What Is The Dielectric Constant Of Water
Muz Play
Apr 03, 2025 · 6 min read
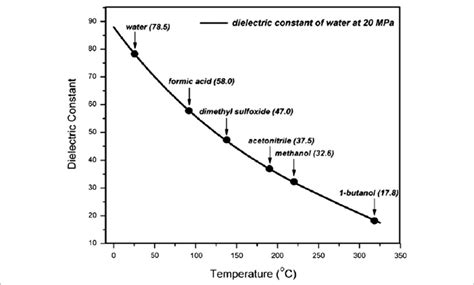
Table of Contents
What is the Dielectric Constant of Water? A Deep Dive
The dielectric constant of water, a seemingly simple value, plays a crucial role in numerous scientific fields and everyday phenomena. Understanding its properties and implications is key to comprehending various processes, from the behavior of biological molecules to the performance of electronic devices. This article delves deep into the dielectric constant of water, exploring its definition, measurement, influencing factors, and significance across diverse applications.
Defining the Dielectric Constant
The dielectric constant, also known as relative permittivity (ε<sub>r</sub>), is a measure of a material's ability to reduce the electric field strength between two charges. It represents the ratio of the electric field strength in a vacuum to the electric field strength in the material. In simpler terms, it describes how effectively a substance can store electrical energy within an electric field. A higher dielectric constant indicates a greater ability to reduce the electric field and store more energy.
For water, the dielectric constant is remarkably high compared to most other substances. This high value is a consequence of water's unique molecular structure and strong dipole moment.
The Role of Water's Polarity
Water (H₂O) is a polar molecule, meaning it possesses a net dipole moment due to the uneven distribution of electron density. The oxygen atom is more electronegative than the hydrogen atoms, attracting electrons more strongly. This creates a partial negative charge (δ-) on the oxygen and partial positive charges (δ+) on the hydrogens. This polarity allows water molecules to effectively align themselves in an electric field, reducing its overall strength. This alignment counteracts the electric field, resulting in the high dielectric constant.
Measuring the Dielectric Constant of Water
The dielectric constant of water isn't a fixed value; it's dependent on several factors, most notably temperature and frequency. Various methods exist for measuring this crucial property, each with its own advantages and limitations.
Time-Domain Reflectometry (TDR)
TDR is a common technique used to measure the dielectric constant of materials, including water. It involves sending a short electrical pulse down a transmission line and measuring the reflections at the interface between the transmission line and the material. The time it takes for the pulse to reflect back provides information about the material's dielectric properties. This method is particularly suitable for measuring the dielectric constant of liquids like water.
Capacitance Measurements
Another widely used method involves measuring the capacitance of a capacitor with water as the dielectric material. The capacitance of a capacitor is directly proportional to the dielectric constant of the material between its plates. By carefully measuring the capacitance, one can determine the dielectric constant of water. This method is relatively straightforward but requires careful calibration and consideration of factors like electrode geometry and temperature.
Factors Affecting the Dielectric Constant of Water
The dielectric constant of water isn't a static number; it varies depending on a range of conditions:
Temperature Dependence
The dielectric constant of water decreases significantly as temperature increases. This is because higher temperatures lead to increased molecular motion, disrupting the alignment of water molecules in an electric field. The reduction in alignment weakens the ability of water to screen the electric field, thereby lowering the dielectric constant. This temperature dependence is non-linear and requires precise temperature control during measurements.
Frequency Dependence
The dielectric constant of water also exhibits frequency dependence. At low frequencies, water molecules have ample time to align themselves with the changing electric field, resulting in a high dielectric constant. As the frequency increases, the water molecules can't keep up with the rapid changes in the field, leading to a decrease in the dielectric constant. This frequency dependence is particularly noticeable at higher frequencies, such as those used in microwave and radio wave applications.
Pressure Dependence
Pressure also influences the dielectric constant of water, albeit to a lesser extent than temperature or frequency. Increased pressure generally leads to a slight increase in the dielectric constant, due to changes in molecular density and interactions.
Salinity and Ionic Strength
The presence of dissolved ions, such as salts in seawater, significantly affects the dielectric constant of water. Ions interfere with the alignment of water molecules, reducing the effective dielectric constant. The higher the ionic strength (concentration of dissolved ions), the lower the dielectric constant will be. This is particularly important in applications related to oceans and biological systems.
The Significance of Water's High Dielectric Constant
The exceptionally high dielectric constant of water is crucial for numerous phenomena and applications:
Biological Systems
The high dielectric constant of water plays a vital role in biological systems. It significantly influences the interactions between charged molecules, such as proteins and DNA. The dielectric constant helps screen the electrostatic interactions between these charged molecules, preventing them from aggregating and allowing them to maintain their functional structures and participate in vital biological processes.
Chemical Reactions
Water's high dielectric constant enhances the solubility of ionic compounds. Its ability to screen the electrostatic interactions between ions makes it an excellent solvent for many salts and other ionic substances, facilitating chemical reactions in aqueous solutions. Many crucial biochemical reactions occur in water, reliant on this unique solvation characteristic.
Electrochemistry
The dielectric constant of water is essential in electrochemistry, affecting the behavior of electrolytes and the performance of electrochemical devices. It influences the ionic conductivity of solutions and the double-layer capacitance at electrode surfaces, which are central to battery operation and corrosion processes.
Material Science
The dielectric constant of water is a crucial factor in material science, particularly in the development of new materials with specific electrical properties. Understanding how water interacts with various materials helps in designing materials for applications in sensors, capacitors, and other electronic devices.
Dielectric Constant: A Complex, Variable Property
In conclusion, the dielectric constant of water is not a simple constant but a dynamic property influenced by various factors, including temperature, frequency, pressure, and ionic strength. Its exceptionally high value is a direct consequence of water's unique molecular structure and polarity, and this value has profound implications across diverse fields. From the intricate workings of biological systems to the design of advanced electronic devices, understanding and accurately measuring the dielectric constant of water remains crucial for scientific progress and technological innovation. Further research into the precise behavior of water's dielectric constant under different conditions will continue to illuminate its significance in various scientific disciplines. Future studies may focus on developing more accurate measurement techniques and exploring the influence of other factors, such as the presence of dissolved gases or nanoparticles. This deeper understanding will undoubtedly lead to advancements in numerous fields, furthering our comprehension of the world around us.
Latest Posts
Latest Posts
-
Dna Biology And Technology Dna And Rna Structure
Apr 04, 2025
-
Narcotics Act On The Central Nervous System By Producing A
Apr 04, 2025
-
Two Plants Heteroygous For Pod Color Are Crossed Refer To
Apr 04, 2025
-
What Two Main Products Result From Photosynthesis
Apr 04, 2025
-
Identify What A Coffee Cup Calorimeter Measures
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Dielectric Constant Of Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
