Proteins Are Polymers Of Amino Acid Monomers
Muz Play
Apr 05, 2025 · 6 min read
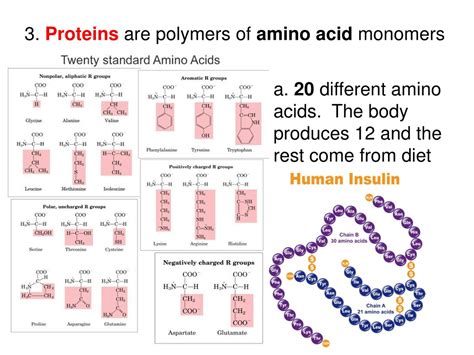
Table of Contents
Proteins: Polymers of Amino Acid Monomers
Proteins are the workhorses of the cell, carrying out a vast array of functions essential for life. From catalyzing biochemical reactions as enzymes to providing structural support as components of hair and nails, proteins are ubiquitous and indispensable. Understanding their structure and function begins with grasping a fundamental concept: proteins are polymers of amino acid monomers. This article delves deep into this relationship, exploring the intricacies of amino acids, the process of polymerization, and the diverse structures and functions that result.
The Building Blocks: Amino Acids
Amino acids, the monomers of proteins, are organic molecules characterized by a specific structure. Each amino acid consists of a central carbon atom (the α-carbon) bonded to four groups:
- An amino group (-NH2): This group is basic and readily accepts a proton (H+), contributing to the amino acid's overall charge.
- A carboxyl group (-COOH): This group is acidic and readily donates a proton (H+), also influencing the amino acid's charge.
- A hydrogen atom (-H): This simple group contributes to the overall structure.
- A side chain (R-group): This variable group is what distinguishes one amino acid from another. The R-group's properties – size, charge, polarity, and reactivity – dictate the amino acid's characteristics and ultimately influence the protein's overall properties.
There are 20 standard amino acids commonly found in proteins, each with a unique R-group. These amino acids can be categorized based on their R-group properties:
Types of Amino Acids Based on R-Group Properties
- Nonpolar, aliphatic amino acids: These amino acids have hydrocarbon side chains, making them hydrophobic (water-repelling). Examples include glycine, alanine, valine, leucine, isoleucine, and methionine.
- Aromatic amino acids: These amino acids have ring structures in their side chains, often contributing to protein absorbance of UV light. Examples include phenylalanine, tyrosine, and tryptophan.
- Polar, uncharged amino acids: These amino acids have polar but uncharged side chains, making them hydrophilic (water-attracting). Examples include serine, threonine, cysteine, asparagine, and glutamine.
- Positively charged (basic) amino acids: These amino acids have positively charged side chains at physiological pH. Examples include lysine, arginine, and histidine.
- Negatively charged (acidic) amino acids: These amino acids have negatively charged side chains at physiological pH. Examples include aspartic acid and glutamic acid.
Peptide Bonds: Linking Amino Acids
Amino acids are linked together through a process called dehydration synthesis, or condensation. In this process, the carboxyl group of one amino acid reacts with the amino group of another amino acid, releasing a water molecule (H2O) and forming a peptide bond (also known as an amide bond). This bond is a strong covalent bond, crucial for maintaining the primary structure of the protein.
The resulting molecule is called a dipeptide if two amino acids are linked, a tripeptide if three are linked, and a polypeptide if many are linked. Proteins are essentially long polypeptide chains composed of hundreds or even thousands of amino acids. The sequence of amino acids in a polypeptide chain is known as its primary structure, and this sequence dictates the protein's higher-order structures and ultimately its function.
Protein Structure: From Primary to Quaternary
The intricate three-dimensional structure of a protein is essential for its function. Protein structure is described in four levels of organization:
1. Primary Structure: The Amino Acid Sequence
As mentioned above, the primary structure is the linear sequence of amino acids in a polypeptide chain. This sequence is determined by the genetic code, which dictates the order in which amino acids are incorporated during protein synthesis. Even a single change in the amino acid sequence (a mutation) can drastically alter the protein's structure and function.
2. Secondary Structure: Local Folding Patterns
The primary structure folds into local patterns stabilized by hydrogen bonds between the backbone atoms (the amino and carboxyl groups). Two common secondary structures are:
- α-helices: A right-handed coiled structure stabilized by hydrogen bonds between the carbonyl oxygen of one amino acid and the amide hydrogen of an amino acid four residues away.
- β-sheets: Extended polypeptide chains arranged side-by-side, stabilized by hydrogen bonds between adjacent chains. β-sheets can be parallel (chains run in the same direction) or antiparallel (chains run in opposite directions).
Other secondary structures, such as loops and turns, also contribute to the overall protein fold.
3. Tertiary Structure: The Overall 3D Arrangement
The tertiary structure refers to the overall three-dimensional arrangement of a polypeptide chain, including the arrangement of its secondary structures. This structure is stabilized by various interactions between amino acid side chains (R-groups), including:
- Hydrophobic interactions: Nonpolar side chains cluster together in the protein's interior, away from the surrounding water.
- Hydrogen bonds: Hydrogen bonds form between polar side chains.
- Ionic bonds (salt bridges): Electrostatic interactions between oppositely charged side chains.
- Disulfide bonds: Covalent bonds formed between cysteine residues. These are strong bonds that contribute significantly to protein stability.
4. Quaternary Structure: Multiple Polypeptide Chains
Some proteins consist of multiple polypeptide chains (subunits) associated together to form a functional protein complex. The arrangement of these subunits is known as the quaternary structure. Hemoglobin, for example, is a tetramer (composed of four subunits). Interactions similar to those stabilizing tertiary structure also stabilize quaternary structure.
Protein Function: Diversity and Specificity
The diverse functions of proteins stem from their highly specific three-dimensional structures. The precise arrangement of amino acid side chains creates binding sites for specific ligands (molecules that bind to proteins), allowing proteins to perform their roles with remarkable efficiency and specificity.
Some major protein functions include:
- Enzymes: Catalyze biochemical reactions.
- Structural proteins: Provide structural support (e.g., collagen, keratin).
- Transport proteins: Carry molecules across membranes (e.g., hemoglobin, ion channels).
- Motor proteins: Generate movement (e.g., myosin, kinesin).
- Hormones: Act as chemical messengers (e.g., insulin, growth hormone).
- Antibodies: Part of the immune system, recognizing and neutralizing foreign substances.
- Receptor proteins: Receive and transmit signals.
Protein Misfolding and Disease
The proper folding of proteins is critical for their function. Errors in protein folding can lead to the formation of misfolded proteins, which can aggregate and cause a variety of diseases, including Alzheimer's disease, Parkinson's disease, and cystic fibrosis. These diseases highlight the importance of maintaining proper protein structure and the devastating consequences of misfolding.
Conclusion: The Intricate World of Proteins
Proteins are remarkable macromolecules, essential for life's processes. Their diverse functions are a direct consequence of their intricate structures, built from the assembly of amino acid monomers. Understanding the relationship between amino acid sequence, protein folding, and protein function is crucial for comprehending the complexity of biological systems and developing new strategies for treating diseases related to protein misfolding. Further research into the intricacies of protein structure and function continues to unlock new insights into the fundamental mechanisms of life itself. The study of proteins remains a vibrant and rapidly evolving field, promising breakthroughs in various areas of biology and medicine.
Latest Posts
Latest Posts
-
How Does A Competitive Inhibitor Slow Enzyme Catalysis
Apr 05, 2025
-
What Is The Difference Between An Equation And Inequality
Apr 05, 2025
-
Does Electric Field Go From Positive To Negative
Apr 05, 2025
-
What Tools Are Used To Measure Humidity
Apr 05, 2025
-
How To Solve Inequalities In Interval Notation
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Proteins Are Polymers Of Amino Acid Monomers . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
