Two Atoms Always Represent The Same Element If They Have
Muz Play
Apr 03, 2025 · 6 min read
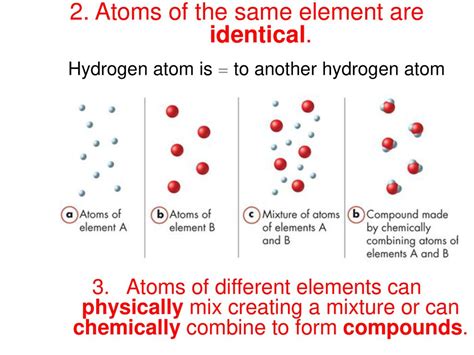
Table of Contents
Two Atoms Always Represent the Same Element If They Have the Same Number of Protons
Understanding the fundamental building blocks of matter is crucial to grasping the complexities of chemistry and physics. At the heart of this understanding lies the atom, the smallest unit of an element that retains its chemical properties. But what truly defines an element, and how do we know when two atoms represent the same element? The answer lies in the number of protons within their nucleus.
The Defining Characteristic: Atomic Number
Atoms, while incredibly small, are composed of even smaller subatomic particles: protons, neutrons, and electrons. Protons carry a positive electrical charge, neutrons are electrically neutral, and electrons carry a negative charge. These particles are arranged in a specific manner: protons and neutrons reside in the atom's dense central core, the nucleus, while electrons orbit the nucleus in shells or energy levels.
The atomic number of an element is the defining characteristic that determines its identity. This number represents the number of protons found in the nucleus of an atom of that element. Crucially, two atoms always represent the same element if they have the same atomic number, meaning they possess the same number of protons.
Isotopes: The Same Element, Different Neutrons
While the number of protons dictates the element, the number of neutrons can vary. Atoms of the same element with different numbers of neutrons are called isotopes. Isotopes have the same atomic number (same number of protons) but different mass numbers (the sum of protons and neutrons).
For example, carbon (C) has an atomic number of 6, meaning all carbon atoms have six protons. However, carbon exists in several isotopic forms: carbon-12 (⁶C), carbon-13 (¹³C), and carbon-14 (¹⁴C). These isotopes all have six protons, but they differ in the number of neutrons: six neutrons in carbon-12, seven in carbon-13, and eight in carbon-14. Despite these differences in neutron count, they all remain carbon atoms because they share the same atomic number.
The different isotopes of an element often have slightly varying properties, especially in terms of mass and radioactivity. Carbon-14, for instance, is radioactive and used in radiocarbon dating, while carbon-12 and carbon-13 are stable isotopes. However, their chemical behavior remains largely similar because the number of electrons, which determine chemical reactivity, is directly influenced by the number of protons.
The Role of Electrons in Chemical Behavior
While protons define the element, electrons play a crucial role in determining an element's chemical properties and how it interacts with other elements. Electrons occupy specific energy levels or shells surrounding the nucleus. The outermost shell, known as the valence shell, contains the valence electrons. These electrons are the ones involved in chemical bonding and reactions.
The number of valence electrons directly relates to an element's reactivity. Elements tend to react in ways that achieve a stable electron configuration, often a full valence shell. This drives the formation of chemical bonds, such as ionic bonds (transfer of electrons) and covalent bonds (sharing of electrons). Because the number of protons dictates the number of electrons in a neutral atom, the atomic number indirectly influences the chemical behavior of an element.
Ions: Atoms with a Charge Imbalance
Atoms can gain or lose electrons, resulting in the formation of ions. Ions are charged atoms or molecules because they have an unequal number of protons and electrons. Cations are positively charged ions (more protons than electrons), while anions are negatively charged ions (more electrons than protons).
The formation of ions significantly impacts the chemical behavior of an atom. For example, sodium (Na) readily loses one electron to become a Na⁺ ion, while chlorine (Cl) readily gains one electron to become a Cl⁻ ion. The electrostatic attraction between Na⁺ and Cl⁻ ions leads to the formation of sodium chloride (NaCl), common table salt.
Importantly, even though an ion has gained or lost electrons, it still represents the same element because the number of protons in its nucleus remains unchanged. A sodium ion (Na⁺) is still sodium, and a chloride ion (Cl⁻) is still chlorine.
Beyond Protons: Mass Number and Isotopic Abundance
While the atomic number definitively identifies an element, the mass number and isotopic abundance provide additional information about the different forms of that element.
The mass number of an atom is the sum of its protons and neutrons. As mentioned earlier, isotopes of the same element have the same atomic number but different mass numbers due to variations in neutron count.
Isotopic abundance refers to the relative proportion of each isotope of an element found in nature. For instance, carbon-12 is the most abundant isotope of carbon, making up about 98.9% of naturally occurring carbon. Carbon-13 makes up about 1.1%, and carbon-14 is present in trace amounts. These abundances are important considerations in various applications, including mass spectrometry and radiocarbon dating.
Understanding isotopic abundance allows us to calculate the average atomic mass of an element, which is a weighted average based on the mass of each isotope and its relative abundance. This average atomic mass is often used in stoichiometric calculations and other chemical computations.
Applications and Significance
The principle that two atoms represent the same element if they have the same number of protons is fundamental to many areas of science and technology:
-
Nuclear Chemistry and Physics: Understanding isotopes is crucial in nuclear chemistry and physics, as different isotopes exhibit varying nuclear properties, including radioactivity and stability. This understanding is applied in nuclear medicine, nuclear power generation, and radioisotope dating techniques.
-
Analytical Chemistry: Various analytical techniques, such as mass spectrometry, are used to determine the isotopic composition of samples. This information can be used for isotopic tracing, identifying sources of pollutants, and determining the age of materials.
-
Material Science: The properties of materials are often influenced by the isotopic composition of the elements they contain. For example, the use of specific isotopes in semiconductors can affect their electronic properties.
-
Geochemistry and Cosmochemistry: Isotopic ratios in rocks and meteorites provide valuable information about the age of the Earth, the formation of the solar system, and the processes that have shaped our planet.
-
Environmental Science: Isotopic tracing is a powerful tool for studying environmental processes, including nutrient cycling, water flow, and the movement of pollutants.
Conclusion: The Proton's Reign
The number of protons in an atom's nucleus is the ultimate determinant of its elemental identity. Two atoms always represent the same element if, and only if, they possess the same number of protons. While the number of neutrons can vary, resulting in different isotopes, the fundamental properties and chemical behavior of an element are primarily defined by its atomic number, reflecting the influence of protons on electron configuration and chemical reactivity. This fundamental concept forms the bedrock of our understanding of matter and underpins countless applications across various scientific disciplines. The proton, therefore, reigns supreme as the defining characteristic that distinguishes one element from another.
Latest Posts
Latest Posts
-
Is Childbirth A Positive Or Negative Feedback
Apr 04, 2025
-
Number Of Atoms In Simple Cubic Unit Cell
Apr 04, 2025
-
What Is The Purpose Of Persuasive Essay
Apr 04, 2025
-
Determine The Oxidation State Of Each Species
Apr 04, 2025
-
How To Find The Domain Of A Multivariable Function
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Two Atoms Always Represent The Same Element If They Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
