Where On The Periodic Table Are The Metals Found
Muz Play
Apr 04, 2025 · 6 min read
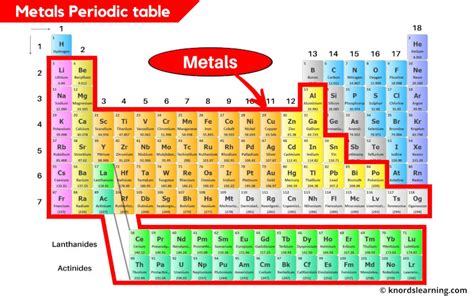
Table of Contents
Where on the Periodic Table are the Metals Found? A Comprehensive Guide
The periodic table, that iconic chart adorning countless science classrooms, is more than just a neatly organized list of elements. It's a powerful tool that reveals fundamental relationships between the building blocks of matter. One of the most striking patterns it reveals is the distribution of metals. Understanding where metals are located on the periodic table is key to comprehending their properties and applications. This comprehensive guide delves deep into the subject, exploring the location, characteristics, and exceptions within the metallic landscape of the periodic table.
The Broad Sweep: Metals Dominate the Left
The most straightforward answer to the question "Where are metals found on the periodic table?" is: predominantly on the left and in the center. A vast majority of the elements residing on the left side of the zig-zag line separating metals from nonmetals are metals. This line, often called the metalloid staircase, isn't a perfectly straight line; its slight undulations reflect the gradual transition in properties between metals and nonmetals.
This left-hand dominance isn't accidental. The arrangement of the periodic table reflects the electronic configuration of atoms. Metals generally have relatively few electrons in their outermost shells (valence electrons). This electronic structure facilitates the loss of electrons, a characteristic feature leading to the formation of positive ions (cations) and contributing to many of their metallic properties.
Groups and Their Metallic Traits:
Let's break down the metallic regions by group (vertical column) on the periodic table:
-
Group 1 (Alkali Metals): Highly reactive, soft, silvery-white metals. Their reactivity increases down the group. Examples include lithium (Li), sodium (Na), and potassium (K). These are all quintessential metals exhibiting classic metallic properties.
-
Group 2 (Alkaline Earth Metals): Also reactive, but less so than alkali metals. They are generally harder and denser than alkali metals. Examples include beryllium (Be), magnesium (Mg), and calcium (Ca). Again, these are strongly metallic.
-
Groups 3-12 (Transition Metals): This is the heart of the metallic region. These elements are known for their variable oxidation states, meaning they can form ions with different charges. This leads to a wide range of compounds and complex behaviors. This group encompasses elements crucial for industry and technology, including iron (Fe), copper (Cu), gold (Au), and many more. Their metallic properties are generally very strong.
-
Lanthanides and Actinides: Often placed separately at the bottom of the table, these series of elements (f-block elements) are all metals. The lanthanides are relatively reactive, while the actinides are radioactive and extremely reactive. Their metallic characteristics are consistent with their placement in the overall table's structure.
The Gray Area: Metalloids – Bridging the Gap
The metalloid staircase mentioned earlier isn't just a visual divider; it represents a gradual transition in properties. Elements along this line, known as metalloids or semimetals, exhibit properties that are intermediate between those of metals and nonmetals. This means they sometimes behave like metals and sometimes like nonmetals, depending on the conditions.
Metalloids like silicon (Si), germanium (Ge), and arsenic (As) possess a combination of metallic and nonmetallic traits. They are semiconductors, meaning their electrical conductivity lies somewhere between that of conductors (metals) and insulators (nonmetals). This property makes them incredibly important in electronics. The presence of metalloids illustrates the continuous spectrum of properties within the periodic table.
Exceptions and Nuances: Not All Shine Equally
While the vast majority of elements on the left and center of the periodic table are metals, there are some exceptions and nuances to consider:
-
Hydrogen (H): While placed above Group 1, hydrogen behaves quite differently. Under standard conditions, it's a nonmetal gas. However, under extremely high pressures, it can exhibit metallic properties. This highlights that an element's behavior can be highly pressure-dependent.
-
Some Post-Transition Metals: Elements like aluminum (Al), tin (Sn), and lead (Pb) are located to the right of the transition metals. While they are undoubtedly metals, some of their properties exhibit a lesser degree of metallic character compared to those in Groups 1 and 2 or even within the transition metal block. This is reflected in their lower melting points or greater reactivity with certain substances.
-
Metallic Character Across a Period: Moving from left to right across a period (horizontal row), the metallic character generally decreases. This is due to the increasing number of protons in the nucleus and the accompanying tighter attraction to the valence electrons. This trend is clearly observed in the transition from highly reactive alkali metals to nonmetals on the right side of the periodic table.
The Significance of Metallic Properties
The location of metals on the periodic table is intrinsically linked to their characteristic properties:
-
Electrical Conductivity: Metals are excellent conductors of electricity due to the ease with which electrons can move through their structure. This is why copper is widely used in electrical wiring.
-
Thermal Conductivity: Metals efficiently transfer heat. This is why many cooking utensils are made of metal.
-
Malleability and Ductility: Metals can be hammered into thin sheets (malleability) and drawn into wires (ductility). This is a direct consequence of the way their atoms are bonded together, allowing for easy reshaping without breaking.
-
Luster: Many metals have a characteristic shiny appearance, a property known as luster. This is due to their interaction with light.
-
Density: Metals typically have high densities; they contain a large number of atoms packed closely together.
Applications and Technological Advancements
The unique properties of metals have led to their widespread use in countless applications:
-
Construction: Steel (an alloy of iron and carbon) and aluminum are essential building materials.
-
Transportation: Cars, trains, and airplanes rely heavily on metals for their structural components.
-
Electronics: Copper, gold, and other metals are critical in the manufacture of electronic devices.
-
Medicine: Many medical implants and instruments are made from biocompatible metals like titanium.
-
Energy: Metals play a vital role in energy generation and storage, particularly in batteries and fuel cells.
Conclusion: A Periodic Pattern of Metallic Mastery
The periodic table serves as a powerful roadmap for understanding the distribution and properties of elements. The preponderance of metals on the left and center of the table underscores their significance in the natural world and in human technology. While exceptions and nuances exist, the overall pattern reveals the deep connection between electronic configuration, atomic structure, and the observable properties of these essential materials. Further exploration into specific metal groups and their unique characteristics will undoubtedly reveal even more fascinating insights into the world of chemistry and materials science. Understanding where metals are found on the periodic table is not just a matter of memorization; it's the key to unlocking their amazing potential and their pervasive role in our daily lives.
Latest Posts
Latest Posts
-
Pretest And Posttest Control Group Design
Apr 04, 2025
-
Max Rate Of Change Directional Derivative
Apr 04, 2025
-
The Energy Of Random Atomic And Molecular Motion Is Called
Apr 04, 2025
-
Is Salt A Compound Or Mixture
Apr 04, 2025
-
What State Of Matter Is Compressible
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Where On The Periodic Table Are The Metals Found . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
