What Is Group 2 In The Periodic Table
Muz Play
Apr 04, 2025 · 6 min read
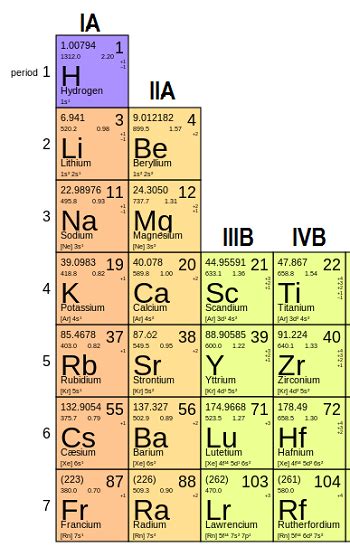
Table of Contents
What is Group 2 in the Periodic Table? Exploring the Alkaline Earth Metals
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Within this organized arrangement, groups represent vertical columns of elements sharing similar characteristics. Group 2, also known as alkaline earth metals, is a fascinating collection of elements exhibiting unique properties and playing crucial roles in various applications. This article delves deep into the world of Group 2, exploring their physical and chemical properties, common reactions, applications, and biological significance.
Understanding the Alkaline Earth Metals: Properties and Trends
Group 2 comprises six elements: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). These elements are all metals, characterized by their metallic luster, good electrical conductivity, and relatively high melting and boiling points compared to other groups. However, there's a clear trend in properties as you move down the group:
Atomic Radius and Ionization Energy
As you proceed down Group 2, the atomic radius increases. This is because each subsequent element adds an extra electron shell, resulting in a larger atomic size. Concurrently, the ionization energy – the energy required to remove an electron – decreases. This is due to the increasing distance between the outermost electrons and the nucleus; the weaker attraction makes it easier to remove an electron.
Electronegativity and Reactivity
Electronegativity, the ability of an atom to attract electrons in a chemical bond, is relatively low for alkaline earth metals. This low electronegativity contributes to their high reactivity, particularly with nonmetals like oxygen and halogens. As you descend Group 2, reactivity generally increases because the outermost electrons are more easily lost.
Density and Melting/Boiling Points
The density of alkaline earth metals increases as you move down the group, mirroring the trend in atomic radius. While the melting and boiling points are generally high compared to other groups, they show a less consistent trend down the group, influenced by the complexities of metallic bonding.
Chemical Reactions of Group 2 Elements
The alkaline earth metals are highly reactive, particularly with nonmetals. Their chemical reactions are largely dominated by their tendency to lose two electrons, forming 2+ ions (M²⁺). Let's examine some common reactions:
Reaction with Oxygen
All alkaline earth metals react with oxygen to form metal oxides (MO). For example, magnesium burns brightly in air, producing magnesium oxide:
2Mg(s) + O₂(g) → 2MgO(s)
The reactivity with oxygen increases down the group. Beryllium reacts slowly, while calcium, strontium, and barium react more readily.
Reaction with Water
Beryllium does not react with water or steam. Magnesium reacts slowly with boiling water but more readily with steam. Calcium, strontium, and barium react vigorously with cold water, producing metal hydroxides and hydrogen gas:
Ca(s) + 2H₂O(l) → Ca(OH)₂(aq) + H₂(g)
The vigorousness of the reaction increases as you go down the group, reflecting the increasing reactivity.
Reaction with Acids
Alkaline earth metals react readily with dilute acids, producing hydrogen gas and metal salts. The reaction with hydrochloric acid (HCl) can be represented as:
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
Again, the reactivity increases down the group.
Reaction with Halogens
Group 2 metals react with halogens (fluorine, chlorine, bromine, iodine) to form metal halides (MX₂). For example, magnesium reacts with chlorine to form magnesium chloride:
Mg(s) + Cl₂(g) → MgCl₂(s)
The reactions are generally exothermic (release heat).
Applications of Alkaline Earth Metals and Their Compounds
The unique properties of alkaline earth metals and their compounds have led to widespread applications in various industries:
Magnesium (Mg)
- Lightweight Alloys: Magnesium's low density makes it ideal for creating lightweight alloys used in automobiles, aircraft, and other transportation applications.
- Pharmaceuticals: Magnesium compounds are used in various medications, including antacids and laxatives.
- Photography: Magnesium is used in flash photography due to its bright and intense flame when it burns.
Calcium (Ca)
- Building Materials: Calcium carbonate (CaCO₃), commonly known as limestone, is a crucial component in cement and concrete production. Gypsum (calcium sulfate) is also widely used in construction.
- Agriculture: Calcium is an essential nutrient for plant growth and is often added to fertilizers.
- Medicine: Calcium salts are used in the treatment of various medical conditions, including hypocalcemia (low blood calcium levels).
Beryllium (Be)
- Aerospace: Beryllium alloys are used in high-performance aircraft and spacecraft due to their high strength-to-weight ratio and excellent thermal conductivity.
- Nuclear Reactors: Beryllium is used as a neutron reflector and moderator in nuclear reactors.
- X-ray Windows: Due to its low atomic mass, beryllium is used in X-ray windows because it allows X-rays to pass through with minimal absorption.
Strontium (Sr), Barium (Ba), and Radium (Ra)
- Pyrotechnics: Strontium salts produce brilliant red flames in fireworks, while barium salts produce green flames.
- Medical Imaging: Barium sulfate is used as a contrast agent in X-ray imaging of the digestive system.
- Radium (Ra): While historically used in luminous paints, radium is now primarily studied for its radioactive properties in scientific research and has limited practical applications due to its high radioactivity.
Biological Significance of Alkaline Earth Metals
Alkaline earth metals play essential roles in biological systems.
Calcium (Ca)
Calcium is crucial for numerous biological processes:
- Bone and Teeth Formation: Calcium is the major structural component of bones and teeth, providing strength and rigidity.
- Muscle Contraction: Calcium ions are essential for muscle contraction and relaxation.
- Nerve Impulse Transmission: Calcium ions play a vital role in nerve impulse transmission.
- Blood Clotting: Calcium is necessary for the blood clotting process.
Magnesium (Mg)
Magnesium is another vital element in biological systems:
- Enzyme Activation: Magnesium acts as a cofactor for many enzymes, enabling them to catalyze biochemical reactions.
- Protein Synthesis: Magnesium is involved in protein synthesis and other metabolic processes.
- DNA Replication: Magnesium plays a role in DNA replication and repair.
Other Group 2 elements have limited or no known biological roles, with the exception of some trace amounts possibly contributing to specific functions in some organisms.
Environmental Considerations
Mining and processing of alkaline earth metals can have environmental impacts. Mining activities can lead to habitat destruction and soil erosion. The disposal of waste materials from industrial processes must be carefully managed to avoid environmental contamination.
Conclusion: The Importance of Group 2
Group 2 elements, the alkaline earth metals, are a unique and vital group with properties and applications spanning many facets of our modern world. From lightweight alloys in transportation to essential roles in biological systems, their importance cannot be overstated. Understanding their chemical behavior, applications, and environmental implications is crucial for sustainable progress in various scientific and industrial fields. Further research and development continually unlock new applications and deepen our understanding of these fascinating elements. The consistent trends in their properties as you move down the group, combined with their significant applications, makes Group 2 a fascinating subject worthy of continuous study and exploration.
Latest Posts
Latest Posts
-
When Does Dna Replication Occur In A Eukaryotic Cell
Apr 04, 2025
-
What Was Shown By Both Redis And Pasteurs Experiments
Apr 04, 2025
-
The Elements In Group 1 Are Called The
Apr 04, 2025
-
Proof Of The Inverse Function Theorem
Apr 04, 2025
-
Nonmetal Halogen Family Atomic Mass 35
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is Group 2 In The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
