What Happens To Ionization Energy Down A Group
Muz Play
Apr 02, 2025 · 6 min read
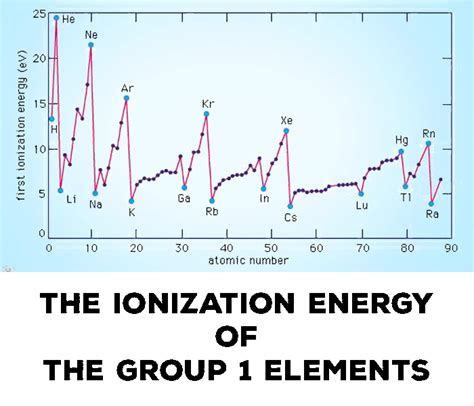
Table of Contents
What Happens to Ionization Energy Down a Group? A Deep Dive into Periodic Trends
Ionization energy, a fundamental concept in chemistry, describes the energy required to remove an electron from a gaseous atom or ion. Understanding how ionization energy changes down a group in the periodic table is crucial for predicting chemical behavior and reactivity. This article delves into the intricacies of this trend, exploring the underlying physics, exceptions, and its implications.
The General Trend: A Decrease Down a Group
The general trend observed for ionization energy down a group is a decrease. As you move down a group, the ionization energy generally becomes smaller. This means it takes less energy to remove an electron from an atom lower in the group compared to one higher up. This seemingly simple trend, however, is underpinned by several key factors:
1. Increasing Atomic Radius
The most significant factor contributing to the decrease in ionization energy down a group is the increase in atomic radius. As you descend a group, each successive element adds an electron shell. This results in a larger atom with electrons further from the nucleus. The increased distance weakens the electrostatic attraction between the positively charged nucleus and the outermost electrons. This weaker attraction makes it easier to remove an electron, hence the lower ionization energy.
Think of it like this: imagine trying to pull a magnet away from a metal surface. If the magnet is very close to the surface, it's difficult to separate. But if the magnet is further away, it's much easier. The nucleus is like the metal surface, and the outermost electron is like the magnet.
2. Increased Shielding Effect
Another crucial factor is the increased shielding effect. As you move down a group, the number of inner electrons (core electrons) increases. These core electrons effectively shield the outermost valence electrons from the full positive charge of the nucleus. This shielding reduces the net positive charge experienced by the valence electrons, weakening the electrostatic attraction and lowering the ionization energy.
The inner electrons act like a buffer, reducing the pull of the nucleus on the outermost electron. The more inner electrons there are, the stronger the shielding effect, and the easier it is to remove a valence electron.
3. Increased Number of Protons
While the atomic radius increases and shielding effect enhances, it's important to note that the number of protons in the nucleus also increases down a group. This increased positive charge should theoretically increase the attraction to the outermost electrons. However, the effects of the increased atomic radius and shielding are significantly more impactful, ultimately leading to the observed decrease in ionization energy.
The increase in protons does contribute to a stronger attraction, but it's outweighed by the increased distance and shielding, which lessen the overall effective nuclear charge experienced by the valence electrons.
Exceptions to the Rule: Why the Trend Isn't Always Straightforward
While the general trend is a decrease in ionization energy down a group, there are some exceptions and irregularities that require closer examination. These deviations often stem from subtle electronic configurations and inter-electronic repulsions.
1. The 'p-block' Irregularities:
In the p-block elements, some irregularities can be observed. For instance, the ionization energy of nitrogen is higher than that of oxygen, despite oxygen having a higher atomic number. This anomaly is due to the half-filled p-orbital in nitrogen. A half-filled p-orbital offers greater stability due to electron-electron repulsion being minimized in this configuration. Removing an electron from this stable configuration requires more energy, resulting in a higher ionization energy than expected.
Similarly, other p-block elements might show slight deviations from the general trend due to variations in electron-electron repulsion and orbital stability.
2. The 'd-block' and 'f-block' complexities:
The d-block and f-block elements exhibit more complex ionization energy trends due to the involvement of d and f orbitals. The poor shielding effect of d and f electrons and the complexities of electron-electron interactions make predicting ionization energies in these blocks less straightforward. Slight increases or decreases can be observed depending on the specific electronic configuration and the interplay of various factors.
Understanding these complexities requires a deeper understanding of atomic orbitals and electron configurations.
Implications of Ionization Energy Trends
The decreasing ionization energy trend down a group has significant implications for the chemical properties and reactivity of elements.
1. Reactivity of Alkali Metals:
The alkali metals (Group 1) exhibit extremely low ionization energies due to their single valence electron and the significant effects of atomic radius and shielding. This makes them highly reactive, readily losing their valence electron to form +1 ions and participating in various chemical reactions.
Their low ionization energy is a key factor in their propensity to form ionic compounds.
2. Reactivity of Halogens:
The halogens (Group 17) have relatively high ionization energies compared to the alkali metals, reflecting the strong attraction between the nucleus and their seven valence electrons. However, their high electron affinity makes them eager to gain an electron, achieving a stable octet. This makes them highly reactive, although in a different way than alkali metals – they are strong oxidizing agents, gaining electrons to form -1 ions.
3. Predicting Chemical Behavior:
The ionization energy trend allows chemists to predict the general chemical behavior of elements within a group. Elements with low ionization energies are more likely to form cations, while those with high ionization energies are more likely to form anions or participate in covalent bonding. This prediction power is invaluable in understanding chemical reactions and designing new materials.
4. Applications in Materials Science:
Understanding ionization energy trends is crucial in materials science. The ability to fine-tune the ionization energies of elements by alloying or doping materials allows researchers to create materials with specific electronic and optical properties. This has significant applications in semiconductor technology, photovoltaics, and catalysis.
Beyond First Ionization Energy: Higher Ionization Energies
It's important to note that the discussion above primarily focuses on the first ionization energy, which is the energy required to remove the first electron. Subsequent ionization energies (second, third, and so on) generally increase. This is because removing an electron leaves a positively charged ion, resulting in a stronger attraction for the remaining electrons. This makes it increasingly difficult to remove subsequent electrons.
The large jump in ionization energy when removing an inner shell electron (core electron) signifies the significant energy required to overcome the shielding effect and the strong nuclear attraction experienced by those tightly bound electrons.
Conclusion: A Trend with Nuances
The decrease in ionization energy down a group is a fundamental periodic trend with far-reaching consequences for understanding the chemical behavior and reactivity of elements. While the general trend is well-established, understanding the exceptions and complexities related to shielding effects, electron-electron repulsions, and the nuances of d- and f-block elements is crucial for a comprehensive grasp of this important concept. Ionization energy is not merely a theoretical concept; it is a cornerstone of our understanding of chemical bonding, reactivity, and material properties. Its importance extends to diverse fields, underscoring its significance in both fundamental and applied chemistry.
Latest Posts
Latest Posts
-
Red Blood Cells In Hypertonic Solution
Apr 03, 2025
-
The Echelon Form Of A Matrix Is Unique
Apr 03, 2025
-
How To Write An Equation For A Vertical Line
Apr 03, 2025
-
The Heterozygote Expresses Phenotype Of Both Homozygotes
Apr 03, 2025
-
Difference Between A Somatic Cell And A Gamete
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Happens To Ionization Energy Down A Group . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
