What Does A Battery Do In A Circuit
Muz Play
Apr 05, 2025 · 6 min read
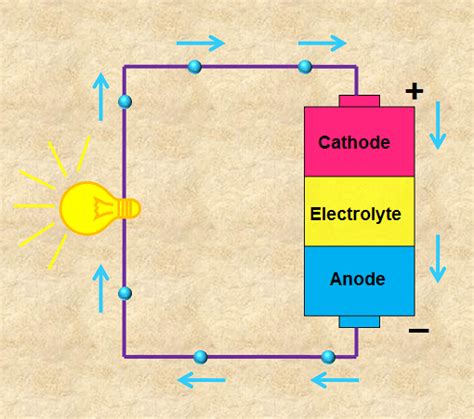
Table of Contents
What Does a Battery Do in a Circuit? A Deep Dive into Electrical Power
A battery, that ubiquitous cylindrical or rectangular device, is far more than just a power source. It's the heart of countless electronic devices, from smartphones and laptops to cars and power tools. But what exactly does a battery do in a circuit? Understanding its function requires delving into the fundamentals of electricity and circuit behavior. This comprehensive guide will explore the role of a battery in detail, covering its internal workings, its interaction with circuits, and the various types available.
The Battery: A Chemical Energy Source
At its core, a battery is a chemical energy storage and conversion device. It doesn't inherently create electricity; instead, it converts stored chemical energy into electrical energy. This conversion is achieved through a controlled chemical reaction involving two electrodes (typically a cathode and an anode) immersed in an electrolyte. The electrodes are made of different materials, each with a different tendency to lose or gain electrons.
The Electrochemical Reaction: A Closer Look
The magic happens at the electrode-electrolyte interface. The chemical reaction at the anode causes it to release electrons, while the reaction at the cathode causes it to accept electrons. This difference in electron affinity creates a potential difference, or voltage, between the two electrodes. This voltage is what drives the flow of electrons when the battery is connected to a circuit.
Think of it like a water pump in a plumbing system. The pump doesn't create water; it creates pressure that pushes the water through the pipes. Similarly, the battery doesn't create electrons; it creates an electrical potential that pushes electrons through the circuit.
Internal Resistance: The Battery's Imperfection
No battery is perfect. Every battery possesses internal resistance, which is the opposition to the flow of current within the battery itself. This resistance arises from the inherent limitations of the chemical reactions and the materials used in its construction. Internal resistance causes a voltage drop within the battery, reducing the voltage available at the terminals. This voltage drop increases with the current drawn from the battery, meaning a battery will deliver less voltage under heavy load.
Types of Batteries: A Diverse Landscape
Batteries come in a wide variety of chemistries, each with its own characteristics and applications. Some common types include:
-
Primary Batteries (Non-rechargeable): These batteries undergo an irreversible chemical reaction during discharge. Once they are depleted, they cannot be recharged. Common examples include alkaline batteries (AA, AAA) and zinc-carbon batteries.
-
Secondary Batteries (Rechargeable): These batteries undergo a reversible chemical reaction. They can be discharged and then recharged multiple times by reversing the chemical reaction using an external power source. Common examples include lead-acid batteries (used in cars), lithium-ion batteries (used in laptops and smartphones), and nickel-metal hydride batteries (used in some hybrid vehicles).
The choice of battery type depends on the specific application. Factors to consider include energy density (energy stored per unit mass or volume), power density (rate at which energy can be delivered), cycle life (number of charge-discharge cycles), cost, and safety.
The Battery in a Simple Circuit
Let's consider a simple circuit comprising a battery, a light bulb, and connecting wires. When the circuit is closed (the wires are connected to the battery terminals), the following happens:
-
Potential Difference: The battery establishes a potential difference (voltage) between its positive and negative terminals.
-
Electron Flow: This potential difference creates an electric field that pushes electrons from the negative terminal of the battery, through the connecting wires, to the light bulb filament.
-
Energy Conversion: As the electrons pass through the filament's high resistance, they collide with atoms, converting electrical energy into heat and light. This is why the light bulb glows.
-
Circuit Completion: The electrons then travel through the other wire and back to the positive terminal of the battery, completing the circuit. This continuous flow of electrons constitutes an electric current.
The battery acts as the driving force for this electron flow, providing the energy necessary for the light bulb to function. The circuit provides a path for the electrons to flow from the negative to the positive terminal of the battery.
Understanding Circuit Parameters: Voltage, Current, and Resistance
To fully understand the battery's role, we need to grasp the fundamental circuit parameters:
-
Voltage (V): The electrical potential difference between two points in a circuit. It's the "push" that drives the electrons. The battery's voltage determines the potential energy available to the circuit.
-
Current (I): The rate of flow of electric charge (electrons) past a given point in the circuit. It's measured in amperes (amps). The current indicates how many electrons are flowing per second.
-
Resistance (R): The opposition to the flow of current in a circuit. It's measured in ohms (Ω). Resistance determines how much the circuit impedes the flow of electrons.
Ohm's Law elegantly relates these three parameters: V = IR. This means that the voltage across a component is equal to the current flowing through it multiplied by its resistance.
Battery's Role in Complex Circuits
While the simple circuit illustrates the basic principles, real-world circuits are far more complex. Batteries still provide the driving force, but their role becomes nuanced:
-
Powering Multiple Components: A single battery can power multiple components, each with its own resistance. The current flowing through each component depends on its resistance and the battery's voltage, governed by Ohm's Law and Kirchhoff's circuit laws.
-
Series and Parallel Connections: Batteries can be connected in series (positive to negative) to increase the voltage, or in parallel (positive to positive, negative to negative) to increase the current capacity and extend battery life.
-
Voltage Regulators and Power Management: Complex circuits often incorporate voltage regulators to maintain a stable voltage for sensitive components, even as the battery's voltage fluctuates during discharge. Power management integrated circuits (ICs) optimize power consumption and extend battery life.
-
Charging Circuits: Rechargeable batteries require sophisticated charging circuits to regulate the charging current and voltage, preventing overcharging and damage to the battery. These circuits employ various techniques, such as constant current charging and constant voltage charging.
Beyond the Basics: Advanced Concepts
-
Battery Capacity: Measured in ampere-hours (Ah) or milliampere-hours (mAh), this indicates the total amount of charge a battery can deliver before being depleted.
-
Battery Life: This depends on factors such as the battery's capacity, the current drawn from it, and its operating temperature.
-
Self-Discharge: Batteries gradually lose their charge even when not in use, a phenomenon called self-discharge. This is due to internal chemical reactions.
-
Battery Health: Rechargeable batteries degrade over time and with repeated charge-discharge cycles. Their capacity and performance decline, a process that can be influenced by charging habits and operating temperature.
-
Battery Management Systems (BMS): These systems are crucial in complex devices and electric vehicles, monitoring the battery's voltage, current, temperature, and state of charge to ensure safe and efficient operation, extend battery life, and prevent damage.
Conclusion: The Unsung Hero of Electronics
The battery's role in a circuit may seem simple at first glance, but it is a critical one, involving complex chemical and electrical processes. From providing the initial driving force for electron flow in a simple circuit to managing the power distribution in sophisticated devices, the battery is the unsung hero of our electronic world. Understanding its function, characteristics, and limitations is crucial for anyone working with electronics, from hobbyists to engineers. By grasping the principles outlined in this article, you'll gain a much deeper appreciation for this seemingly simple yet remarkably complex device and its integral contribution to the modern world.
Latest Posts
Latest Posts
-
Practicing Dna Transcription And Translation Answer Key
Apr 06, 2025
-
How To Prove A Transformation Is Linear
Apr 06, 2025
-
Is The Mutant Allele Dominant Or Recessive
Apr 06, 2025
-
Why Do Covalent Compounds Have Low Melting Points
Apr 06, 2025
-
Which Of The Following Determines The Length Of The Base
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Does A Battery Do In A Circuit . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
