Red Blood Cells In Hypertonic Solution
Muz Play
Apr 03, 2025 · 6 min read
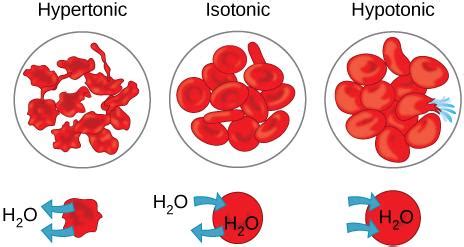
Table of Contents
Red Blood Cells in a Hypertonic Solution: A Comprehensive Overview
Red blood cells (RBCs), also known as erythrocytes, are the most abundant cells in the blood, playing a crucial role in oxygen transport throughout the body. Their behavior in different solutions, particularly hypertonic solutions, provides a fascinating case study in osmosis and cell membrane dynamics. Understanding this interaction is fundamental to grasping basic biological principles and has important implications in various medical fields. This article delves deep into the effects of hypertonic solutions on red blood cells, exploring the underlying mechanisms, observable changes, and the broader significance of this phenomenon.
Understanding Osmosis and Tonicity
Before examining the specifics of RBCs in hypertonic solutions, it's crucial to define osmosis and tonicity. Osmosis is the passive movement of water across a selectively permeable membrane from a region of high water concentration (low solute concentration) to a region of low water concentration (high solute concentration). This movement continues until equilibrium is reached, meaning the water concentration is equal on both sides of the membrane.
Tonicity refers to the relative concentration of solutes in two solutions separated by a selectively permeable membrane. There are three main types of tonicity:
- Isotonic: The solute concentration is equal inside and outside the cell. There is no net movement of water, and the cell maintains its normal shape and size.
- Hypotonic: The solute concentration is lower outside the cell than inside. Water moves into the cell, causing it to swell and potentially lyse (burst).
- Hypertonic: The solute concentration is higher outside the cell than inside. Water moves out of the cell, causing it to shrink and crenate.
Red Blood Cells in a Hypertonic Environment
When red blood cells are placed in a hypertonic solution, the concentration of solutes (e.g., salts, sugars) is higher outside the cell than inside. Due to osmosis, water molecules move across the cell membrane from the area of higher water concentration (inside the RBC) to the area of lower water concentration (the hypertonic solution). This outward movement of water causes the red blood cell to lose volume, leading to a process called crenation.
The Process of Crenation
Crenation is characterized by the cell shrinking and developing a spiky, wrinkled appearance. The extent of crenation depends on the concentration of the hypertonic solution and the duration of exposure. Mild hypertonicity might cause only slight shrinkage, while a highly concentrated solution can lead to significant cell deformation and potential damage.
Cellular Mechanisms Involved
The process of crenation is governed by the properties of the red blood cell membrane. This membrane is selectively permeable, allowing water to pass through but restricting the movement of larger solutes. Aquaporins, specialized water channels, facilitate the rapid movement of water across the membrane, contributing to the efficient osmotic response. The cell's cytoskeleton, a network of proteins beneath the membrane, plays a role in maintaining cell shape and integrity. However, in hypertonic conditions, the cytoskeleton's ability to maintain the normal biconcave shape is challenged by the significant loss of water.
Observable Changes
The changes in red blood cells subjected to hypertonic solutions are readily observable under a microscope. The transition from a normal, smooth, biconcave disc shape to a crenated, spiky form is dramatic and visually striking. The degree of crenation can be used to quantify the tonicity of the solution.
Factors Affecting Crenation
Several factors influence the degree of crenation observed in red blood cells exposed to hypertonic solutions:
- Concentration of the hypertonic solution: Higher solute concentrations lead to more pronounced water loss and greater crenation.
- Duration of exposure: Prolonged exposure to a hypertonic solution will result in increased water loss and more severe crenation.
- Temperature: Temperature affects the permeability of the cell membrane and can influence the rate of water movement.
- Type of solute: Different solutes may have different effects on the cell membrane, potentially influencing the extent of crenation. For instance, some solutes might interact with the membrane, affecting its permeability.
- Cell age: Older red blood cells may be more susceptible to damage from hypertonic conditions compared to younger cells due to changes in membrane fluidity and integrity.
Physiological Implications and Medical Relevance
The response of red blood cells to hypertonic solutions has several significant physiological and medical implications:
- Dehydration: Severe dehydration, where the body loses significant amounts of water, can create a hypertonic environment in the blood. This can lead to crenation of red blood cells and compromise their function in oxygen transport.
- Hypernatremia: This condition involves abnormally high levels of sodium in the blood, creating a hypertonic state. Hypernatremia can cause significant cellular dehydration, including crenation of red blood cells, and can have serious consequences for various organ systems.
- Diabetes Mellitus: In poorly controlled diabetes, high blood glucose levels create a hypertonic environment. This can lead to cellular dehydration and affect various cell types, including red blood cells.
- Intravenous fluid therapy: Careful attention must be given to the tonicity of intravenous fluids administered to patients. The use of hypertonic solutions can have serious consequences if not properly managed. Isotonic solutions are generally preferred to avoid causing crenation or lysis of blood cells.
- Diagnostic tools: Observing the response of red blood cells to solutions of varying tonicity can be used as a diagnostic tool to assess the health and integrity of the cell membrane.
Experimental Investigation of Crenation
The effects of hypertonic solutions on red blood cells are commonly demonstrated in laboratory settings. These experiments often involve:
- Preparing a blood smear: A drop of blood is spread thinly on a microscope slide.
- Adding a hypertonic solution: A hypertonic solution (e.g., a concentrated saline solution) is added to the blood smear.
- Observing under a microscope: The changes in the shape of the red blood cells are observed using a light microscope.
Conclusion
The behavior of red blood cells in hypertonic solutions offers a valuable illustration of fundamental principles in biology, particularly osmosis and cell membrane function. Understanding the process of crenation is critical for comprehending the consequences of various physiological conditions and for making informed decisions in medical settings. From the microscopic level of cellular changes to the macroscopic effects on overall health, the impact of hypertonicity on red blood cells highlights the intricate interplay between cellular processes and physiological homeostasis. The continued study of this interaction contributes to advancements in medical diagnostics, treatments, and our overall understanding of cellular biology.
Further Research and Exploration
This topic extends beyond the basics. Deeper exploration could include researching specific proteins involved in the red blood cell membrane's response to hypertonic stress, investigating the role of different ions besides sodium, and exploring advanced techniques like flow cytometry to quantitatively analyze crenation. Further research into the long-term effects of chronic hypertonicity on RBC function and the potential development of compensatory mechanisms would also be significant contributions to the field. The intricacies of this seemingly simple biological process continue to present opportunities for groundbreaking discoveries.
Latest Posts
Latest Posts
-
How Many Lobes Does The Frogs Liver Have
Apr 04, 2025
-
Gibbs Free Energy Vs Temperature Graph
Apr 04, 2025
-
Practice Problems For Newtons Second Law Of Motion
Apr 04, 2025
-
Atom Molecule Cell Tissue Organ Organ System Organism
Apr 04, 2025
-
Where On The Periodic Table Are The Metals Found
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Red Blood Cells In Hypertonic Solution . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
