How Does A Competitive Inhibitor Slow Enzyme Catalysis
Muz Play
Apr 05, 2025 · 6 min read
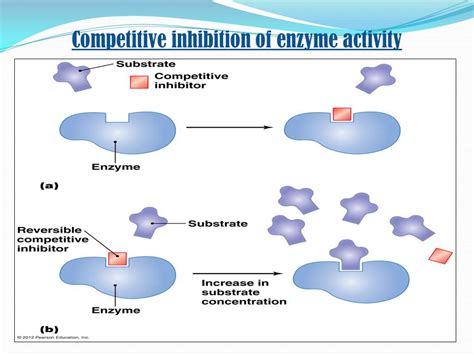
Table of Contents
How Does a Competitive Inhibitor Slow Enzyme Catalysis?
Enzymes are biological catalysts that significantly speed up the rate of chemical reactions within living organisms. They achieve this remarkable feat by lowering the activation energy required for a reaction to proceed. However, enzyme activity isn't always unimpeded. A variety of molecules can interfere with enzyme function, and competitive inhibitors are a significant class of these enzyme inhibitors. Understanding how these inhibitors work is crucial for comprehending enzyme kinetics, drug design, and metabolic regulation.
Understanding Enzyme-Substrate Interactions
Before delving into the mechanism of competitive inhibition, let's establish a basic understanding of how enzymes interact with their substrates. Enzymes possess a specific three-dimensional structure, including an active site – a region with a unique shape and chemical properties that precisely binds the substrate(s). This interaction, often likened to a "lock and key" or "induced fit" model, brings the substrates into close proximity and optimal orientation for the reaction to occur. The enzyme-substrate complex then undergoes a series of conformational changes, facilitating the chemical transformation and the release of the product(s).
The Lock and Key Model:
This classic model portrays the enzyme's active site as a rigid structure, perfectly complementary to the substrate's shape. Only substrates with the correct shape can fit and bind to the active site.
The Induced Fit Model:
This more refined model recognizes that the active site is flexible and can undergo conformational changes upon substrate binding. The binding process itself induces changes in the enzyme's structure, optimizing the interaction for catalysis.
Competitive Inhibition: A Detailed Look
Competitive inhibitors are molecules that resemble the enzyme's substrate in structure. This structural similarity allows them to bind to the enzyme's active site, competing directly with the substrate for binding. Crucially, competitive inhibitors do not undergo the catalytic reaction. Their binding is reversible, meaning the inhibitor can dissociate from the enzyme, allowing the substrate to bind subsequently.
The Mechanism of Inhibition:
-
Binding to the Active Site: The competitive inhibitor binds to the enzyme's active site, preventing the substrate from binding. This forms an enzyme-inhibitor complex (EI).
-
Competition with the Substrate: The inhibitor and the substrate are competing for the same binding site on the enzyme. The relative concentrations of the inhibitor and substrate determine the outcome. A high concentration of substrate can outcompete the inhibitor, while a high inhibitor concentration can effectively block the enzyme's activity.
-
Reversible Binding: The binding of the competitive inhibitor is reversible. The EI complex can dissociate, releasing the inhibitor and making the enzyme available for substrate binding again. This reversibility is a key characteristic differentiating competitive inhibition from other types of inhibition.
-
No Change to Vmax: While the inhibitor slows down the reaction rate at lower substrate concentrations, the maximum velocity (Vmax) of the enzyme remains unchanged. At sufficiently high substrate concentrations, the substrate can outcompete the inhibitor for the active site, achieving the enzyme's maximum catalytic rate.
-
Increased Km: The Michaelis-Menten constant (Km), which represents the substrate concentration at half-maximal velocity, is increased in the presence of a competitive inhibitor. This reflects the apparent decrease in the enzyme's affinity for the substrate due to the inhibitor's presence.
Visualizing Competitive Inhibition: Lineweaver-Burk Plots
The effects of competitive inhibition are clearly visualized using Lineweaver-Burk plots (double reciprocal plots). These plots illustrate the reciprocal of the reaction velocity (1/V) against the reciprocal of the substrate concentration (1/ [S]).
-
No Inhibitor: In the absence of an inhibitor, the plot shows a straight line with a y-intercept of 1/Vmax and an x-intercept of -1/Km.
-
Competitive Inhibitor: In the presence of a competitive inhibitor, the lines intersect on the y-axis (at 1/Vmax), indicating that Vmax remains unchanged. However, the x-intercept shifts to the left, reflecting the increased Km.
Factors Influencing the Effectiveness of Competitive Inhibitors
Several factors influence the effectiveness of competitive inhibitors:
-
Inhibitor Concentration: Higher inhibitor concentrations lead to greater inhibition.
-
Substrate Concentration: Higher substrate concentrations can overcome the inhibition, as the substrate outcompetes the inhibitor for binding to the active site.
-
Inhibitor Affinity: Inhibitors with higher affinity for the enzyme's active site are more effective inhibitors.
-
Structural Similarity to the Substrate: The greater the structural similarity between the inhibitor and the substrate, the more effective the inhibition will be.
Examples of Competitive Inhibitors
Competitive inhibitors play important roles in various biological processes and have significant therapeutic applications. Here are some notable examples:
-
Methotrexate: This drug acts as a competitive inhibitor of dihydrofolate reductase, an enzyme crucial for nucleotide biosynthesis. By inhibiting this enzyme, methotrexate disrupts DNA synthesis and cell proliferation, making it effective against certain cancers.
-
Statins: Statins are competitive inhibitors of HMG-CoA reductase, an enzyme involved in cholesterol biosynthesis. By inhibiting this enzyme, statins lower cholesterol levels in the blood, reducing the risk of cardiovascular disease.
-
Sulfonamides: Sulfonamides are competitive inhibitors of para-aminobenzoic acid (PABA) synthesis in bacteria. PABA is a precursor to folic acid, essential for bacterial growth. By inhibiting PABA synthesis, sulfonamides prevent bacterial growth, making them effective antibiotics.
Distinguishing Competitive Inhibition from Other Types of Inhibition
It's crucial to differentiate competitive inhibition from other types of enzyme inhibition, such as non-competitive and uncompetitive inhibition. These other types of inhibitors do not compete directly with the substrate for the active site and exhibit different effects on Vmax and Km.
-
Non-competitive inhibition: The inhibitor binds to a site other than the active site (allosteric site), causing a conformational change that reduces the enzyme's catalytic activity. Both Vmax and Km are affected.
-
Uncompetitive inhibition: The inhibitor binds only to the enzyme-substrate complex, preventing the release of products. Both Vmax and Km are affected, but in a manner distinct from non-competitive inhibition.
Conclusion: The Significance of Competitive Inhibition
Competitive inhibition is a fundamental mechanism of enzyme regulation with far-reaching implications in biology and medicine. Understanding how competitive inhibitors function is essential for interpreting enzyme kinetics, designing new drugs, and manipulating metabolic pathways. The ability to design and utilize competitive inhibitors provides a powerful tool for influencing biological processes and treating various diseases. The reversible nature of competitive inhibition offers a level of control and reversibility that makes it particularly useful in therapeutic applications, allowing for precise modulation of enzyme activity and minimizing unintended side effects. Continued research in this area promises to yield further insights and applications of competitive inhibition in the future. Further exploration into the intricacies of enzyme-inhibitor interactions promises exciting advances in both fundamental biological understanding and therapeutic development.
Latest Posts
Latest Posts
-
What Causes Semilunar Valves To Open
Apr 05, 2025
-
A Covalent Bond Is Formed As A Result Of
Apr 05, 2025
-
What Is A Net Change In Math
Apr 05, 2025
-
Is Corrosion A Physical Or Chemical Property
Apr 05, 2025
-
Find The Equation Of The Tangent Plane To The Surface
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Does A Competitive Inhibitor Slow Enzyme Catalysis . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
