The Final Electron Acceptor Of Cellular Respiration Is
Muz Play
Apr 02, 2025 · 6 min read
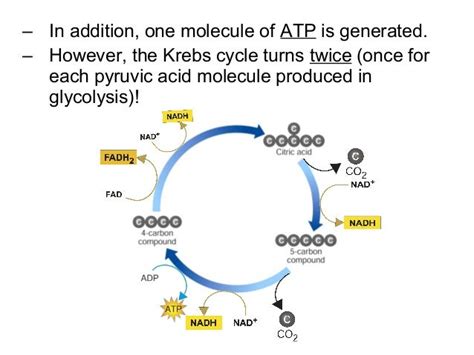
Table of Contents
The Final Electron Acceptor of Cellular Respiration Is… Oxygen! Understanding the Crucial Role of Oxygen in Energy Production
Cellular respiration, the process by which cells break down glucose to generate energy in the form of ATP (adenosine triphosphate), is fundamental to life as we know it. This intricate metabolic pathway involves a series of redox reactions, where electrons are passed from one molecule to another. Understanding the final electron acceptor in this chain is crucial to grasping the entire process and its implications for life. The answer, simply put, is oxygen. But the story goes far beyond this simple statement. This article will delve deep into the significance of oxygen as the terminal electron acceptor, exploring its role in the electron transport chain, its impact on ATP production, and the consequences of its absence.
The Electron Transport Chain: A Cascade of Redox Reactions
Cellular respiration can be broadly divided into four main stages: glycolysis, pyruvate oxidation, the Krebs cycle (also known as the citric acid cycle), and oxidative phosphorylation. While the first three stages generate a small amount of ATP directly, the majority of ATP synthesis occurs during oxidative phosphorylation, which takes place in the inner mitochondrial membrane. This stage is where the final electron acceptor, oxygen, plays its crucial role.
Oxidative phosphorylation centers around the electron transport chain (ETC), a series of protein complexes embedded within the inner mitochondrial membrane. These complexes, labeled Complex I through Complex IV, act as electron carriers, sequentially accepting and donating electrons. Electrons enter the ETC from electron carriers like NADH and FADH2, generated during glycolysis and the Krebs cycle. As electrons move down the chain, they lose energy, which is harnessed to pump protons (H+) from the mitochondrial matrix into the intermembrane space, establishing a proton gradient.
This proton gradient is essential because it drives chemiosmosis, the process by which ATP synthase, an enzyme located in the inner mitochondrial membrane, utilizes the energy stored in the gradient to synthesize ATP from ADP and inorganic phosphate (Pi). The flow of protons back into the matrix through ATP synthase powers the rotation of a molecular motor, facilitating the phosphorylation of ADP to ATP.
The Role of Oxygen: The Final Electron Destination
The electron transport chain wouldn't function without a final electron acceptor to receive the electrons at the end of the chain. This is where oxygen comes into the picture. Oxygen is highly electronegative, meaning it has a strong affinity for electrons. Complex IV, also known as cytochrome c oxidase, reduces oxygen (O2) to water (H2O), thereby accepting the electrons and completing the electron transport chain. This reaction is essential because it prevents the ETC from becoming "backed up," ensuring the continuous flow of electrons and the generation of the proton gradient.
Without oxygen as the final electron acceptor, the electron transport chain would halt. The buildup of reduced electron carriers (NADH and FADH2) would prevent further oxidation of glucose and significantly reduce ATP production. This is precisely what happens during anaerobic respiration, where alternative electron acceptors are used, resulting in far less ATP production.
The Significance of Oxygen in ATP Synthesis
The amount of ATP produced during cellular respiration is directly linked to the presence of oxygen. The process of oxidative phosphorylation, driven by the electron transport chain and oxygen's role as the final electron acceptor, is responsible for the vast majority of ATP generated. In contrast, the earlier stages of cellular respiration (glycolysis and the Krebs cycle) produce a comparatively small amount of ATP. Therefore, oxygen is crucial for efficient ATP generation, providing the energy necessary for cellular processes.
The efficiency of ATP production is significantly influenced by the electron carriers. NADH, generated in glycolysis and the Krebs cycle, donates its electrons earlier in the ETC, resulting in a greater proton gradient and, consequently, more ATP molecules synthesized. FADH2, another electron carrier, enters the ETC at a later stage, resulting in less ATP production per molecule.
The exact number of ATP molecules produced per glucose molecule varies depending on the shuttle systems used to transport NADH from glycolysis into the mitochondria. However, the overall yield is significantly higher when oxygen is present as the final electron acceptor. In aerobic conditions, the complete oxidation of one glucose molecule can yield approximately 30-32 ATP molecules, a considerable amount compared to the few ATP molecules produced under anaerobic conditions.
Anaerobic Respiration: Alternative Electron Acceptors
When oxygen is absent, cells resort to anaerobic respiration, also known as fermentation. This process allows for the continuation of glycolysis, albeit at a much lower ATP yield. However, since the ETC is halted without oxygen, the reduced electron carriers (NADH and FADH2) accumulate. To regenerate NAD+, which is necessary for glycolysis to proceed, cells utilize alternative electron acceptors.
Common examples include:
- Lactic acid fermentation: Pyruvate is reduced to lactate, regenerating NAD+ and allowing glycolysis to continue. This is a common process in muscle cells during strenuous exercise when oxygen supply is limited.
- Alcoholic fermentation: Pyruvate is converted to acetaldehyde, which is then reduced to ethanol, regenerating NAD+. This process is used by yeast and certain bacteria.
These fermentation pathways produce only a small amount of ATP (2 ATP molecules per glucose molecule), significantly less than the ATP produced during aerobic respiration. The lack of oxygen significantly restricts the energy available for cellular processes.
The Consequences of Oxygen Deprivation
The absence of oxygen has profound consequences for cellular function and overall health. Oxygen deprivation, or hypoxia, can lead to a variety of problems, including:
- Reduced ATP production: The drastic reduction in ATP synthesis leads to impaired cellular function and energy depletion.
- Cellular damage: The accumulation of reactive oxygen species (ROS) due to incomplete reduction of oxygen can damage cellular components like DNA, proteins, and lipids.
- Metabolic acidosis: Lactic acid fermentation can lead to a buildup of lactic acid, resulting in metabolic acidosis, which can further impair cellular function.
- Tissue damage and death: Prolonged hypoxia can lead to tissue damage and cell death, potentially resulting in organ failure.
Conditions like heart attacks, strokes, and severe infections can cause hypoxia, highlighting the critical importance of oxygen in maintaining proper cellular function and overall health.
Oxygen and the Evolution of Life
The evolution of oxygenic photosynthesis significantly altered the course of life on Earth. The emergence of photosynthetic organisms capable of releasing oxygen as a byproduct revolutionized the planet's atmosphere. This increase in atmospheric oxygen paved the way for the evolution of aerobic respiration, a far more efficient process of energy production than anaerobic respiration. The higher ATP yield from aerobic respiration allowed for the development of more complex multicellular organisms. Oxygen's role as the terminal electron acceptor was pivotal in the diversification and complexity of life on Earth.
Conclusion: Oxygen's Indispensable Role in Cellular Respiration
In conclusion, oxygen serves as the final electron acceptor in cellular respiration, a crucial role that ensures efficient ATP production. Its high electronegativity allows for the complete oxidation of glucose and the generation of a large proton gradient, which drives ATP synthesis. Without oxygen, the electron transport chain would cease, dramatically reducing ATP production and leading to impaired cellular function. Oxygen's role is not just limited to energy production; it’s fundamental to the very survival and complexity of life, highlighting its significance in the grand scheme of biological processes. Further research into the intricacies of oxidative phosphorylation and the electron transport chain continues to unveil new insights into this fundamental process of life.
Latest Posts
Latest Posts
-
Tangential And Normal Components Of Acceleration Calculator
Apr 03, 2025
-
Does Higher Bond Order Mean Stronger Bond
Apr 03, 2025
-
Does Sohcahtoa Work On Non Right Triangles
Apr 03, 2025
-
What Are The Four Main Types Of Context
Apr 03, 2025
-
How To Know If A Graph Is Symmetric
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Final Electron Acceptor Of Cellular Respiration Is . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
