Two Glucose Molecules Joined Together Form
Muz Play
Apr 03, 2025 · 6 min read
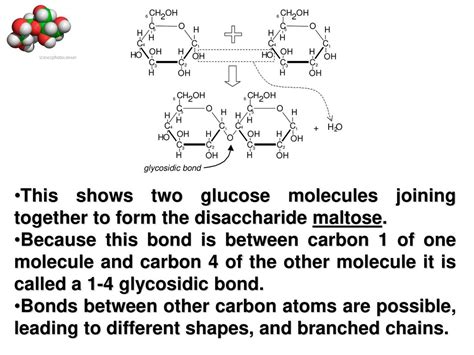
Table of Contents
Two Glucose Molecules Joined Together Form: A Deep Dive into Disaccharides and Glycosidic Bonds
When two glucose molecules join together, they form a disaccharide. This seemingly simple process is fundamental to biochemistry and plays a crucial role in energy storage and metabolism in living organisms. Understanding how this happens, the types of disaccharides formed, and their significance is essential for comprehending many biological processes. This article explores the fascinating world of disaccharides, focusing specifically on the joining of two glucose molecules.
The Chemistry of Joining Glucose Molecules: Glycosidic Bonds
The union of two glucose molecules isn't a simple fusion; it involves the formation of a glycosidic bond. This covalent bond forms between the hemiacetal group of one glucose molecule and a hydroxyl group of the other. This reaction is a dehydration synthesis, meaning a water molecule (H₂O) is released during the bond formation.
Let's break it down:
-
Hemiacetal group: Glucose exists primarily in a cyclic form, either α or β, due to the internal reaction of its aldehyde group with a hydroxyl group. This creates a hemiacetal group, characterized by a carbon atom bonded to both an -OH group and an -OR group (where R is the rest of the glucose molecule).
-
Hydroxyl group (-OH): Glucose molecules contain several hydroxyl groups, each capable of participating in the glycosidic bond formation. The specific hydroxyl group involved influences the type of disaccharide formed.
-
Dehydration Synthesis: The reaction involves the removal of a hydroxyl group (-OH) from one glucose molecule and a hydrogen atom (H) from the other, forming water. The remaining oxygen atom bridges the two glucose molecules, creating the glycosidic bond.
α-1,4-Glycosidic Bond: Maltose
When the glycosidic bond forms between the carbon atom at position 1 (C1) of one glucose molecule (in its α form) and the carbon atom at position 4 (C4) of the other glucose molecule, the resulting disaccharide is maltose. Maltose is a reducing sugar, meaning it has a free hemiacetal group that can be oxidized. This is because only one of the glucose units is involved in the glycosidic linkage, leaving the other free to act as a reducing agent.
Key Characteristics of Maltose:
- Source: Found in germinating grains, malt beverages, and partially digested starches.
- Structure: Two α-D-glucose units joined by an α-1,4-glycosidic bond.
- Sweetness: Moderately sweet.
- Digestion: Easily digested by the enzyme maltase, breaking it down into two glucose molecules.
β-1,4-Glycosidic Bond: Cellobiose
If the glycosidic bond forms between the C1 of one glucose molecule (in its β form) and the C4 of the other, the disaccharide formed is cellobiose. Unlike maltose, cellobiose is a less common disaccharide. It's a component of cellulose and is not easily digested by humans because they lack the necessary enzyme, cellulase. Cellulase is found in the digestive systems of certain animals, such as ruminants (cows, goats, sheep), that rely on symbiotic microorganisms to break down cellulose.
Key Characteristics of Cellobiose:
- Source: A breakdown product of cellulose.
- Structure: Two β-D-glucose units joined by a β-1,4-glycosidic bond.
- Sweetness: Slightly sweet.
- Digestion: Not easily digested by humans.
Beyond Maltose and Cellobiose: Other Possibilities
While maltose and cellobiose are the most common disaccharides formed from two glucose molecules, other configurations are theoretically possible depending on which hydroxyl groups participate in the glycosidic bond formation. These might involve linkages at different carbon positions (e.g., 1-6 linkages) or involve a combination of α and β glucose units. However, such disaccharides are less prevalent in nature. The prevalence of maltose and cellobiose highlights the importance of specific structural arrangements in biological systems.
The Importance of Disaccharides in Biology
Disaccharides serve several crucial roles in living organisms:
-
Energy Source: Maltose, in particular, serves as an easily accessible energy source. Its breakdown into glucose provides readily available fuel for cellular respiration.
-
Energy Storage: Disaccharides, particularly in their polysaccharide forms (many disaccharide units linked together), serve as significant energy storage molecules. Think of starch (amylose and amylopectin) in plants and glycogen in animals. These are essentially long chains of glucose molecules linked by glycosidic bonds.
-
Structural Components: While less common than starch and glycogen, certain disaccharides contribute to structural components in organisms. Cellobiose, a component of cellulose, is a prime example. Cellulose is the primary structural component of plant cell walls, providing rigidity and support.
-
Transport: Sugars are transported in plants in the form of sucrose, a disaccharide of glucose and fructose, not strictly two glucose units. The use of disaccharides provides efficient transport mechanisms, preventing the osmotic effects of large amounts of monosaccharides.
The Role of Enzymes in Disaccharide Metabolism
The digestion and metabolism of disaccharides rely heavily on the action of specific enzymes. These enzymes catalyze the hydrolysis of the glycosidic bond, breaking the disaccharide into its constituent monosaccharides. For example:
- Maltase: Breaks down maltose into two glucose molecules.
- Cellulase: Breaks down cellobiose and cellulose into glucose (humans lack this enzyme).
- Lactase: Breaks down lactose (glucose and galactose) into glucose and galactose. Lactose intolerance results from a deficiency in lactase.
These enzymes are crucial for energy acquisition and efficient utilization of carbohydrates in living organisms. The specific enzymes present in an organism's digestive system determine its ability to utilize different disaccharides.
Further Exploration: Polysaccharides
The joining of two glucose molecules represents a fundamental step toward understanding larger carbohydrate structures. Repeated glycosidic linkages between glucose units lead to the formation of polysaccharides, which include:
- Starch: A major energy storage polysaccharide in plants.
- Glycogen: The main energy storage polysaccharide in animals.
- Cellulose: The primary structural polysaccharide in plant cell walls.
Polysaccharides possess unique properties dictated by the type and arrangement of glycosidic bonds within their structures. Their diverse roles highlight the versatility of glucose and the importance of glycosidic bond formation in biological systems.
Conclusion: The Significance of a Simple Bond
The seemingly simple act of two glucose molecules joining together to form a disaccharide carries profound implications for life. The resulting disaccharides, along with their polysaccharide counterparts, are crucial for energy storage, structural support, and cellular processes. Understanding the chemistry of glycosidic bonds, the types of disaccharides formed, and their metabolic pathways is key to grasping the intricate complexities of biological systems. Further research continues to unravel the subtle nuances of these fundamental molecules and their impact on life as we know it. The continued investigation into the properties and function of disaccharides will undoubtedly unveil further insights into their vital roles in the realm of biology and beyond. From the humble beginnings of two glucose molecules, the vast world of carbohydrate chemistry unfolds, providing a compelling example of the power of simplicity in shaping the complexity of life.
Latest Posts
Latest Posts
-
Are Ion Channels Active Or Passive
Apr 04, 2025
-
Prokaryotes That Obtain Their Energy From Chemical Compounds Are Called
Apr 04, 2025
-
What Does High Absorbance Mean In Spectrophotometry
Apr 04, 2025
-
Electric Field Due To A Disk Of Charge
Apr 04, 2025
-
Compare And Contrast Skeletal Smooth And Cardiac Muscle
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Two Glucose Molecules Joined Together Form . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
