What Are The Reactants Of A Neutralization Reaction
Muz Play
Apr 04, 2025 · 6 min read
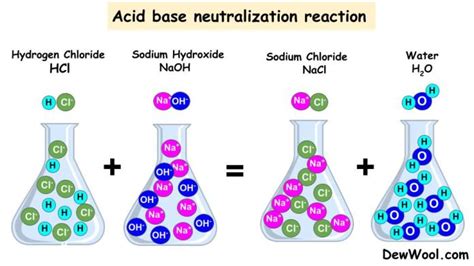
Table of Contents
What Are the Reactants of a Neutralization Reaction? A Deep Dive into Acid-Base Chemistry
Neutralization reactions are fundamental chemical processes that underpin many natural and industrial applications. Understanding the reactants involved is key to grasping their significance and predicting their outcomes. This comprehensive guide will delve into the intricacies of neutralization reactions, exploring the types of reactants, their properties, and the resulting products. We will also touch upon the applications of neutralization reactions in various fields.
Understanding Neutralization Reactions
A neutralization reaction is a chemical reaction in which an acid and a base react quantitatively with each other. The hallmark of this reaction is the formation of water and a salt. The reaction effectively neutralizes the acidic and basic properties of the reactants, resulting in a solution that is closer to neutral (pH 7). The exact pH of the resulting solution depends on the strength of the acid and base involved.
The Key Reactants: Acids and Bases
The core components of a neutralization reaction are, unsurprisingly, acids and bases. Let's examine each in detail:
Acids: Proton Donors
Acids are substances that donate protons (H⁺ ions) when dissolved in water. They exhibit several characteristic properties:
- Sour taste: This is a classic, though not recommended, way to identify an acid.
- Turn blue litmus paper red: This is a common laboratory test for acidity.
- React with metals to produce hydrogen gas: This reaction is often exothermic (releases heat).
- React with bases to form salts and water: This is the essence of neutralization reactions.
Acids are broadly classified into two categories based on their ability to donate protons:
-
Strong acids: These acids completely dissociate in water, meaning they donate all their protons. Examples include hydrochloric acid (HCl), sulfuric acid (H₂SO₄), and nitric acid (HNO₃). Strong acids have a significant impact on the pH of a solution, drastically lowering it.
-
Weak acids: These acids only partially dissociate in water, meaning they donate only some of their protons. Examples include acetic acid (CH₃COOH, found in vinegar), carbonic acid (H₂CO₃), and formic acid (HCOOH). Weak acids have a less pronounced effect on the pH of a solution.
Bases: Proton Acceptors
Bases are substances that accept protons (H⁺ ions) when dissolved in water. Like acids, they have distinctive properties:
- Bitter taste: Again, this is a characteristic property, but tasting chemicals is strongly discouraged.
- Turn red litmus paper blue: This is the opposite effect of acids on litmus paper.
- Feel slippery: Many bases feel soapy or slippery to the touch.
- React with acids to form salts and water: This is their role in neutralization reactions.
Bases are also categorized into strong and weak:
-
Strong bases: These bases completely dissociate in water, accepting all available protons. Examples include sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca(OH)₂). Strong bases significantly raise the pH of a solution.
-
Weak bases: These bases only partially dissociate in water, accepting only some protons. Examples include ammonia (NH₃) and many metal hydroxides. Weak bases have a less drastic effect on the pH of a solution.
Types of Neutralization Reactions
The specifics of a neutralization reaction depend on the strength of the acid and base involved. Here are some examples:
Strong Acid – Strong Base Neutralization
This type of reaction leads to the formation of a neutral salt and water. The pH of the resulting solution is approximately 7. For example:
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
In this reaction, hydrochloric acid (strong acid) reacts with sodium hydroxide (strong base) to produce sodium chloride (a neutral salt) and water.
Strong Acid – Weak Base Neutralization
The resulting solution will be slightly acidic (pH < 7). The weak base does not completely neutralize the strong acid. For instance:
HCl(aq) + NH₃(aq) → NH₄Cl(aq)
Here, hydrochloric acid neutralizes ammonia, a weak base, forming ammonium chloride. However, the solution remains slightly acidic due to the incomplete neutralization.
Weak Acid – Strong Base Neutralization
The resulting solution will be slightly alkaline (pH > 7). The weak acid does not completely neutralize the strong base. For example:
CH₃COOH(aq) + NaOH(aq) → CH₃COONa(aq) + H₂O(l)
Acetic acid, a weak acid, reacts with sodium hydroxide, a strong base, to form sodium acetate and water. The solution's pH will be greater than 7.
Weak Acid – Weak Base Neutralization
Predicting the pH of the resulting solution is more complex in this case, as it depends on the relative strengths of the acid and base. The solution might be slightly acidic, slightly alkaline, or approximately neutral, depending on the specific reactants.
The Products: Salts and Water
The products of a neutralization reaction are always a salt and water.
-
Salt: A salt is an ionic compound formed from the cation (positive ion) of the base and the anion (negative ion) of the acid. Salts can have various properties, ranging from neutral to acidic or basic, depending on the nature of the acid and base from which they are formed. For example, NaCl (sodium chloride) is a neutral salt, while NH₄Cl (ammonium chloride) is an acidic salt, and CH₃COONa (sodium acetate) is a basic salt.
-
Water: Water (H₂O) is formed by the combination of H⁺ ions from the acid and OH⁻ ions from the base.
Applications of Neutralization Reactions
Neutralization reactions have widespread applications across various fields:
-
Acid spills: Neutralization reactions are crucial for cleaning up acid spills. Bases like calcium carbonate (CaCO₃) or sodium bicarbonate (NaHCO₃) can be used to neutralize spilled acids, reducing their harmful effects.
-
Digestion: Our stomachs produce hydrochloric acid (HCl) to aid in digestion. When the stomach produces excess acid, antacids containing bases like magnesium hydroxide (Mg(OH)₂) or aluminum hydroxide (Al(OH)₃) can neutralize the excess acid, relieving heartburn or indigestion.
-
Wastewater treatment: Neutralization reactions are vital in wastewater treatment plants to adjust the pH of wastewater before discharge, making it environmentally safe.
-
Industrial processes: Many industrial processes involve acids and bases. Neutralization reactions are used to control pH levels in these processes, ensuring optimal reaction conditions.
-
Soil pH adjustment: The pH of soil can significantly influence plant growth. Neutralization reactions can be used to adjust the pH of acidic or alkaline soils to make them more suitable for specific crops.
-
Production of salts: Neutralization reactions are utilized in the industrial production of various salts. These salts have numerous applications in various sectors.
Conclusion
Neutralization reactions are fundamental chemical processes involving the reaction between acids and bases, resulting in the formation of a salt and water. Understanding the types of acids and bases, their strengths, and the resulting products is crucial for predicting the outcomes of these reactions and their diverse applications in various fields, from everyday life to industrial processes and environmental protection. The precise nature of the products and the resulting pH depends heavily on the strength of the reacting acid and base. This comprehensive understanding is essential for anyone working with chemistry, whether in a laboratory setting or in other applied fields.
Latest Posts
Latest Posts
-
Convert Rectangular Coordinates To Polar Coordinates
Apr 05, 2025
-
Single Double And Triple Covalent Bonds
Apr 05, 2025
-
Explain Why Each Function Is Continuous Or Discontinuous
Apr 05, 2025
-
Describe The Development Of The Modern Periodic Table
Apr 05, 2025
-
Which Is Most Likely A Physical Change
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Are The Reactants Of A Neutralization Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
