Two Different Ionic Compounds Each Contain
Muz Play
Apr 02, 2025 · 6 min read
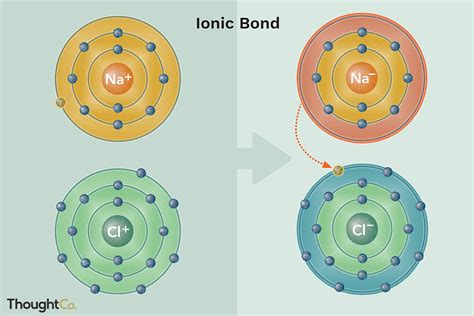
Table of Contents
Exploring Two Distinct Ionic Compounds: Sodium Chloride (NaCl) and Calcium Carbonate (CaCO₃)
Ionic compounds are fundamental substances in chemistry, formed through the electrostatic attraction between oppositely charged ions—cations (positively charged) and anions (negatively charged). Understanding their properties, formation, and applications is crucial in various fields, from medicine and materials science to environmental studies. This article delves into the fascinating world of ionic compounds, focusing on two distinct examples: sodium chloride (NaCl), common table salt, and calcium carbonate (CaCO₃), a ubiquitous component of limestone and seashells. We'll explore their unique characteristics, contrasting their structures, properties, and everyday applications.
Sodium Chloride (NaCl): The Everyday Wonder
Sodium chloride, more commonly known as table salt, is perhaps the most recognizable ionic compound. Its simple cubic structure and readily available nature make it an excellent case study for understanding ionic bonding and crystal structures.
Formation and Structure:
NaCl forms through an ionic bond between a sodium cation (Na⁺) and a chloride anion (Cl⁻). Sodium, an alkali metal, readily loses one electron to achieve a stable octet configuration, forming a positively charged ion. Chlorine, a halogen, readily gains one electron to achieve a stable octet, forming a negatively charged ion. The strong electrostatic attraction between these oppositely charged ions results in the formation of a crystalline structure. This structure is characterized by a repeating cubic arrangement, where each sodium ion is surrounded by six chloride ions, and vice versa. This arrangement maximizes electrostatic attraction and minimizes repulsion, leading to a stable, three-dimensional lattice. The crystal structure is remarkably strong, contributing to the hardness of sodium chloride crystals.
Properties of Sodium Chloride:
- High Melting and Boiling Points: The strong electrostatic forces between the ions require significant energy to overcome, resulting in high melting (801 °C) and boiling (1413 °C) points. This is typical of ionic compounds due to the strong ionic bonds.
- Solubility in Water: NaCl is highly soluble in water. The polar water molecules effectively surround and interact with the charged ions, weakening the ionic bonds and allowing the ions to dissolve. This solubility is crucial for its biological functions and various industrial applications.
- Electrical Conductivity: While solid NaCl is an insulator, molten NaCl and aqueous solutions of NaCl are good conductors of electricity. This is because the ions are free to move and carry electric charge when the compound is molten or dissolved.
- Brittle Nature: The strong ionic bonds, while providing structural strength, also make NaCl brittle. When subjected to stress, the aligned ions repel each other, causing the crystal to fracture along cleavage planes.
Applications of Sodium Chloride:
The widespread applications of NaCl stem from its unique properties and abundant availability. These include:
- Food Preservation: Salt has been used for centuries as a food preservative, inhibiting the growth of microorganisms by osmosis.
- Seasoning and Flavoring: Salt's taste and ability to enhance flavors make it an essential ingredient in countless culinary preparations.
- De-icing Agent: NaCl is commonly used to melt ice and snow on roads and pavements during winter. The dissolved ions lower the freezing point of water.
- Industrial Uses: NaCl is used in various industrial processes, including the production of chlorine, sodium hydroxide, and other chemicals. It’s also utilized in the manufacturing of soaps, detergents, and other products.
- Medical Applications: NaCl is a crucial component of intravenous fluids used in medical settings for hydration and electrolyte balance.
Calcium Carbonate (CaCO₃): The Building Block of Life
Calcium carbonate, a compound found extensively in nature, serves as a vital building block for various biological structures and geological formations. Its unique properties and chemical behavior contribute significantly to its diverse roles in the environment and various industries.
Formation and Structure:
Calcium carbonate is formed by the ionic bonding between a calcium cation (Ca²⁺) and a carbonate anion (CO₃²⁻). Calcium, an alkaline earth metal, loses two electrons to form a divalent cation. The carbonate ion is a polyatomic anion with a 2- charge, carrying a resonance structure which provides stability. The electrostatic attraction between these ions leads to the formation of several different crystalline structures, most notably calcite and aragonite. Calcite is the most common form, exhibiting a trigonal crystal system, while aragonite features an orthorhombic structure. Both forms exhibit strong ionic bonding within their crystalline structures.
Properties of Calcium Carbonate:
- Insolubility in Water: Unlike NaCl, CaCO₃ is relatively insoluble in water. This low solubility contributes to its role in geological formations and its use in various applications.
- High Melting Point: Similar to NaCl, CaCO₃ has a high melting point (around 825 °C) due to the strong electrostatic attractions between the calcium and carbonate ions. However, it decomposes before reaching its true melting point, releasing carbon dioxide gas.
- Reaction with Acids: CaCO₃ readily reacts with acids, producing carbon dioxide gas, water, and a soluble calcium salt. This reaction is commonly used to identify the presence of carbonates. The equation for this reaction with hydrochloric acid is: CaCO₃(s) + 2HCl(aq) → CaCl₂(aq) + H₂O(l) + CO₂(g)
- Hardness and Abrasiveness: The crystalline structure of CaCO₃ contributes to its hardness and abrasive properties, making it useful in various applications like polishing and cleaning.
Applications of Calcium Carbonate:
The diverse applications of CaCO₃ reflect its abundance in nature and its unique properties:
- Building Materials: Limestone, a sedimentary rock primarily composed of CaCO₃, is extensively used in construction as a building material, for cement production, and as a component of concrete.
- Antacids: Calcium carbonate is a common ingredient in antacids, neutralizing stomach acid due to its reaction with hydrochloric acid.
- Dietary Supplements: Calcium carbonate is used as a calcium supplement to prevent and treat calcium deficiency.
- Paper Manufacturing: CaCO₃ is used as a filler in paper production to improve brightness and opacity.
- Pigments and Paints: Ground calcium carbonate is used as a pigment and filler in paints and coatings.
- Agricultural Applications: It can be used to adjust soil pH, providing calcium for plant growth.
- Toothpaste: Calcium carbonate is used as an abrasive agent in many toothpastes.
Contrasting NaCl and CaCO₃: A Comparative Overview
While both NaCl and CaCO₃ are ionic compounds, their properties and applications differ significantly. Here's a comparative overview:
| Feature | Sodium Chloride (NaCl) | Calcium Carbonate (CaCO₃) |
|---|---|---|
| Structure | Simple cubic | Calcite (trigonal), Aragonite (orthorhombic) |
| Solubility | Highly soluble in water | Relatively insoluble in water |
| Melting Point | 801 °C | Decomposes before melting (around 825°C) |
| Reaction with Acids | No significant reaction | Reacts readily, producing CO₂ |
| Hardness | Relatively soft | Relatively hard |
| Applications | Food preservation, de-icing, industrial processes | Building materials, antacids, dietary supplements |
Conclusion
Sodium chloride and calcium carbonate, despite both being ionic compounds, showcase the vast diversity within this class of chemical substances. Their contrasting properties and applications highlight the importance of understanding the nuances of ionic bonding, crystal structure, and the influence of these factors on the physical and chemical behavior of materials. From the everyday utility of table salt to the geological significance of limestone, these compounds play crucial roles in our lives and the world around us. Further exploration into the vast array of ionic compounds reveals an even richer tapestry of fascinating chemistry and impactful applications across various fields. The study of these compounds serves as a foundation for a deeper understanding of materials science, geology, and many other scientific disciplines.
Latest Posts
Latest Posts
-
What Is A Word Equation In Chemistry
Apr 03, 2025
-
How Many A Columns Are On The Periodic Table
Apr 03, 2025
-
Kelvin Celsius And Fahrenheit Are Three Types Of Scales
Apr 03, 2025
-
Difference Between Applied And Basic Science
Apr 03, 2025
-
Identify The Characteristics Of A Spontaneous Reaction
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Two Different Ionic Compounds Each Contain . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
