:
KE = ½mv²
Where:
- KE represents kinetic energy (measured in Joules)
- m represents the mass of the object (measured in kilograms)
- v represents the velocity of the object (measured in meters per second)
Average Kinetic Energy and Temperature: A Direct Relationship
The particles within a substance—be it a gas, liquid, or solid—are constantly in motion, albeit with varying speeds and directions. This motion is random and chaotic, reflecting the inherent thermal energy within the substance. The average kinetic energy of these particles is directly proportional to the absolute temperature of the substance. This fundamental relationship is a cornerstone of the kinetic theory of matter.
Absolute temperature, measured in Kelvin (K), is a measure of the average kinetic energy of the particles. Zero Kelvin (0 K), also known as absolute zero, represents the theoretical point where all particle motion ceases. Importantly, this relationship holds true regardless of the substance's mass or the type of particles involved.
Understanding the Implications
This direct proportionality between average kinetic energy and absolute temperature has profound implications:
- Higher Temperature = Higher Average Kinetic Energy: As the temperature of a substance increases, the average kinetic energy of its particles increases proportionally. This leads to faster particle movement.
- Lower Temperature = Lower Average Kinetic Energy: Conversely, as the temperature decreases, the average kinetic energy decreases, resulting in slower particle movement.
- Phase Transitions: Changes in the state of matter (solid to liquid to gas) are directly linked to changes in the average kinetic energy of the particles. The energy required to overcome the intermolecular forces holding the substance together in a particular phase is directly related to the average kinetic energy needed for a phase transition.
Average Kinetic Energy in Different States of Matter
The average kinetic energy of particles significantly influences the properties and behavior of matter in its various states:
Gases
Gas particles are characterized by their high average kinetic energy and weak intermolecular forces. They move freely and independently, colliding frequently with each other and the walls of their container. The pressure exerted by a gas is directly related to the average kinetic energy of its particles—higher average kinetic energy results in higher pressure. The ideal gas law, PV = nRT, directly incorporates this relationship.
Liquids
Liquid particles possess a moderate average kinetic energy. While they are not as free to move as gas particles, they can still move past each other, leading to the fluidity characteristic of liquids. The intermolecular forces in liquids are stronger than in gases, resulting in a more structured arrangement. The average kinetic energy determines the rate of diffusion and viscosity within the liquid.
Solids
Solid particles have the lowest average kinetic energy among the three states of matter. Their motion is largely restricted to vibrations around fixed positions within a rigid structure. The strong intermolecular forces in solids maintain the substance’s fixed shape and volume. While the particles vibrate, the average kinetic energy is not sufficient to overcome these forces and allow for significant movement.
Calculating Average Kinetic Energy
The precise calculation of average kinetic energy can be complex, depending on the complexity of the system. For ideal gases, the following equation is used to relate the average kinetic energy to temperature:
KE_avg = (3/2)kT
Where:
- KE_avg represents the average kinetic energy per particle
- k represents the Boltzmann constant (1.38 x 10⁻²³ J/K)
- T represents the absolute temperature in Kelvin
This equation shows the direct proportionality between average kinetic energy and temperature. It’s crucial to remember that this is a simplification that assumes ideal gas behavior; real gases deviate from ideality under certain conditions. For more complex systems, statistical mechanics and computational methods are often employed.
Applications of Average Kinetic Energy
Understanding average kinetic energy is crucial across multiple scientific disciplines:
- Thermodynamics: The concept forms the foundation for understanding heat transfer, work, and entropy. The flow of heat is essentially the transfer of average kinetic energy from regions of higher temperature (higher average kinetic energy) to regions of lower temperature (lower average kinetic energy).
- Chemical Kinetics: The rate of chemical reactions is strongly influenced by the average kinetic energy of reacting particles. Higher average kinetic energy leads to more frequent and energetic collisions, increasing the probability of a successful reaction. Activation energy, the minimum energy required for a reaction to occur, is directly related to the average kinetic energy needed for the reaction to proceed.
- Material Science: The properties of materials, such as their strength, ductility, and thermal conductivity, are strongly linked to the average kinetic energy of their constituent particles. Understanding this relationship allows scientists to engineer materials with specific properties.
- Atmospheric Science: The average kinetic energy of atmospheric particles plays a vital role in weather patterns and climate models. The movement of air masses and the formation of clouds and precipitation are influenced by the kinetic energy of air molecules.
Beyond the Average: Distribution of Kinetic Energies
It's important to note that the average kinetic energy only provides a single representative value. In reality, the kinetic energies of individual particles within a substance are distributed across a range of values, following a statistical distribution known as the Maxwell-Boltzmann distribution. This distribution describes the probability of finding a particle with a particular kinetic energy at a given temperature. The peak of this distribution corresponds to the most probable kinetic energy, which is related to the average kinetic energy.
Conclusion: A Fundamental Concept
The average kinetic energy of particles is a fundamental concept in physics and chemistry, providing crucial insights into the behavior of matter. Its direct relationship with temperature, its influence on phase transitions, and its applications in diverse fields highlight its importance. Understanding this concept is not merely an academic pursuit; it’s essential for tackling real-world challenges in areas ranging from material science and engineering to environmental science and climate modeling. Further exploration into the Maxwell-Boltzmann distribution and statistical mechanics provides even deeper understanding of this dynamic and essential aspect of the physical world. As research continues, our comprehension of the subtleties of particle motion and its macroscopic consequences will only continue to grow.
Latest Posts
Latest Posts
-
The Families On The Periodic Table
Apr 05, 2025
-
What Is The Base Pairing Rule For Rna
Apr 05, 2025
-
What Is The Iupac Name Of This Alkane
Apr 05, 2025
-
What Does A Battery Do In A Circuit
Apr 05, 2025
-
Definition Of Word Equation In Chemistry
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Average Kinetic Energy Of The Particles Of A Substance . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
