The Families On The Periodic Table
Muz Play
Apr 05, 2025 · 7 min read
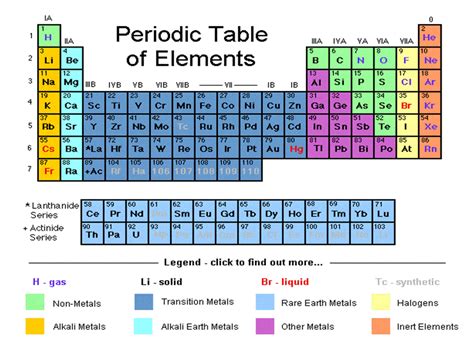
Table of Contents
The Families on the Periodic Table: A Deep Dive into the Elements' Relationships
The periodic table, a cornerstone of chemistry, isn't just a random arrangement of elements. It's a meticulously organized system reflecting the fundamental properties and relationships between atoms. Understanding these relationships, particularly within the element families or groups, is crucial to grasping chemical behavior and predicting reactions. This comprehensive guide delves into the major families of the periodic table, exploring their unique characteristics, trends, and applications.
The Alkali Metals: The Highly Reactive Family (Group 1)
The alkali metals, located in Group 1, are highly reactive metals characterized by their single valence electron. This lone electron readily participates in chemical reactions, making these elements extremely reactive, especially with water and oxygen. Their reactivity increases as you move down the group.
Key Characteristics:
- Highly reactive: They readily lose their single valence electron to form +1 ions.
- Soft and silvery: They are easily cut with a knife.
- Low melting and boiling points: Compared to other metals.
- Good conductors of heat and electricity: Due to their mobile valence electrons.
- React violently with water: Producing hydrogen gas and a metal hydroxide.
Notable Members:
- Lithium (Li): Used in batteries, ceramics, and lubricants.
- Sodium (Na): Essential for human life (electrolyte balance), found in table salt (NaCl).
- Potassium (K): Also crucial for human health (nerve function), used in fertilizers.
- Rubidium (Rb) and Cesium (Cs): Used in atomic clocks and specialized applications.
- Francium (Fr): A radioactive element, extremely rare and unstable.
Applications:
Alkali metals find numerous applications in various industries, from batteries (lithium-ion batteries are ubiquitous) to medicine (sodium and potassium are essential electrolytes) and agriculture (potassium fertilizers are crucial for plant growth). Their reactivity, while posing safety challenges, also makes them invaluable in specific chemical processes.
The Alkaline Earth Metals: Reactive, but Less So (Group 2)
The alkaline earth metals, in Group 2, possess two valence electrons, making them less reactive than the alkali metals but still significantly reactive. These elements also exhibit trends in reactivity and properties similar to the alkali metals, although less pronounced.
Key Characteristics:
- Reactive, but less so than alkali metals: They lose two valence electrons to form +2 ions.
- Silvery-white in appearance: Though some may appear slightly yellowish.
- Higher melting and boiling points than alkali metals: Reflecting stronger metallic bonding.
- Good conductors of heat and electricity: Similar to alkali metals.
- React with water (though less violently than alkali metals): Forming metal hydroxides and hydrogen gas.
Notable Members:
- Beryllium (Be): Used in aerospace alloys due to its high strength-to-weight ratio.
- Magnesium (Mg): Used in lightweight alloys, also plays a vital role in human biology.
- Calcium (Ca): Essential for bones and teeth, used in cement and plaster.
- Strontium (Sr) and Barium (Ba): Used in fireworks for their vibrant colors.
- Radium (Ra): A radioactive element, historically used in medical applications, now largely replaced by safer alternatives.
Applications:
Alkaline earth metals are vital in construction (calcium in cement), aerospace (beryllium in alloys), and biological systems (magnesium and calcium are essential for life). Their properties make them suitable for a range of applications, from creating strong yet lightweight materials to providing essential nutrients.
The Transition Metals: The Colorful and Versatile Group (Groups 3-12)
The transition metals, spanning Groups 3-12, form the central block of the periodic table. They are characterized by their partially filled d orbitals, which gives rise to their unique properties, including variable oxidation states and often vibrant colors in their compounds.
Key Characteristics:
- Variable oxidation states: They can exist in multiple oxidation states, leading to a wide range of compounds.
- Colorful compounds: Due to the absorption and emission of light by their d electrons.
- Good conductors of heat and electricity: Due to the delocalized d electrons.
- High melting and boiling points: Generally higher than those of alkali and alkaline earth metals.
- Catalytic activity: Many transition metals and their compounds act as catalysts in various chemical reactions.
Notable Members:
- Iron (Fe): Crucial for oxygen transport in blood, used in steel production.
- Copper (Cu): Excellent conductor of electricity, used in wiring and plumbing.
- Zinc (Zn): Used in galvanization (corrosion protection), essential trace element.
- Nickel (Ni): Used in stainless steel and nickel-cadmium batteries.
- Gold (Au) and Platinum (Pt): Precious metals, used in jewelry and catalysis.
Applications:
Transition metals are essential in countless applications, from building materials (steel) and electronics (copper wiring) to catalysis (platinum in catalytic converters) and medicine (iron in hemoglobin). Their versatile properties make them invaluable across diverse fields.
The Halogens: The Reactive Nonmetals (Group 17)
The halogens, located in Group 17, are a family of highly reactive nonmetals. They readily gain an electron to achieve a stable noble gas configuration, forming -1 anions. Their reactivity decreases as you move down the group.
Key Characteristics:
- Highly reactive: They readily gain one electron to form -1 ions.
- Diatomic molecules: They exist as diatomic molecules (e.g., Cl₂, Br₂) at room temperature.
- Varying states of matter: Fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid.
- Strong oxidizing agents: They readily accept electrons.
- Form various compounds: With metals and other nonmetals.
Notable Members:
- Fluorine (F): The most reactive element, used in toothpaste and refrigerants.
- Chlorine (Cl): Used in water purification and the production of various chemicals.
- Bromine (Br): Used in flame retardants and photographic film.
- Iodine (I): Essential for thyroid function, used as a disinfectant.
- Astatine (At): A radioactive element, extremely rare.
Applications:
Halogens are crucial in numerous applications, from water purification (chlorine) and dentistry (fluoride) to medicine (iodine) and various industrial processes. Their high reactivity makes them both beneficial and potentially hazardous, requiring careful handling.
The Noble Gases: The Inert Elements (Group 18)
The noble gases, in Group 18, are a unique family of elements known for their extreme inertness. They possess a complete valence electron shell (octet), making them exceptionally stable and unreactive.
Key Characteristics:
- Inert: They rarely participate in chemical reactions due to their stable electron configuration.
- Colorless, odorless gases: At room temperature.
- Low boiling points: Reflecting weak interatomic forces.
- Used in lighting and lasers: Due to their ability to emit light when excited.
- Generally monatomic: They exist as single atoms, not molecules.
Notable Members:
- Helium (He): Used in balloons and cryogenics.
- Neon (Ne): Used in neon signs.
- Argon (Ar): Used in welding and as a protective atmosphere.
- Krypton (Kr) and Xenon (Xe): Used in specialized lighting applications.
- Radon (Rn): A radioactive gas, a health hazard.
Applications:
Noble gases find applications in lighting (neon signs), welding (argon), and cryogenics (helium). Their inertness makes them ideal for creating protective atmospheres in various industrial processes. While generally unreactive, some heavier noble gases can form compounds under special conditions, showcasing the exceptions that prove the rule.
Beyond the Main Families: Lanthanides and Actinides
Beyond the main groups, the periodic table also contains the lanthanides and actinides, two series of elements with unique properties.
Lanthanides:
These elements, also known as rare earth elements, are characterized by their filling of the 4f orbitals. They exhibit similar chemical properties, making their separation and purification challenging. They are essential in various technological applications, such as magnets, catalysts, and lighting.
Actinides:
The actinides are characterized by the filling of the 5f orbitals. Many are radioactive, with applications in nuclear energy and medicine (although increasingly limited due to safety concerns). Their radioactivity makes them both powerful and hazardous, requiring extremely careful handling.
Conclusion: The Interconnectedness of the Elements
The periodic table, with its families and groups, beautifully illustrates the interconnectedness of the elements. Understanding the properties and trends within each family is crucial for predicting chemical behavior, designing new materials, and advancing various scientific and technological fields. From the highly reactive alkali metals to the inert noble gases, each family contributes uniquely to the richness and complexity of the chemical world. The families are not isolated entities; they interact and react with each other in countless ways, shaping the world around us. Further exploration of these families and their intricate relationships will continue to unlock new possibilities in chemistry and beyond. The periodic table is a living document, constantly evolving as our understanding of the elements deepens.
Latest Posts
Latest Posts
-
A Gas Has No Definite Shape And Volume
Apr 06, 2025
-
What Are The Three Factors That Affect Gas Pressure
Apr 06, 2025
-
Expected Number Of Trials Until Success
Apr 06, 2025
-
Determinants Of The Elasticity Of Supply
Apr 06, 2025
-
What Does A Replicated Chromosome Look Like
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about The Families On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
