A Covalent Bond Is Formed As A Result Of
Muz Play
Apr 05, 2025 · 6 min read
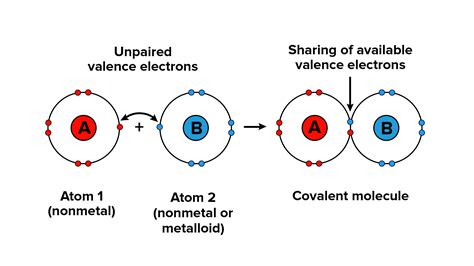
Table of Contents
A Covalent Bond is Formed as a Result of: Sharing Electrons to Achieve Stability
Covalent bonds are fundamental to chemistry, forming the backbone of countless molecules essential to life and countless industrial applications. Understanding how these bonds form is crucial for grasping the properties and behaviors of matter. This article delves deep into the process of covalent bond formation, exploring the underlying principles, types of covalent bonds, factors influencing bond strength, and the implications of these bonds in various contexts.
The Driving Force: Octet Rule and Stability
At the heart of covalent bond formation lies the quest for stability. Atoms, the building blocks of matter, strive to achieve a stable electron configuration, most commonly resembling that of the nearest noble gas. This is often referred to as the octet rule, stating that atoms tend to gain, lose, or share electrons to obtain eight electrons in their outermost shell (valence shell). Exceptions exist, particularly for elements with low atomic numbers like hydrogen and lithium, which aim for a duet (two electrons) and helium's stable configuration.
This drive for stability is the primary reason atoms engage in bonding. When atoms share electrons, they effectively complete their valence shells, lowering their overall energy and increasing stability. This shared electron pair constitutes the covalent bond.
Energy Considerations: Lowering Potential Energy
Bond formation is an energetically favorable process. Isolated atoms possess higher potential energy compared to atoms bonded together. When a covalent bond forms, the system's potential energy decreases, signifying increased stability. This energy difference is the bond energy, representing the energy required to break the bond. A higher bond energy indicates a stronger, more stable bond.
The Mechanics of Covalent Bond Formation: Overlapping Orbitals
Covalent bonds arise from the overlap of atomic orbitals. Atomic orbitals are regions of space around the nucleus where there's a high probability of finding an electron. When two atoms approach each other, their valence orbitals overlap, allowing electrons to be shared between the two nuclei. This overlap leads to a build-up of electron density between the nuclei, creating the attractive force that holds the atoms together.
Types of Orbital Overlap: Sigma and Pi Bonds
The type of orbital overlap dictates the characteristics of the covalent bond. Two primary types exist:
-
Sigma (σ) bonds: These are formed by the direct, head-on overlap of atomic orbitals. They are the strongest type of covalent bond and are typically found as the single bond between two atoms. Examples include the C-H bond in methane and the O-H bond in water. Sigma bonds allow free rotation around the bond axis.
-
Pi (π) bonds: These are formed by the sideways overlap of p orbitals. They are weaker than sigma bonds and are typically found in multiple bonds (double or triple bonds). In a double bond, one bond is a sigma bond, and the other is a pi bond. In a triple bond, one sigma bond and two pi bonds exist. Pi bonds restrict rotation around the bond axis, leading to structural rigidity in molecules.
Factors Influencing Covalent Bond Strength
Several factors influence the strength of a covalent bond:
-
Bond length: The distance between the nuclei of two bonded atoms. Shorter bond lengths generally correlate with stronger bonds, as the atoms are held closer together by stronger attractive forces.
-
Electronegativity: The ability of an atom to attract electrons in a covalent bond. When atoms with similar electronegativities bond, the electrons are shared equally, forming a nonpolar covalent bond. When atoms with significantly different electronegativities bond, the electrons are shared unequally, leading to a polar covalent bond. In polar bonds, one atom carries a partial negative charge (δ-), and the other carries a partial positive charge (δ+).
-
Bond order: The number of electron pairs shared between two atoms. Higher bond orders generally indicate stronger bonds. A single bond has a bond order of 1, a double bond has a bond order of 2, and a triple bond has a bond order of 3.
-
Hybridization: The mixing of atomic orbitals to form hybrid orbitals, which are involved in covalent bonding. Different hybridization schemes lead to varying bond strengths and geometries. For instance, sp3 hybridized carbon atoms form stronger bonds than sp hybridized carbon atoms.
Types of Covalent Bonds: Exploring the Spectrum
The nature of covalent bonds encompasses a range of possibilities:
-
Nonpolar Covalent Bonds: These bonds form between atoms with similar electronegativities. The electron pair is shared equally between the atoms, resulting in a symmetrical distribution of charge. Examples include bonds between identical atoms (e.g., H-H in hydrogen gas) or bonds between atoms with very similar electronegativities (e.g., C-H bonds in many organic molecules).
-
Polar Covalent Bonds: These bonds form between atoms with different electronegativities. The electron pair is shared unequally, with the more electronegative atom attracting the electrons more strongly. This creates a dipole moment, a separation of positive and negative charge within the molecule. Examples include O-H bonds in water and C-O bonds in many organic molecules.
-
Coordinate Covalent Bonds (Dative Bonds): In these bonds, both electrons in the shared pair come from the same atom. This is common in molecules with lone pairs of electrons that can be donated to an atom lacking electrons. Examples include the bonds in ammonium ion (NH4+) and in many metal complexes.
Covalent Bonding in Different Contexts: Examples and Applications
Covalent bonds are ubiquitous in chemistry and biology. They are responsible for the formation of:
-
Organic Molecules: The vast majority of organic molecules are held together by covalent bonds. This includes hydrocarbons, carbohydrates, proteins, nucleic acids, and countless other vital biomolecules. The carbon atom's ability to form four covalent bonds allows for the construction of complex, diverse structures.
-
Inorganic Molecules: Many inorganic molecules are also held together by covalent bonds. Examples include water (H2O), carbon dioxide (CO2), ammonia (NH3), and silicon dioxide (SiO2), a major component of sand.
-
Polymers: Polymers, large molecules composed of repeating units, are often held together by covalent bonds between the repeating units. Examples include plastics, synthetic fibers, and natural polymers like cellulose and proteins.
-
Network Solids: Network solids are characterized by a continuous network of covalent bonds extending throughout the entire structure. Examples include diamond (a network of carbon atoms) and silicon carbide (SiC). These materials are typically very strong and hard.
Understanding Covalent Bonds: A Cornerstone of Chemistry
Covalent bonds are a fundamental concept in chemistry, explaining the structure and properties of countless molecules. Their formation is driven by the quest for stability, achieved through electron sharing and orbital overlap. Understanding the various types of covalent bonds, the factors affecting their strength, and their prevalence across different chemical contexts is crucial for anyone seeking a deep understanding of the chemical world. From the simplest molecules to the most complex biological systems, covalent bonds are the invisible glue that holds our universe together. The principles discussed here provide a solid foundation for further exploration of more advanced chemical concepts and applications.
Latest Posts
Latest Posts
-
Why Are Ionic Compounds Soluble In Water
Apr 06, 2025
-
How To Find Center Of Data
Apr 06, 2025
-
Type Of Molecule With An Uneven Distribution Of Electrons
Apr 06, 2025
-
Differential Rate Law Vs Integrated Rate Law
Apr 06, 2025
-
How Do You Test An Equation For Symmetry
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about A Covalent Bond Is Formed As A Result Of . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
