Single Double And Triple Covalent Bonds
Muz Play
Apr 05, 2025 · 7 min read
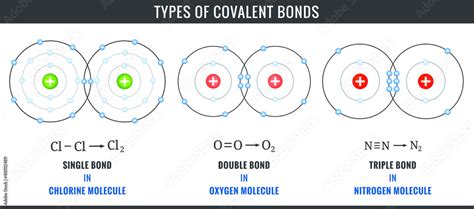
Table of Contents
Single, Double, and Triple Covalent Bonds: A Deep Dive into Chemical Bonding
Covalent bonds are the fundamental forces holding atoms together in molecules, forming the basis of countless compounds in the universe. Understanding the different types of covalent bonds – single, double, and triple – is crucial for grasping the properties and behaviors of these molecules. This comprehensive guide will explore the intricacies of each bond type, explaining their formation, characteristics, and significance in chemistry. We'll delve into their impact on molecular geometry, polarity, and reactivity, offering examples to solidify your understanding.
What is a Covalent Bond?
Before we dive into the specifics of single, double, and triple bonds, let's establish a foundational understanding of covalent bonding. A covalent bond forms when two atoms share one or more pairs of electrons. This sharing occurs to achieve a more stable electron configuration, typically resembling that of a noble gas (a full outer electron shell). Atoms achieve this stability by satisfying the octet rule (eight electrons in their valence shell), although there are exceptions, particularly with elements beyond the second row of the periodic table.
Unlike ionic bonds, which involve the transfer of electrons, covalent bonds involve the sharing of electrons. This sharing creates a strong attractive force between the atoms, holding them together as a molecule. The strength of this bond depends on several factors, including the number of shared electron pairs and the electronegativity difference between the atoms.
Single Covalent Bonds
A single covalent bond, also known as a sigma (σ) bond, is the simplest type of covalent bond. It involves the sharing of one pair of electrons (two electrons) between two atoms. This shared pair occupies a region of space between the nuclei of the bonded atoms.
Characteristics of Single Covalent Bonds:
- One shared electron pair: Only one pair of electrons is shared between the atoms.
- Rotation: Single bonds allow free rotation around the bond axis. This means the atoms can rotate relative to each other without breaking the bond.
- Bond length: Single bonds are relatively long compared to double and triple bonds.
- Bond strength: Single bonds are relatively weaker compared to double and triple bonds.
- Examples: The bond in methane (CH₄), where each carbon-hydrogen bond is a single bond, is a classic example. Ethane (C₂H₆) also features single bonds between the two carbon atoms and the hydrogen atoms. The bonds in most organic molecules are predominantly single bonds.
Understanding Sigma Bonds: An Orbital Perspective
At a deeper level, single covalent bonds are best understood using the concept of atomic orbitals. When two atoms approach each other, their atomic orbitals overlap. This overlap concentrates electron density between the nuclei, forming a sigma bond. The overlap is greatest along the internuclear axis, resulting in a strong bond. This explains the relative strength and stability associated with sigma bonds.
Double Covalent Bonds
A double covalent bond consists of two pairs of shared electrons (four electrons) between two atoms. This involves one sigma (σ) bond and one pi (π) bond.
Characteristics of Double Covalent Bonds:
- Two shared electron pairs: Two pairs of electrons are shared between the atoms.
- Restricted Rotation: The presence of the pi bond restricts rotation around the bond axis. Rotation would require breaking the pi bond, which requires significant energy.
- Shorter Bond Length: Double bonds are shorter than single bonds because the two pairs of electrons create a stronger attractive force between the nuclei.
- Higher Bond Strength: Double bonds are stronger than single bonds due to the additional electron density and the stronger attractive force.
- Examples: The carbon-oxygen bond in carbon dioxide (CO₂) is a double bond. Ethene (C₂H₄) possesses a double bond between the two carbon atoms, which significantly impacts its reactivity and shape.
Understanding Pi Bonds: Orbital Overlap
While the sigma bond in a double bond is formed by head-on overlap of atomic orbitals, the pi (π) bond results from the sideways overlap of p orbitals. This sideways overlap forms an electron cloud above and below the internuclear axis. This is a less effective overlap than the head-on overlap of sigma bonds, explaining why pi bonds are generally weaker than sigma bonds.
Triple Covalent Bonds
A triple covalent bond involves the sharing of three pairs of electrons (six electrons) between two atoms. It consists of one sigma (σ) bond and two pi (π) bonds.
Characteristics of Triple Covalent Bonds:
- Three shared electron pairs: Three pairs of electrons are shared between the atoms.
- Very Restricted Rotation: The presence of two pi bonds severely restricts rotation around the bond axis. Breaking these pi bonds to allow rotation requires substantial energy input.
- Shortest Bond Length: Triple bonds are the shortest among the three types due to the very strong attractive force between the nuclei resulting from the high electron density.
- Highest Bond Strength: Triple bonds are the strongest type of covalent bond due to the three shared electron pairs.
- Examples: The quintessential example is the nitrogen molecule (N₂), where a triple bond exists between the two nitrogen atoms, making it exceptionally stable and unreactive. Acetylene (C₂H₂) also contains a triple bond between the two carbon atoms.
Delving Deeper into Pi Bond Formation
As with double bonds, the sigma bond in a triple bond is formed from the head-on overlap of atomic orbitals. The two pi bonds, however, result from the sideways overlap of two pairs of p orbitals, creating two electron clouds above and below, and two electron clouds in front and behind the internuclear axis.
Comparing Single, Double, and Triple Bonds
| Feature | Single Bond | Double Bond | Triple Bond |
|---|---|---|---|
| Number of Bonds | 1 | 2 | 3 |
| Number of Electron Pairs | 1 | 2 | 3 |
| Bond Type | σ | σ + π | σ + 2π |
| Bond Length | Longest | Medium | Shortest |
| Bond Strength | Weakest | Medium | Strongest |
| Rotation | Free | Restricted | Highly Restricted |
Impact on Molecular Geometry and Polarity
The type of covalent bond significantly impacts a molecule's shape and polarity. Single bonds allow for greater flexibility in bond angles, leading to a wider range of molecular geometries. Double and triple bonds, with their restricted rotation, often impose more rigid structures.
Molecular polarity is also influenced by bond type. While the presence of a double or triple bond doesn't automatically dictate polarity, the electronegativity difference between the bonded atoms plays a crucial role. If the atoms have different electronegativities, the shared electrons will be drawn more towards the more electronegative atom, resulting in a polar bond. The overall polarity of the molecule depends on the arrangement of these polar bonds and the molecule's symmetry.
Bond Order and Reactivity
The bond order is the number of chemical bonds between a pair of atoms. It directly correlates with bond strength and length. A higher bond order (triple > double > single) indicates a stronger and shorter bond. This directly impacts the molecule's reactivity. Molecules with higher bond orders are typically less reactive because the bonds require more energy to break.
Applications and Examples in Organic and Inorganic Chemistry
Single, double, and triple bonds are ubiquitous in both organic and inorganic chemistry. Understanding these bonds is essential for predicting molecular properties and reactions.
Organic Chemistry: Alkanes are saturated hydrocarbons containing only single bonds between carbon atoms. Alkenes and alkynes contain double and triple bonds, respectively, which introduce unsaturation and enhanced reactivity. These functional groups profoundly affect the molecule's chemical behavior.
Inorganic Chemistry: Many inorganic compounds, including nitrogen gas (N₂), carbon dioxide (CO₂), and various metal complexes, feature multiple bonds influencing their properties and reactivity. The strong triple bond in nitrogen makes it very stable and unreactive, playing a crucial role in atmospheric chemistry.
Conclusion
The nature of the covalent bond – single, double, or triple – is a critical factor in determining the properties and behavior of molecules. The number of shared electron pairs, bond length, bond strength, and rotational freedom are all directly impacted by the bond type. Understanding these differences is crucial for predicting and interpreting the reactivity, molecular structure, and overall behavior of a vast range of chemical compounds, from simple diatomic molecules to complex organic and inorganic structures. By grasping the fundamental principles discussed here, you build a solid foundation for further exploration of advanced chemical concepts.
Latest Posts
Latest Posts
-
Does Proton And Neutron Have The Same Mass
Apr 05, 2025
-
Water Has High Heat Of Vaporization
Apr 05, 2025
-
Find The Tangent Plane To The Surface At The Point
Apr 05, 2025
-
What Are The Characteristics Of A Covalent Bond
Apr 05, 2025
-
Match Each Linear Equation With Its Graph
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Single Double And Triple Covalent Bonds . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
