What Is Produced When An Acid Reacts With A Base
Muz Play
Apr 03, 2025 · 6 min read
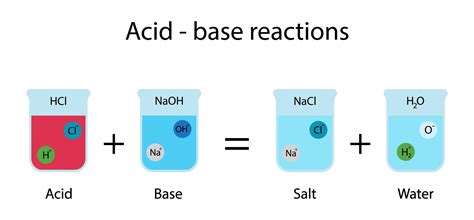
Table of Contents
What is Produced When an Acid Reacts with a Base?
The reaction between an acid and a base is a fundamental concept in chemistry, known as neutralization. Understanding what's produced in this reaction is crucial for various applications, from everyday life to industrial processes. This comprehensive guide delves deep into the products of acid-base reactions, exploring different types of acids and bases, the nuances of the reaction, and the practical implications of neutralization.
Understanding Acids and Bases
Before diving into the products, let's refresh our understanding of acids and bases. Several definitions exist, but the most commonly used are the Arrhenius, Brønsted-Lowry, and Lewis definitions.
Arrhenius Definition
The Arrhenius definition, one of the earliest, defines acids as substances that produce hydrogen ions (H⁺) when dissolved in water, and bases as substances that produce hydroxide ions (OH⁻) when dissolved in water. This definition is simple but limited, as it only applies to aqueous solutions.
Brønsted-Lowry Definition
The Brønsted-Lowry definition expands upon the Arrhenius definition. It defines acids as proton (H⁺) donors and bases as proton acceptors. This definition is broader and applies to reactions not necessarily involving water.
Lewis Definition
The Lewis definition, the broadest of the three, defines acids as electron-pair acceptors and bases as electron-pair donors. This definition encompasses a wider range of reactions, including those that don't involve protons.
The Neutralization Reaction: The Core Process
The core of an acid-base reaction is the neutralization reaction, where an acid and a base react to form water and a salt. The general equation is:
Acid + Base → Salt + Water
This seemingly simple equation hides a wealth of chemical complexity. The nature of the salt formed depends entirely on the specific acid and base involved. Let's explore this in more detail.
Types of Salts Formed During Neutralization
The salt produced in a neutralization reaction isn't always table salt (sodium chloride). The type of salt formed depends on the cation (positive ion) from the base and the anion (negative ion) from the acid.
Salts from Strong Acids and Strong Bases
When a strong acid (e.g., hydrochloric acid, HCl; sulfuric acid, H₂SO₄; nitric acid, HNO₃) reacts with a strong base (e.g., sodium hydroxide, NaOH; potassium hydroxide, KOH; calcium hydroxide, Ca(OH)₂), the resulting salt is neutral. This means the resulting solution has a pH of approximately 7. For example:
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
Here, sodium chloride (NaCl) is the salt, and water (H₂O) is formed. The solution is neutral because neither the sodium cation nor the chloride anion significantly affects the pH of the water.
Salts from Strong Acids and Weak Bases
When a strong acid reacts with a weak base (e.g., ammonia, NH₃), the resulting salt is acidic. This is because the cation of the weak base can act as a weak acid, slightly increasing the concentration of H⁺ ions in the solution, lowering the pH below 7. For example:
HCl(aq) + NH₃(aq) → NH₄Cl(aq)
Ammonium chloride (NH₄Cl) is acidic because the ammonium ion (NH₄⁺) can donate a proton to water.
Salts from Weak Acids and Strong Bases
When a weak acid (e.g., acetic acid, CH₃COOH; carbonic acid, H₂CO₃) reacts with a strong base, the resulting salt is basic. This occurs because the anion of the weak acid can act as a weak base, increasing the concentration of OH⁻ ions in the solution, raising the pH above 7. For example:
CH₃COOH(aq) + NaOH(aq) → CH₃COONa(aq) + H₂O(l)
Sodium acetate (CH₃COONa) is basic because the acetate ion (CH₃COO⁻) can accept a proton from water.
Salts from Weak Acids and Weak Bases
The pH of the salt formed from the reaction of a weak acid and a weak base is more complex to predict. It depends on the relative strengths of the conjugate acid and conjugate base formed. In some cases, the resulting solution might be acidic, basic, or even neutral, depending on the specific acid and base involved.
Beyond Simple Neutralization: Understanding the Equilibrium
Acid-base reactions often don't go to completion; they reach an equilibrium. This means that at a certain point, the rate of the forward reaction (acid + base → salt + water) equals the rate of the reverse reaction (salt + water → acid + base). The position of this equilibrium depends on the strengths of the acid and base involved. Strong acids and strong bases tend to have equilibria that strongly favor the products (salt and water), while weak acids and weak bases have equilibria that are less strongly shifted toward the products.
Practical Applications of Neutralization Reactions
Neutralization reactions are crucial in numerous applications:
-
Digestion: Our stomachs produce hydrochloric acid (HCl) to digest food. When the stomach produces too much acid, antacids (bases) are used to neutralize the excess acid, relieving heartburn.
-
Wastewater Treatment: Industrial wastewater often contains acids or bases. Neutralization is a critical step in treating this wastewater before it's released into the environment.
-
Soil pH Adjustment: The pH of soil significantly impacts plant growth. Farmers use neutralization reactions to adjust the soil pH, making it suitable for specific crops.
-
Chemical Synthesis: Many chemical processes rely on controlled acid-base reactions to synthesize specific compounds.
-
Titration: Neutralization reactions are fundamental to titrations, a quantitative analytical technique used to determine the concentration of an unknown acid or base.
Factors Affecting Neutralization Reaction Rate
Several factors influence the rate of a neutralization reaction:
-
Concentration: Higher concentrations of acid and base lead to faster reactions.
-
Temperature: Increasing temperature generally increases the reaction rate.
-
Nature of Acid and Base: Strong acids and strong bases react faster than weak acids and weak bases.
-
Presence of Catalysts: Some catalysts can accelerate the rate of neutralization.
Further Considerations: Polyprotic Acids and Bases
The discussion so far has primarily focused on monoprotic acids (acids that donate one proton) and monobasic bases (bases that accept one proton). However, many acids and bases are polyprotic (donate more than one proton) or polybasic (accept more than one proton). These reactions are more complex but still follow the fundamental principle of neutralization: the reaction proceeds until all available protons are transferred. For example, sulfuric acid (H₂SO₄) is a diprotic acid, meaning it can donate two protons in successive steps.
Conclusion: The Ubiquity of Neutralization
The reaction between an acid and a base, resulting in the formation of water and a salt, is a cornerstone of chemistry. Understanding the nuances of this reaction, including the types of salts formed and the factors influencing the reaction rate, is vital for appreciating its widespread applications in various fields, from everyday life to advanced chemical synthesis. The seemingly simple equation, Acid + Base → Salt + Water, masks a rich tapestry of chemical processes that are fundamental to our understanding of the world around us. The versatility of this reaction and its significance in various scientific and technological applications highlight its enduring importance in the realm of chemistry. Furthermore, continued research into the intricacies of acid-base reactions will undoubtedly lead to further advancements and applications in the future.
Latest Posts
Latest Posts
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
-
What Is The Net Change In Math
Apr 04, 2025
-
Why Cant The Subscripts Be Changed In A Chemical Equation
Apr 04, 2025
-
How Many Bonds Does Boron Form
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is Produced When An Acid Reacts With A Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
