What Is An Abbreviated Electron Configuration
Muz Play
Apr 02, 2025 · 6 min read
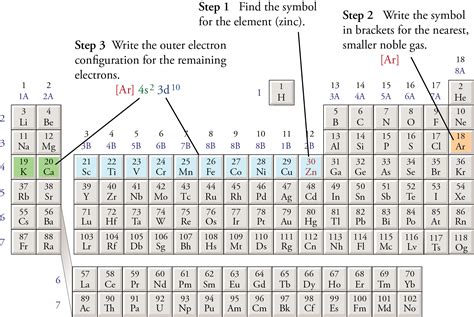
Table of Contents
What is an Abbreviated Electron Configuration? A Comprehensive Guide
Electron configuration describes the arrangement of electrons in an atom's orbitals. Understanding this arrangement is crucial for predicting an element's chemical properties and reactivity. While the full electron configuration provides a complete picture, the abbreviated electron configuration offers a more concise and efficient way to represent the electron arrangement, particularly for elements with high atomic numbers. This article will delve into the intricacies of abbreviated electron configurations, exploring its significance, how to write it, and its applications in chemistry.
Understanding Electron Configuration: The Foundation
Before diving into abbreviated electron configurations, let's establish a firm understanding of the basic principles of electron configuration. Electrons occupy specific energy levels, or shells, surrounding the nucleus. Each shell is further divided into subshells, designated as s, p, d, and f. These subshells can hold a specific number of electrons:
- s subshell: Holds a maximum of 2 electrons.
- p subshell: Holds a maximum of 6 electrons.
- d subshell: Holds a maximum of 10 electrons.
- f subshell: Holds a maximum of 14 electrons.
The filling of electrons follows the Aufbau principle, which states that electrons fill the lowest energy levels first. This is often represented by the Aufbau diagram or by using the principle quantum number (n) and the azimuthal quantum number (l). Furthermore, Hund's rule dictates that electrons will individually occupy each orbital within a subshell before pairing up. The Pauli exclusion principle ensures that no two electrons within an atom can have the same set of four quantum numbers (n, l, ml, and ms).
Example: Full Electron Configuration of Sodium (Na)
Sodium (Na) has an atomic number of 11, meaning it has 11 electrons. Its full electron configuration is: 1s²2s²2p⁶3s¹. This indicates that:
- Two electrons occupy the 1s orbital.
- Two electrons occupy the 2s orbital.
- Six electrons occupy the 2p orbitals (three 2p orbitals, each with two electrons).
- One electron occupies the 3s orbital.
The Power of Abbreviated Electron Configuration: Efficiency and Clarity
Writing out the full electron configuration for elements with many electrons can be lengthy and cumbersome. This is where the abbreviated electron configuration comes into play. It simplifies the representation by using the noble gas notation. Noble gases are elements in Group 18 of the periodic table, characterized by their extremely stable electron configurations (full outer electron shells).
The abbreviated electron configuration replaces the core electrons (electrons in the inner shells) with the symbol of the preceding noble gas in brackets. The remaining valence electrons (electrons in the outermost shell) are then written explicitly.
How to Write an Abbreviated Electron Configuration
The process involves these steps:
-
Identify the noble gas that precedes the element: Look at the periodic table and find the noble gas element that comes before the element you are working with. For example, for sodium (Na), the preceding noble gas is neon (Ne).
-
Determine the electron configuration of the noble gas: The electron configuration of neon (Ne) is 1s²2s²2p⁶.
-
Replace the core electrons with the noble gas symbol in brackets: In sodium's case, this means replacing 1s²2s²2p⁶ with [Ne].
-
Write the remaining valence electrons: The remaining valence electron in sodium is in the 3s orbital, so we add 3s¹.
Therefore, the abbreviated electron configuration of sodium (Na) is [Ne]3s¹. This concise notation effectively communicates the electron arrangement without sacrificing essential information.
Examples of Abbreviated Electron Configurations
Let's illustrate with more examples:
-
Chlorine (Cl, atomic number 17): The full electron configuration is 1s²2s²2p⁶3s²3p⁵. The abbreviated electron configuration is [Ne]3s²3p⁵. (Neon is the preceding noble gas.)
-
Iron (Fe, atomic number 26): The full electron configuration is 1s²2s²2p⁶3s²3p⁶4s²3d⁶. The abbreviated electron configuration is [Ar]4s²3d⁶. (Argon is the preceding noble gas.)
-
Gold (Au, atomic number 79): The full electron configuration is [Xe] 6s¹ 4f¹⁴ 5d¹⁰. The abbreviated electron configuration is [Xe] 6s¹ 4f¹⁴ 5d¹⁰. (Xenon is the preceding noble gas). Note the order of filling of 6s and 4f orbitals.
-
Lead (Pb, atomic number 82): The full electron configuration is [Xe] 6s² 4f¹⁴ 5d¹⁰ 6p². The abbreviated electron configuration is [Xe] 6s² 4f¹⁴ 5d¹⁰ 6p². (Xenon is the preceding noble gas).
These examples highlight how the abbreviated electron configuration drastically reduces the length and complexity of representing the electron arrangement, especially for heavier elements.
Significance of Abbreviated Electron Configuration in Chemistry
Abbreviated electron configurations play a crucial role in various aspects of chemistry:
-
Predicting Chemical Properties: The valence electrons, explicitly shown in the abbreviated configuration, directly determine an element's chemical behavior and reactivity. Elements with similar valence electron configurations often exhibit similar chemical properties. This is the basis for the organization of the periodic table.
-
Understanding Bonding: The number and arrangement of valence electrons dictate how atoms will form bonds with other atoms. For instance, the presence of one valence electron in sodium ([Ne]3s¹) explains its tendency to lose this electron to form a +1 ion, while chlorine's seven valence electrons ([Ne]3s²3p⁵) explain its tendency to gain one electron to form a -1 ion. The ionic bond between sodium and chlorine arises from this electron transfer.
-
Explaining Periodic Trends: Abbreviated electron configurations help explain periodic trends such as atomic radius, ionization energy, and electronegativity. For example, the increasing nuclear charge across a period, while keeping the outermost shell relatively similar, leads to a decrease in atomic radius.
-
Spectroscopy: Understanding the electron configuration is essential to interpreting atomic spectra. Transitions between energy levels (orbitals) result in the absorption or emission of light at specific wavelengths. Abbreviated configurations provide a simplified view of the electronic transitions.
-
Coordination Chemistry: In coordination chemistry, which involves transition metal complexes, the abbreviated electron configuration of the central metal ion is crucial in determining the geometry, magnetic properties, and reactivity of the complex. The d-orbital configuration is particularly important in this context.
Exceptions to the Aufbau Principle and Abbreviated Configuration
It's important to note that there are exceptions to the Aufbau principle. Some elements exhibit slight variations in their electron configurations due to factors such as electron-electron repulsion and orbital stability. For instance, chromium (Cr) and copper (Cu) have slightly different electron configurations than what would be predicted by strictly following the Aufbau principle. Their abbreviated configurations are [Ar]4s¹3d⁵ and [Ar]4s¹3d¹⁰, respectively, instead of the expected [Ar]4s²3d⁴ and [Ar]4s²3d⁹. These exceptions are related to the stability achieved by having half-filled or completely filled d subshells.
Despite these exceptions, the abbreviated electron configuration remains a highly useful tool for predicting and understanding the chemical behavior of most elements. The exceptions are relatively few and are often discussed in advanced chemistry courses.
Conclusion: A Powerful Tool for Chemists
The abbreviated electron configuration is a powerful tool that simplifies the representation of electron arrangements in atoms. Its concise notation makes it easier to understand and predict the chemical properties and reactivity of elements, especially those with high atomic numbers. While the full electron configuration provides a detailed picture, the abbreviated version offers an efficient and clear way to highlight the crucial valence electrons that dictate chemical behavior. Understanding both full and abbreviated electron configurations is essential for a comprehensive understanding of atomic structure and chemical bonding. The use of the abbreviated electron configuration, coupled with the understanding of the underlying principles, forms a fundamental basis for advanced topics in chemistry and related fields.
Latest Posts
Latest Posts
-
Finding Domain Of A Composite Function
Apr 03, 2025
-
Image Formation In A Plane Mirror
Apr 03, 2025
-
Us Air Force Tongue And Quill
Apr 03, 2025
-
The Coefficients In A Chemical Equation Represent The
Apr 03, 2025
-
How To Calculate The Gradient Of A Stream
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is An Abbreviated Electron Configuration . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
