Term Used For Compounds With Properties For Neutralizing Acids
Muz Play
Apr 05, 2025 · 5 min read
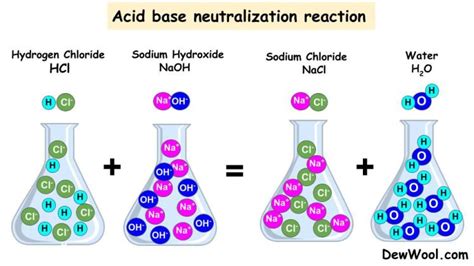
Table of Contents
Term Used for Compounds with Properties for Neutralizing Acids
Compounds with the property of neutralizing acids are broadly classified as bases or alkalis. While the terms are often used interchangeably, there's a subtle difference. Understanding this difference, along with the diverse applications and properties of these compounds, is crucial for various fields, from chemistry and manufacturing to environmental science and everyday life. This comprehensive article delves into the terminology, properties, reactions, and applications of these acid-neutralizing compounds.
Bases and Alkalis: A Clarification
The terms "base" and "alkali" are frequently used to describe substances capable of neutralizing acids. However, while all alkalis are bases, not all bases are alkalis. This distinction lies in their behavior in aqueous solutions:
-
Base: A base is any substance that can accept a proton (H⁺ ion) or donate a lone pair of electrons. This is a broader definition encompassing a wide range of chemical species. Bases can be soluble or insoluble in water.
-
Alkali: An alkali is a specific type of base that is soluble in water and produces hydroxide ions (OH⁻) when dissolved. These hydroxide ions are responsible for the alkaline nature of the solution, increasing its pH. Therefore, alkalis are a subset of bases.
Properties of Acid-Neutralizing Compounds
Acid-neutralizing compounds, whether bases or alkalis, exhibit several key properties:
1. pH Value
The most defining characteristic is their ability to raise the pH of a solution. Acids have a pH below 7, while bases have a pH above 7. Neutral solutions have a pH of 7. The stronger the base, the higher the pH it will raise the solution to. This pH change is a direct result of the neutralization reaction.
2. Reaction with Acids
The fundamental property is their reaction with acids, leading to a neutralization reaction. This reaction typically produces salt and water. For example, the reaction between a strong acid (like hydrochloric acid, HCl) and a strong base (like sodium hydroxide, NaOH) is:
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
This reaction is exothermic, meaning it releases heat. The strength of the acid and base influences the heat released.
3. Taste and Feel
Many bases have a bitter taste and a soapy or slippery feel. However, it's crucial to never taste or touch unknown chemicals, as many can be corrosive or poisonous.
4. Conductivity
Strong bases and alkalis, when dissolved in water, conduct electricity due to the presence of mobile ions (OH⁻ and the cation from the base). Weak bases conduct electricity less effectively.
Common Examples of Acid-Neutralizing Compounds
Numerous compounds effectively neutralize acids. Here are some examples, categorized for clarity:
1. Metal Hydroxides (Alkalis):
- Sodium hydroxide (NaOH): A strong alkali, commonly known as lye or caustic soda, used in many industrial processes and drain cleaners.
- Potassium hydroxide (KOH): Another strong alkali, similar in properties to NaOH, used in soap making and other chemical applications.
- Calcium hydroxide (Ca(OH)₂): A weaker alkali, commonly known as slaked lime, used in agriculture and construction. It's less soluble than NaOH or KOH.
- Magnesium hydroxide (Mg(OH)₂): A weak alkali, also known as milk of magnesia, used as an antacid.
2. Metal Carbonates and Bicarbonates:
- Sodium carbonate (Na₂CO₃): Also known as washing soda, used in detergents and water softening. Reacts with acids to produce carbon dioxide gas.
- Sodium bicarbonate (NaHCO₃): Baking soda, a weak base used in baking and as an antacid. Also reacts with acids to produce carbon dioxide gas.
- Calcium carbonate (CaCO₃): Limestone, a common mineral used in construction and as an antacid. Reacts with acids to produce carbon dioxide gas.
3. Metal Oxides:
- Calcium oxide (CaO): Quicklime, reacts with water to form calcium hydroxide. Used in construction and agriculture.
- Magnesium oxide (MgO): Used as a refractory material and in antacids.
4. Ammonia (NH₃):
- A weak base, commonly used in cleaning products and as a refrigerant. It reacts with water to form ammonium hydroxide (NH₄OH), a weak alkali.
Applications of Acid-Neutralizing Compounds
The applications of acid-neutralizing compounds are extensive and span various industries and everyday life:
1. Industrial Applications:
- Neutralizing industrial waste: Many industrial processes generate acidic waste. Bases are used to neutralize this waste before disposal to prevent environmental damage.
- Chemical manufacturing: Bases are essential reactants in numerous chemical processes, including the production of fertilizers, plastics, and pharmaceuticals.
- Water treatment: Alkalis are used to adjust the pH of water, making it suitable for drinking or industrial purposes.
- Soap and detergent manufacturing: Alkalis play a vital role in the saponification process, which converts fats and oils into soap.
2. Environmental Applications:
- Soil remediation: Alkalis are used to neutralize acidic soils, making them suitable for agriculture.
- Acid rain mitigation: While not directly neutralizing acid rain in the atmosphere, bases can help mitigate the effects on water bodies and soil.
- Wastewater treatment: Bases are used to adjust the pH of wastewater before discharge to protect aquatic life.
3. Everyday Applications:
- Antacids: Many over-the-counter antacids contain weak bases like magnesium hydroxide or calcium carbonate to neutralize stomach acid.
- Baking: Sodium bicarbonate (baking soda) is a crucial ingredient in baking, reacting with acidic ingredients to produce carbon dioxide gas, making baked goods rise.
- Cleaning products: Many cleaning products contain bases to remove grease and grime, which are often acidic.
Safety Precautions
Working with acid-neutralizing compounds requires careful attention to safety precautions:
- Protective equipment: Always wear appropriate protective equipment, including gloves, goggles, and lab coats, when handling bases.
- Ventilation: Ensure adequate ventilation when working with bases, especially strong alkalis, as some can release harmful fumes.
- Dilution: Always add bases to water, never water to bases, as this can cause a violent exothermic reaction.
- Disposal: Dispose of bases according to local regulations to avoid environmental damage.
Conclusion: The Importance of Acid-Neutralizing Compounds
Acid-neutralizing compounds, encompassing bases and alkalis, are indispensable in a wide range of applications, from industrial processes and environmental remediation to everyday products. Understanding their properties, reactions, and safe handling is crucial for anyone working with or encountering these substances. Their importance in maintaining a balanced environment and facilitating various industrial processes cannot be overstated. The ongoing research and development in this field continue to expand the applications and refine the understanding of these vital chemical compounds. This article serves as a comprehensive overview, providing a solid foundation for further exploration into the fascinating world of acid-neutralization.
Latest Posts
Latest Posts
-
Final Electron Acceptor In Electron Transport Chain
Apr 06, 2025
-
Accessory Structures Of The Integumentary System
Apr 06, 2025
-
How Do Water Particles Move In A Wave
Apr 06, 2025
-
Why Is Density An Intensive Property
Apr 06, 2025
-
Which Of The Following Bones Belong To The Axial Skeleton
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Term Used For Compounds With Properties For Neutralizing Acids . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
