Plant Cell In A Isotonic Solution
Muz Play
Apr 02, 2025 · 6 min read
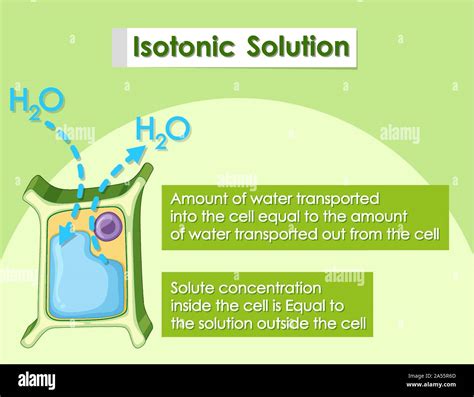
Table of Contents
Plant Cells in an Isotonic Solution: A Deep Dive into Osmosis and Turgor Pressure
Plant cells, unlike animal cells, possess a rigid cell wall surrounding their delicate plasma membrane. This structural difference significantly impacts how they respond to changes in their external environment, particularly concerning the concentration of solutes in the surrounding solution. This article will delve into the fascinating world of plant cells immersed in an isotonic solution, exploring the intricate interplay of osmosis, turgor pressure, and the implications for plant health and function.
Understanding Osmosis: The Driving Force
Before diving into the specifics of plant cells in isotonic solutions, it's crucial to understand the fundamental principle of osmosis. Osmosis is the passive movement of water molecules across a selectively permeable membrane from a region of higher water concentration (lower solute concentration) to a region of lower water concentration (higher solute concentration). This movement continues until an equilibrium is reached, or until the opposing force (such as turgor pressure) becomes too strong. Think of it like this: water always wants to move to where it's "more crowded" with solutes.
The selectively permeable membrane, in the case of plant cells, is the plasma membrane. It allows the passage of water molecules but restricts the movement of many solutes, creating the conditions necessary for osmosis to occur. The direction and rate of water movement depend on the concentration gradient across the membrane – the difference in solute concentration between the inside and outside of the cell.
Isotonic Solutions: A Balancing Act
An isotonic solution is one in which the concentration of solutes is equal both inside and outside the plant cell. This means there's no net movement of water across the cell membrane. The water potential inside the cell is equal to the water potential outside the cell.
When a plant cell is placed in an isotonic solution, a dynamic equilibrium is established. While water molecules still move across the membrane in both directions, the rate of movement is equal in both directions. This results in no overall change in the cell's volume or shape. The cell maintains its normal shape and size; neither shrinking nor swelling.
Implications of Isotonic Solutions for Plant Cells
-
No Net Water Movement: The most significant implication is the absence of net water movement. This is in contrast to hypotonic and hypertonic solutions, where water moves either into or out of the cell, respectively.
-
Stable Cell Volume and Shape: The cell maintains its normal size and shape. This is crucial for the cell's structural integrity and its ability to perform its functions effectively.
-
Optimal Turgor Pressure Maintenance: While there's no net water movement, the cell still maintains some turgor pressure. This pressure is still sufficient for maintaining the cell's shape and preventing wilting. However, it is not as high as in a hypotonic solution.
-
Balanced Intracellular Environment: The isotonic solution provides a stable and balanced environment for the cell's internal components, allowing for the efficient functioning of cellular processes.
-
Absence of Plasmolysis: Plasmolysis, the shrinking of the cytoplasm away from the cell wall due to water loss in a hypertonic solution, doesn't occur in an isotonic environment. The cell remains fully turgid, albeit at a relatively lower pressure.
-
Impact on Metabolic Processes: The maintenance of a stable cellular environment ensures that metabolic processes operate optimally. Enzyme activity, nutrient transport, and other cellular functions are not negatively impacted by drastic changes in water potential.
Contrasting Isotonic Solutions with Hypotonic and Hypertonic Solutions
To fully appreciate the significance of isotonic solutions for plant cells, it's helpful to compare them with hypotonic and hypertonic solutions:
Hypotonic Solutions: A Swelling Affair
A hypotonic solution has a lower solute concentration (higher water potential) than the inside of the plant cell. In this scenario, water moves from the solution into the cell via osmosis. This influx of water causes the cell to swell, and the plasma membrane pushes against the cell wall. This pressure is known as turgor pressure.
Turgor pressure is essential for maintaining plant cell rigidity and overall plant structure. It's what keeps herbaceous plants upright. However, excessive turgor pressure can lead to cell bursting (lysis) if the cell wall is unable to withstand the pressure. Fortunately, the plant cell wall provides a strong and relatively inelastic barrier preventing such lysis.
Hypertonic Solutions: Shrinking to a Halt
A hypertonic solution has a higher solute concentration (lower water potential) than the inside of the plant cell. Here, water moves out of the cell via osmosis, causing the cell to lose volume and the plasma membrane to pull away from the cell wall. This process is called plasmolysis. Plasmolysis significantly compromises cell function and can lead to wilting and even cell death if prolonged.
Practical Applications and Real-World Examples
Understanding the effects of isotonic solutions on plant cells has several practical applications in various fields:
-
Plant Tissue Culture: Isotonic solutions are crucial in maintaining the viability of plant cells and tissues during in vitro propagation and other tissue culture techniques. Providing the cells with an optimal isotonic environment promotes growth and prevents cell damage.
-
Plant Physiology Research: Isotonic solutions are used extensively in plant physiology experiments to investigate the transport of water, solutes, and other substances across plant cell membranes. By controlling the osmotic environment, researchers can study the effects of various factors on plant cell behavior.
-
Agriculture and Horticulture: Maintaining appropriate soil water potential and nutrient levels is critical for plant health. Understanding isotonic principles helps in optimizing irrigation practices and fertilizer application to prevent both water stress and nutrient imbalances that can lead to plasmolysis or excessive turgor.
-
Food Preservation: Controlling the osmotic environment is also important in food preservation. Isotonic solutions or solutions with carefully balanced osmotic pressure can be used to prevent microbial growth and maintain the quality of food products.
-
Plant Disease Resistance: Plant cell response to pathogens and environmental stresses is often influenced by the osmotic state of the cell. Understanding the interplay between osmotic conditions and disease resistance is critical for developing disease-resistant varieties and effective management strategies.
Conclusion: The Importance of Osmotic Balance
The interaction between plant cells and isotonic solutions underscores the critical importance of maintaining osmotic balance for plant health and survival. While an isotonic solution provides a stable cellular environment without inducing drastic water movement, it's vital to remember that plant cells naturally operate within a dynamic range of water potentials. Factors like soil moisture, humidity, and temperature all influence this range, making understanding osmosis and its impact on plant cells an essential aspect of plant biology and related fields. Understanding isotonic solutions is therefore, crucial for ensuring plant health and addressing many agricultural and horticultural challenges. This knowledge paves the way for innovative solutions in agriculture, plant biotechnology, and related disciplines.
Latest Posts
Latest Posts
-
Difference Between A Somatic Cell And A Gamete
Apr 03, 2025
-
Match The Structure Process To The Letter
Apr 03, 2025
-
Find The Basis Of The Subspace
Apr 03, 2025
-
Why Is Immersion Oil Used With The 100x Objective
Apr 03, 2025
-
What Is Shared In A Covalent Bond
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Plant Cell In A Isotonic Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
