What Are The Characteristics Of A Covalent Bond
Muz Play
Apr 05, 2025 · 7 min read
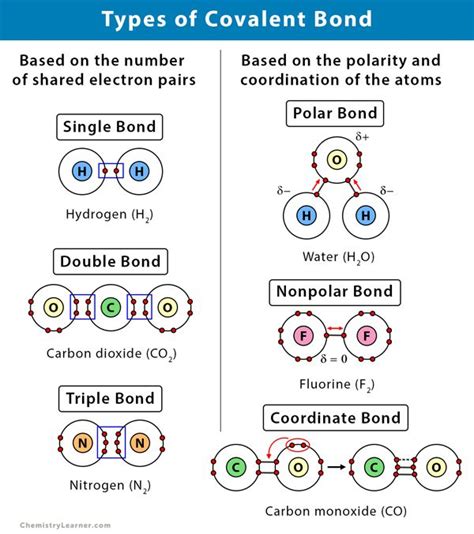
Table of Contents
What are the Characteristics of a Covalent Bond?
A covalent bond, a fundamental concept in chemistry, represents the sharing of electron pairs between atoms. Unlike ionic bonds, where electrons are transferred, covalent bonds involve a mutual attraction to the shared electrons, resulting in a stable molecular structure. Understanding the characteristics of a covalent bond is crucial for comprehending the properties of numerous compounds, from simple molecules like water to complex biomolecules like DNA. This comprehensive guide delves deep into the defining features of covalent bonds, exploring their formation, types, and influence on molecular properties.
Defining Covalent Bonds: A Shared Electron Pair Dance
At the heart of a covalent bond lies the shared electron pair. This shared pair of electrons is attracted to the positively charged nuclei of both atoms involved, creating a stable bond that holds the atoms together. This sharing effectively completes the outermost electron shell (valence shell) of each atom, achieving a more stable, lower-energy configuration – a principle often referred to as the octet rule (although exceptions exist, particularly for elements beyond the second row of the periodic table).
Key Characteristics:
-
Electron Sharing: The most defining characteristic. Atoms contribute one or more electrons to the shared pair. The number of shared electron pairs determines the bond order (single, double, or triple bonds).
-
Strong Bond Strength (relatively): While weaker than some ionic bonds, covalent bonds are still relatively strong, requiring significant energy to break. The strength of the bond depends on factors like the number of shared electron pairs and the electronegativity difference between the atoms.
-
Directional Bonds: Unlike ionic bonds, which are non-directional electrostatic attractions, covalent bonds are highly directional. The shared electron pair is localized between the two atoms, creating a specific bond angle and geometry. This directionality has a profound impact on the shape and properties of molecules.
-
Formation from Nonmetals: Covalent bonds predominantly form between nonmetal atoms. Nonmetals have high electronegativities, meaning they have a strong tendency to attract electrons. Sharing electrons is a more energetically favorable process than transferring them between nonmetals.
-
Low Melting and Boiling Points (generally): Covalent compounds often have relatively low melting and boiling points compared to ionic compounds. This is because the intermolecular forces (forces between molecules) are weaker than the strong electrostatic forces in ionic crystals. However, network covalent solids like diamond and silicon carbide are exceptions, possessing exceptionally high melting points due to their extensive three-dimensional networks of covalent bonds.
-
Poor Electrical Conductivity: Covalent compounds generally do not conduct electricity in either solid or liquid states. This is because there are no freely moving charged particles (ions or electrons) to carry the electric current. Exceptions include some molecules in aqueous solutions that can ionize, allowing for some degree of conductivity.
-
Solubility Variations: The solubility of covalent compounds varies greatly depending on the polarity of the molecule and the polarity of the solvent. Polar covalent molecules (those with uneven electron distribution) tend to dissolve in polar solvents (like water), while nonpolar covalent molecules dissolve in nonpolar solvents (like oil).
Types of Covalent Bonds: A Spectrum of Sharing
Covalent bonds aren't monolithic; they exhibit variation based on several factors, leading to distinct subtypes:
1. Nonpolar Covalent Bonds: Equal Sharing
In a nonpolar covalent bond, the electrons are shared almost equally between the two atoms. This occurs when the atoms involved have similar or identical electronegativities. The electronegativity difference is close to zero. Examples include bonds within diatomic molecules like H₂ (hydrogen), O₂ (oxygen), and N₂ (nitrogen).
2. Polar Covalent Bonds: Unequal Sharing
In a polar covalent bond, the electrons are shared unequally between the atoms. This asymmetry arises from a difference in electronegativity between the atoms. The atom with the higher electronegativity attracts the shared electron pair more strongly, creating a partial negative charge (δ-) on that atom and a partial positive charge (δ+) on the other atom. This leads to a dipole moment, a measure of the polarity of the bond. Water (H₂O) is a classic example, with oxygen having a higher electronegativity than hydrogen.
3. Coordinate Covalent Bonds (Dative Bonds): A One-Sided Contribution
A coordinate covalent bond, also known as a dative bond, is a special type of covalent bond where both shared electrons originate from the same atom. One atom acts as an electron pair donor (Lewis base), and the other atom acts as an electron pair acceptor (Lewis acid). Ammonium ion (NH₄⁺) is a good example; the nitrogen atom donates a lone pair to form a bond with a hydrogen ion (proton).
Factors Influencing Covalent Bond Strength
Several factors impact the strength of a covalent bond:
-
Atomic Size: Smaller atoms generally form stronger covalent bonds because the shared electrons are closer to the positively charged nuclei.
-
Electronegativity: A larger difference in electronegativity leads to a more polar bond. While polar bonds can be stronger than nonpolar bonds in some cases, extremely high electronegativity differences favor ionic bond formation.
-
Bond Order: Multiple bonds (double and triple bonds) are stronger than single bonds because they involve more shared electron pairs, leading to a stronger attraction between the atoms.
Covalent Bonds and Molecular Geometry: Shape Matters
The arrangement of atoms in a molecule, its geometry, is directly influenced by the number and arrangement of covalent bonds. This geometry has a significant impact on the molecule's physical and chemical properties. Concepts like VSEPR (Valence Shell Electron Pair Repulsion) theory help predict molecular geometry based on the electron pairs around the central atom. Molecular shapes such as linear, bent, trigonal planar, tetrahedral, and octahedral arise from different arrangements of electron pairs and bonds.
Covalent Bonds and Intermolecular Forces: Beyond the Bond
While covalent bonds hold atoms within a molecule, intermolecular forces are weaker attractions between different molecules. These forces influence the physical properties of substances, including boiling points, melting points, and solubility. Types of intermolecular forces include:
-
London Dispersion Forces (LDFs): Present in all molecules, these weak forces arise from temporary fluctuations in electron distribution.
-
Dipole-Dipole Forces: Occur between polar molecules due to the attraction between their permanent dipoles.
-
Hydrogen Bonding: A particularly strong type of dipole-dipole force that occurs when a hydrogen atom is bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) and is attracted to another electronegative atom in a nearby molecule.
Covalent Bonds in Organic Chemistry: The Foundation of Life
Covalent bonds are the cornerstone of organic chemistry, the study of carbon-containing compounds. Carbon's ability to form four covalent bonds allows for the formation of a vast array of organic molecules, including hydrocarbons, alcohols, aldehydes, ketones, carboxylic acids, and countless other functional groups. These molecules are the building blocks of life, forming proteins, carbohydrates, lipids, and nucleic acids.
Covalent Network Solids: An Exceptionally Strong Bond
Network covalent solids represent a distinct class of covalent compounds where atoms are covalently bonded in a continuous three-dimensional network. These materials are incredibly strong and have high melting points. Examples include diamond (carbon atoms bonded in a tetrahedral network) and silicon carbide (SiC).
Covalent Bonds and Resonance: Delocalized Electrons
In some molecules, the electron density is not localized to a single bond but is delocalized over multiple atoms. This phenomenon is known as resonance, and it occurs when multiple valid Lewis structures can be drawn for a molecule. The actual structure is a hybrid of these contributing resonance structures, resulting in greater stability. Benzene is a classic example of a molecule exhibiting resonance.
Applications of Covalent Bonding Understanding
Understanding the characteristics of covalent bonds has far-reaching implications across various fields:
-
Materials Science: Designing new materials with specific properties, such as strength, conductivity, and reactivity, relies on understanding the nature of covalent bonds.
-
Medicine: Developing drugs and understanding their interactions with biological systems often involves analyzing covalent bonds in biomolecules and drug molecules.
-
Environmental Science: Studying chemical reactions and their impact on the environment necessitates understanding covalent bonds and their role in various chemical processes.
-
Industrial Chemistry: Optimizing chemical reactions for industrial applications requires a deep understanding of covalent bond formation and reactivity.
Conclusion: The Ubiquity of Covalent Bonds
Covalent bonds are ubiquitous in nature and are essential for understanding the structure and properties of a vast array of molecules. From the simplest diatomic gases to the complex biomolecules that make up life, the sharing of electron pairs is a fundamental force shaping the world around us. A thorough grasp of the characteristics, types, and implications of covalent bonds is indispensable for anyone pursuing studies in chemistry, biology, materials science, and related fields. Further exploration into the nuances of bond energies, bond lengths, and the various spectroscopic techniques used to study covalent bonds can provide even deeper insights into this crucial area of chemistry.
Latest Posts
Latest Posts
-
How Many Valence Electrons Does Sulfer Have
Apr 05, 2025
-
Power Of A Quotient Property Examples
Apr 05, 2025
-
Which Electron Carrier S Function In The Citric Acid Cycle
Apr 05, 2025
-
Human Body Temperature In Kelvin Scale
Apr 05, 2025
-
Both State And Federal Courts Hear Matters That Involve
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Are The Characteristics Of A Covalent Bond . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
