What Are Characteristics Of Covalent Compounds
Muz Play
Apr 03, 2025 · 6 min read
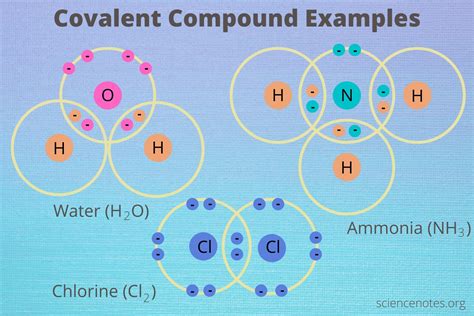
Table of Contents
What are the Characteristics of Covalent Compounds?
Covalent compounds, also known as molecular compounds, are formed when atoms share electrons to achieve a stable electron configuration, typically resembling a noble gas. This sharing creates a strong bond between the atoms, resulting in the formation of molecules. Unlike ionic compounds, which are formed through the electrostatic attraction between oppositely charged ions, covalent compounds exhibit a distinct set of characteristics that differentiate them. Understanding these characteristics is crucial in various fields, including chemistry, materials science, and biology.
Key Characteristics of Covalent Compounds
Covalent compounds display several key characteristics that set them apart from ionic and metallic compounds. These characteristics are a direct result of the nature of the covalent bond – the sharing of electrons between atoms.
1. Low Melting and Boiling Points:
One of the most noticeable characteristics of covalent compounds is their generally low melting and boiling points. This is because the intermolecular forces (forces between molecules) in covalent compounds are relatively weak compared to the strong electrostatic forces in ionic compounds. These weak intermolecular forces, such as van der Waals forces, hydrogen bonds, and dipole-dipole interactions, require less energy to overcome, leading to lower melting and boiling points. Many covalent compounds exist as gases or liquids at room temperature, while ionic compounds are typically solids.
2. Poor Electrical Conductivity:
Covalent compounds are generally poor conductors of electricity in both solid and liquid states. This is because, unlike ionic compounds which have freely moving ions, covalent compounds lack freely moving charged particles. The electrons are involved in strong covalent bonds, localized between atoms, and are not free to move and carry an electric current. However, some covalent compounds, particularly those that ionize in solution (acids, for example), can conduct electricity when dissolved in water.
3. Poor Thermal Conductivity:
Similar to their electrical conductivity, covalent compounds are also generally poor conductors of heat. This is again due to the absence of freely moving charged particles that can readily transfer thermal energy. The weak intermolecular forces hinder the efficient transfer of heat energy through the material.
4. Low Hardness and Brittleness:
Covalent compounds typically exhibit low hardness and are often brittle. This is in contrast to many ionic compounds which can be relatively hard. The relatively weak intermolecular forces in covalent compounds make them easily deformed under stress. The lack of strong electrostatic forces means there's less resistance to breakage under pressure.
5. Solubility:
The solubility of covalent compounds varies greatly depending on the polarity of the molecule and the solvent. Polar covalent compounds (compounds with a significant difference in electronegativity between the atoms) tend to be soluble in polar solvents like water. This is because the polar solvent molecules can interact with the dipole moment of the covalent molecule. Nonpolar covalent compounds, on the other hand, are typically soluble in nonpolar solvents like organic solvents (e.g., benzene, hexane). This principle is often summarized by the saying "like dissolves like."
6. Formation of Molecules:
Covalent compounds exist as discrete molecules. This means that the atoms are bonded together in specific, defined units. This molecular nature contrasts with ionic compounds, which exist as a continuous network of ions. The formation of discrete molecules significantly influences the physical properties of covalent compounds.
7. Variety of Physical States:
Covalent compounds can exist in all three physical states at room temperature: solid, liquid, and gas. The physical state depends on the strength of the intermolecular forces and the molecular weight of the compound. Smaller molecules with weaker intermolecular forces are more likely to be gases or liquids, while larger molecules with stronger intermolecular forces are more likely to be solids.
8. Reactivity:
The reactivity of covalent compounds varies greatly depending on the specific compound and the conditions. Some covalent compounds are very reactive, while others are relatively inert. Factors influencing reactivity include bond strength, bond polarity, and the presence of functional groups.
9. Isomerism:
Covalent compounds can exhibit isomerism. Isomers are molecules with the same molecular formula but different structural arrangements of atoms. This phenomenon is a direct consequence of the way atoms can share electrons in different spatial configurations. Isomerism leads to a vast diversity in the properties of compounds with the same chemical formula. There are several types of isomerism, including structural isomerism (different bonding arrangements) and stereoisomerism (different spatial arrangements of atoms).
10. Lower Density:
Generally, covalent compounds tend to have lower densities compared to ionic compounds. This is because the molecules are held together by weaker forces, resulting in less compact packing and greater intermolecular spacing.
Examples of Covalent Compounds and their Properties
To further illustrate the characteristics of covalent compounds, let's examine a few examples:
-
Water (H₂O): A polar covalent compound with a relatively high boiling point (100°C) due to strong hydrogen bonding. It's a liquid at room temperature and an excellent solvent for polar substances.
-
Methane (CH₄): A nonpolar covalent compound with a very low boiling point (-161.5°C). It's a gas at room temperature and soluble in nonpolar solvents.
-
Carbon dioxide (CO₂): A nonpolar covalent compound that is a gas at room temperature and poorly soluble in water.
-
Sugar (Sucrose, C₁₂H₂₂O₁₁): A polar covalent compound with a high melting point and soluble in water.
-
Diamond (C): A giant covalent structure with extremely high melting point, hardness, and is an electrical insulator.
-
Silicon dioxide (SiO₂): Another giant covalent network, forming the basis of sand and quartz, having high melting points and hardness.
The wide range of properties exhibited by these examples underscores the diversity within the class of covalent compounds.
Distinguishing Covalent from Ionic Compounds
It's important to be able to distinguish between covalent and ionic compounds based on their properties. While there's a spectrum of bonding types (polar covalent, nonpolar covalent, etc.), some general guidelines exist:
- Melting and Boiling Points: Covalent compounds generally have much lower melting and boiling points than ionic compounds.
- Electrical Conductivity: Covalent compounds are poor conductors of electricity, while ionic compounds are good conductors when molten or dissolved in water.
- Solubility: Polar covalent compounds dissolve in polar solvents, while nonpolar covalent compounds dissolve in nonpolar solvents. Ionic compounds often dissolve in polar solvents.
- Hardness: Covalent compounds are typically softer and more brittle than ionic compounds.
- Bonding: Ionic bonds involve the transfer of electrons, while covalent bonds involve the sharing of electrons.
Advanced Considerations: Polarity and Intermolecular Forces
The characteristics of covalent compounds are significantly influenced by the polarity of the molecule and the types of intermolecular forces present.
-
Polarity: If the atoms in a covalent bond have different electronegativities, the shared electrons are pulled closer to the more electronegative atom, creating a dipole moment. This polarity affects the compound's solubility and other properties.
-
Intermolecular Forces: The forces between molecules (intermolecular forces) include van der Waals forces (London dispersion forces, dipole-dipole interactions), and hydrogen bonds. Stronger intermolecular forces lead to higher melting and boiling points. Hydrogen bonds, a particularly strong type of dipole-dipole interaction, are crucial in determining the properties of many biological molecules like proteins and DNA.
Conclusion
Covalent compounds, formed through the sharing of electrons, exhibit a diverse range of properties stemming from the nature of the covalent bond and intermolecular forces. Their generally low melting and boiling points, poor electrical conductivity, and variable solubility make them distinct from ionic compounds. Understanding these characteristics is fundamental to comprehending the behavior of a vast array of substances in chemistry, biology, and materials science. Further exploration into the intricacies of polarity and intermolecular forces provides a deeper understanding of the rich diversity within the world of covalent compounds. This knowledge is invaluable for predicting the properties of new molecules and designing materials with specific desired characteristics.
Latest Posts
Latest Posts
-
Example Of Stoichiometry In Real Life
Apr 04, 2025
-
Dna Biology And Technology Dna And Rna Structure
Apr 04, 2025
-
Narcotics Act On The Central Nervous System By Producing A
Apr 04, 2025
-
Two Plants Heteroygous For Pod Color Are Crossed Refer To
Apr 04, 2025
-
What Two Main Products Result From Photosynthesis
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Are Characteristics Of Covalent Compounds . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
