What Is The Electron Configuration Of Beryllium
Muz Play
Mar 31, 2025 · 6 min read
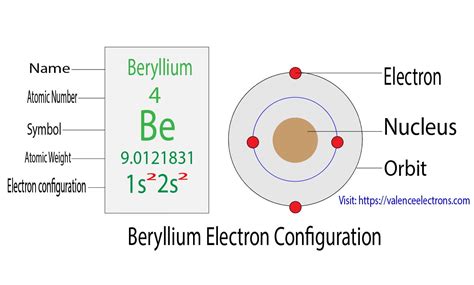
Table of Contents
What is the Electron Configuration of Beryllium? A Deep Dive into Atomic Structure
Beryllium, a fascinating element with a unique set of properties, holds a special place in the periodic table. Understanding its electron configuration is key to unlocking its chemical behavior and its applications in various fields. This comprehensive guide will delve into the electron configuration of beryllium, explaining its derivation, significance, and implications for its reactivity and applications.
Understanding Electron Configuration
Before diving into the specifics of beryllium, let's establish a fundamental understanding of electron configuration. An electron configuration describes how electrons are distributed among the various energy levels and sublevels within an atom. It's a shorthand notation that dictates an atom's electronic structure, a crucial determinant of its chemical properties. This distribution follows specific rules dictated by quantum mechanics, primarily the Aufbau principle, the Pauli exclusion principle, and Hund's rule.
-
The Aufbau Principle: Electrons fill the lowest energy levels first. This is like filling a container from the bottom up; you wouldn't start filling the top before the bottom is full.
-
The Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons, and these two electrons must have opposite spins. Think of it as an apartment with only two rooms; each room can house one tenant with a specific "spin" (let's say an assigned color).
-
Hund's Rule: When filling orbitals of equal energy (degenerate orbitals), electrons will individually occupy each orbital before pairing up in any one orbital. This is like having multiple empty chairs around a table; you'll sit in a separate chair before doubling up.
Determining the Electron Configuration of Beryllium (Be)
Beryllium (Be) is an element with an atomic number of 4. This means a neutral beryllium atom contains 4 protons in its nucleus and 4 electrons surrounding the nucleus. To determine its electron configuration, we follow the Aufbau principle.
The electron subshells are filled in the order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on. Each subshell has a specific capacity for electrons:
- s subshell: Holds a maximum of 2 electrons.
- p subshell: Holds a maximum of 6 electrons.
- d subshell: Holds a maximum of 10 electrons.
- f subshell: Holds a maximum of 14 electrons.
Therefore, following the Aufbau principle, the electron configuration of beryllium is:
1s² 2s²
This notation indicates that:
- Two electrons occupy the 1s orbital (the lowest energy level).
- Two electrons occupy the 2s orbital (the next lowest energy level).
Visualizing Beryllium's Electron Configuration
It's helpful to visualize the electron configuration. We can represent it using orbital diagrams:
1s: ↑↓
2s: ↑↓
Each arrow represents an electron, and the up and down arrows represent opposite spins. As you can see, both the 1s and 2s orbitals are completely filled.
The Significance of Beryllium's Electron Configuration
The electron configuration of beryllium (1s² 2s²) profoundly influences its chemical properties and behavior:
-
Reactivity: Beryllium has two valence electrons (electrons in the outermost shell). These valence electrons are relatively loosely bound and readily participate in chemical bonding. However, compared to other alkali earth metals, beryllium’s small size and high ionization energy make it less reactive. It readily forms covalent bonds instead of simply losing its two valence electrons to form ionic bonds.
-
Oxidation State: Due to its two valence electrons, beryllium predominantly exhibits a +2 oxidation state. This means it tends to lose two electrons to achieve a stable electron configuration similar to helium (1s²).
-
Chemical Bonding: Beryllium's tendency to form covalent bonds arises from its high electronegativity. Covalent bonds involve the sharing of electrons between atoms, rather than the complete transfer of electrons as in ionic bonding.
-
Formation of Compounds: The electron configuration dictates the types of compounds beryllium can form. For instance, beryllium readily reacts with halogens (like fluorine and chlorine) to form beryllium halides (BeF₂, BeCl₂), showcasing its covalent bonding behavior. It also forms stable compounds with oxygen, such as beryllium oxide (BeO).
Applications of Beryllium and its Relationship to Electron Configuration
Beryllium's unique properties, directly linked to its electron configuration, lead to its use in several high-tech applications:
-
Aerospace: Beryllium's high strength-to-weight ratio and stiffness make it ideal for lightweight aerospace components. This is crucial for aircraft and spacecraft where weight reduction translates to increased fuel efficiency and payload capacity.
-
Nuclear Applications: Beryllium is used as a neutron reflector and moderator in nuclear reactors. Its ability to moderate neutron speed facilitates the nuclear chain reaction.
-
Electronics: Its high thermal conductivity and electrical conductivity make beryllium useful in electronic devices and heat sinks, crucial in managing heat dissipation in high-performance electronics.
-
X-ray Windows: Beryllium's low atomic number means it's relatively transparent to X-rays. This property makes it valuable in X-ray windows for applications requiring high X-ray transmission.
Comparing Beryllium's Electron Configuration to Other Elements
Understanding beryllium's electron configuration allows us to compare it with other elements and understand their similarities and differences in chemical behavior:
-
Helium (He): Helium has an electron configuration of 1s². Like beryllium, its valence shell is full, making it chemically inert (unreactive). However, beryllium, with an additional electron shell, exhibits far more chemical reactivity than helium.
-
Lithium (Li): Lithium (1s² 2s¹) has only one valence electron, making it highly reactive and readily forming +1 ions. Its reactivity is significantly higher than that of beryllium, which has two valence electrons.
-
Boron (B): Boron (1s² 2s² 2p¹) has three valence electrons, leading to a different bonding pattern compared to beryllium. Boron forms covalent bonds but with a different geometry and reactivity compared to beryllium.
Beyond the Basics: Excited States and Ionization
While the 1s² 2s² configuration represents beryllium's ground state (lowest energy level), it can be excited to higher energy states by absorbing energy. This excitation promotes an electron to a higher energy level, resulting in a different electron configuration.
Ionization, the process of removing an electron from an atom, also affects the electron configuration. For instance, removing one electron from beryllium would leave it with a +1 charge and an electron configuration of 1s² 2s¹. Removing two electrons would produce a +2 ion with the configuration 1s², which is isoelectronic (having the same number of electrons) with helium.
Conclusion: The Importance of Understanding Electron Configuration
The electron configuration of beryllium (1s² 2s²) is fundamental to understanding its properties, reactivity, and applications. This seemingly simple notation encapsulates a wealth of information about the atom's electronic structure, which dictates its chemical behavior and its significance in diverse technological applications. By understanding the principles behind electron configuration, we gain valuable insight into the behavior of beryllium and numerous other elements, enabling us to predict their chemical interactions and explore their potential uses. Its relatively simple configuration provides a solid foundation for further studies in chemistry and material science.
Latest Posts
Latest Posts
-
Which Of The Following Are Examples Of Homologous Structures
Apr 03, 2025
-
Which Plasma Component Is Not Present In Serum
Apr 03, 2025
-
Physical Features Map Of South America
Apr 03, 2025
-
What Is The Basic Idea Behind Disengagement Theory
Apr 03, 2025
-
Is Bromine A Good Leaving Group
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is The Electron Configuration Of Beryllium . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
