State Of Matter That Has Definite Shape And Volume
Muz Play
Apr 02, 2025 · 7 min read
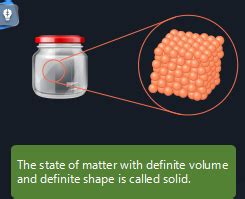
Table of Contents
The Solid State: A Deep Dive into Matter with Definite Shape and Volume
The world around us is a tapestry woven from diverse forms of matter, each exhibiting unique properties and behaviors. One fundamental categorization of matter divides it into four primary states: solid, liquid, gas, and plasma. This article focuses on the solid state of matter, characterized by its definite shape and volume. We will delve deep into the microscopic structure, macroscopic properties, different types of solids, and the fascinating phenomena that govern their behavior.
Understanding the Solid State: A Microscopic Perspective
At the heart of a solid's unique characteristics lies its microscopic structure. Unlike liquids and gases where particles move relatively freely, the constituent particles of a solid – atoms, ions, or molecules – are tightly packed and held together by strong intermolecular forces. This strong cohesive force is responsible for the solid's rigidity and resistance to change in shape or volume.
The Role of Intermolecular Forces
The strength of the intermolecular forces dictates the solidity of a material. These forces can be:
- Ionic Bonds: These strong electrostatic forces hold oppositely charged ions together, resulting in solids with high melting points and hardness, like sodium chloride (table salt).
- Covalent Bonds: Atoms share electrons to form strong covalent bonds, leading to solids with high melting points and often intricate structures, exemplified by diamond and quartz.
- Metallic Bonds: In metals, valence electrons are delocalized, forming a "sea" of electrons that surrounds positively charged metal ions. This results in solids that are often good conductors of heat and electricity, malleable, and ductile.
- Van der Waals Forces: These are weaker forces that arise from temporary fluctuations in electron distribution. They play a crucial role in the structure of molecular solids, such as ice and many organic compounds. These solids generally have lower melting points compared to those with ionic or covalent bonds.
- Hydrogen Bonds: A special type of dipole-dipole attraction that occurs when hydrogen is bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine). Hydrogen bonds are relatively strong and are responsible for the unique properties of water and many biological molecules.
Crystalline vs. Amorphous Solids
Solids are broadly classified into two categories based on their microscopic arrangement:
-
Crystalline Solids: These solids exhibit a highly ordered, three-dimensional arrangement of their constituent particles. This ordered structure, known as a crystal lattice, repeats periodically in all directions. Crystalline solids often possess well-defined shapes, sharp melting points, and anisotropic properties (meaning their properties vary with direction). Examples include diamonds, quartz, and table salt.
-
Amorphous Solids: In contrast to crystalline solids, amorphous solids lack a long-range ordered structure. Their constituent particles are arranged randomly, similar to a liquid but with much lower mobility. They generally do not have sharp melting points but rather soften over a range of temperatures. Examples include glass, rubber, and plastics.
Macroscopic Properties of Solids
The macroscopic properties of a solid, the properties we can observe directly, are a direct consequence of its microscopic structure and the nature of its intermolecular forces. Key properties include:
-
Shape and Volume: Solids possess a definite shape and volume, a defining characteristic that distinguishes them from liquids and gases. This is due to the strong intermolecular forces that hold the particles firmly in place.
-
Density: Solids generally have high densities compared to liquids and gases because their particles are tightly packed. Density varies depending on the type of solid and its crystal structure.
-
Melting Point: The temperature at which a solid transforms into a liquid is its melting point. This temperature is determined by the strength of the intermolecular forces. Stronger forces lead to higher melting points.
-
Boiling Point: While not directly related to the solid state itself, understanding the boiling point of a solid's liquid phase provides valuable insight into the intermolecular forces. Stronger intermolecular forces mean a higher boiling point.
-
Hardness: This refers to a solid's resistance to scratching or indentation. Hardness is strongly correlated with the strength of the intermolecular forces and the crystal structure.
-
Elasticity: This property describes a solid's ability to deform under stress and return to its original shape when the stress is removed. Elasticity is governed by the nature of the intermolecular forces and the crystal structure.
-
Brittleness: This property describes a solid's tendency to fracture or shatter upon impact. Brittleness is linked to the strength and directionality of the intermolecular forces, and the presence of defects in the crystal structure.
-
Malleability and Ductility: These properties relate to a solid's ability to be shaped by hammering (malleability) or drawn into wires (ductility). Metals, with their delocalized electrons, are particularly malleable and ductile.
Types of Solids
Solids can be further classified based on the type of bonding and the arrangement of particles:
-
Ionic Solids: These solids are formed by the electrostatic attraction between positively and negatively charged ions. They are generally hard, brittle, and have high melting points. Examples include NaCl (table salt), MgO (magnesium oxide), and CaF₂ (calcium fluoride).
-
Covalent Solids: These solids are formed by the sharing of electrons between atoms. They are generally very hard, have high melting points, and are poor conductors of electricity. Examples include diamond, graphite, and silicon dioxide (quartz).
-
Metallic Solids: These solids are composed of metal atoms held together by metallic bonds. They are generally good conductors of heat and electricity, malleable, ductile, and lustrous. Examples include iron, copper, gold, and aluminum.
-
Molecular Solids: These solids are composed of molecules held together by relatively weak intermolecular forces. They generally have low melting points and are poor conductors of electricity. Examples include ice (H₂O), dry ice (CO₂), and iodine (I₂).
Solid State Physics: A Deeper Look
Solid-state physics delves into the physical properties of solids at a fundamental level. This field explores:
-
Crystallography: The study of the arrangement of atoms in crystals. Techniques like X-ray diffraction are used to determine crystal structures.
-
Band Theory: This explains the electrical conductivity of solids by examining the energy levels of electrons in the solid. This theory differentiates conductors, insulators, and semiconductors.
-
Defects in Crystals: Imperfections in the crystal lattice, such as vacancies, interstitials, and dislocations, significantly affect the mechanical and electrical properties of solids.
-
Phase Transitions: The study of transitions between different solid phases, such as between different crystalline forms or between a solid and a liquid.
-
Nanomaterials: The study of materials with dimensions on the nanoscale (1-100 nanometers) exhibits unique properties that differ significantly from their bulk counterparts.
Applications of Solids
Solids are ubiquitous in our daily lives, playing a critical role in virtually every aspect of modern society. Their applications are vast and diverse, including:
-
Structural Materials: Steel, concrete, and wood are fundamental building materials in construction and infrastructure.
-
Electronic Components: Silicon and other semiconductors are essential components in microchips and other electronic devices.
-
Biomaterials: Solids such as titanium alloys and polymers are used in medical implants and prosthetics.
-
Optical Materials: Crystalline solids such as quartz and various glasses are used in lenses, prisms, and optical fibers.
-
Energy Storage: Solid-state batteries are being developed for various applications, from electric vehicles to portable electronics.
-
Catalysis: Solid catalysts are used in a wide range of chemical reactions, from industrial processes to biological systems.
Conclusion: The Ever-Evolving World of Solids
The solid state of matter, characterized by its definite shape and volume, is a realm of remarkable diversity and complexity. From the intricate structures of crystals to the amorphous nature of glass, solids exhibit a vast range of properties that make them indispensable for countless applications. Continued research in solid-state physics and materials science promises to unlock even more fascinating properties and lead to innovative technologies that will reshape our world. Understanding the fundamental principles governing the solid state is crucial for advancements in various fields, from electronics and energy to medicine and construction. The journey of exploring the solid state is far from over, promising exciting discoveries and applications in the years to come.
Latest Posts
Latest Posts
-
Heat Of Vaporization Of Water J Kg
Apr 03, 2025
-
What Happens To Plant Cells In A Hypertonic Solution
Apr 03, 2025
-
What Is A Disadvantage Of A Corporation
Apr 03, 2025
-
What Is Kf In Freezing Point Depression
Apr 03, 2025
-
What Are Some Disadvantages Of A Corporation
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about State Of Matter That Has Definite Shape And Volume . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
