What Is The Number Of Protons For Sulfur
Muz Play
Apr 04, 2025 · 5 min read
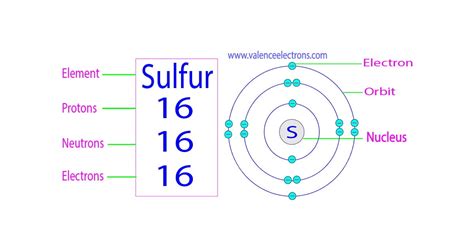
Table of Contents
- What Is The Number Of Protons For Sulfur
- Table of Contents
- What is the Number of Protons for Sulfur? A Deep Dive into Atomic Structure and Sulfur's Properties
- Understanding Atomic Number and Protons
- Sulfur's Atomic Number: 16
- The Significance of Sulfur's 16 Protons
- 1. Chemical Bonding and Reactivity:
- 2. Isotopic Variations:
- 3. Physical and Chemical Properties:
- Sulfur's Role in Various Fields
- 1. Industrial Applications:
- 2. Biological Significance:
- 3. Environmental Concerns:
- Conclusion: The Importance of Atomic Number in Defining Sulfur
- Latest Posts
- Latest Posts
- Related Post
What is the Number of Protons for Sulfur? A Deep Dive into Atomic Structure and Sulfur's Properties
Sulfur, a vibrant yellow nonmetal, plays a crucial role in various biological and industrial processes. Understanding its atomic structure, particularly its number of protons, is fundamental to comprehending its chemical behavior and applications. This comprehensive article delves into the atomic number of sulfur, exploring its implications for chemical bonding, isotopic variations, and the element's unique properties. We will also examine how this fundamental atomic characteristic influences sulfur's role in various fields.
Understanding Atomic Number and Protons
The atomic number of an element is defined as the number of protons found in the nucleus of each atom of that element. This number is unique to each element and is what differentiates one element from another on the periodic table. Protons, along with neutrons, constitute the nucleus of an atom, while electrons orbit the nucleus in electron shells. Protons carry a positive charge, while electrons carry a negative charge. The number of protons determines the element's chemical properties and its position on the periodic table. A neutral atom has an equal number of protons and electrons.
Sulfur's Atomic Number: 16
The atomic number of sulfur (S) is 16. This signifies that every sulfur atom contains 16 protons in its nucleus. This fundamental fact dictates all the other properties and behaviors of sulfur. This number is consistently found across all isotopes of sulfur (variations in the number of neutrons), ensuring its consistent chemical identity. The unique configuration of 16 protons and its corresponding electron arrangement in its shells defines sulfur's reactivity and bonding capabilities.
The Significance of Sulfur's 16 Protons
The presence of 16 protons in the sulfur atom has profound implications for its:
1. Chemical Bonding and Reactivity:
Sulfur's 16 protons result in an electron configuration of 1s²2s²2p⁶3s²3p⁴. This electronic structure means sulfur has six valence electrons—electrons in the outermost shell. These valence electrons are crucial for forming chemical bonds. Sulfur frequently achieves a stable octet (eight electrons in its outermost shell) through covalent bonding, sharing electrons with other atoms. This tendency to form covalent bonds explains sulfur's presence in numerous compounds, including sulfides, sulfates, and organic sulfur compounds. The electronegativity of sulfur (its ability to attract electrons in a chemical bond) is relatively high, contributing to the polar nature of many sulfur-containing compounds.
2. Isotopic Variations:
While the number of protons remains constant at 16, the number of neutrons in the sulfur nucleus can vary. These variations lead to different isotopes of sulfur. The most common isotopes are ³²S, ³³S, ³⁴S, and ³⁶S. Isotopes have the same number of protons and therefore the same chemical properties, but their different neutron numbers lead to variations in their mass and stability. The relative abundance of these isotopes varies slightly in different geological locations and is sometimes used in environmental and geological studies.
3. Physical and Chemical Properties:
The 16 protons in sulfur's nucleus contribute directly to its physical and chemical properties. These properties include:
-
Yellow color: Sulfur's characteristic yellow color arises from the interaction of light with its electrons. This interaction is a direct consequence of its atomic structure, specifically the arrangement of electrons determined by the 16 protons.
-
Low melting and boiling points: Compared to other nonmetals, sulfur has relatively low melting and boiling points. This is related to the strength of the bonds between sulfur atoms, which is influenced by the electronic structure determined by the 16 protons.
-
Multiple allotropic forms: Sulfur exists in various allotropic forms (different structural modifications of the same element). This property is again tied to the electronic configuration, enabling different bonding arrangements and crystal structures. The most common allotrope is rhombic sulfur.
-
Oxidation states: Sulfur can exist in a variety of oxidation states, ranging from -2 to +6. This versatility is a direct result of its six valence electrons and its ability to gain or lose electrons in chemical reactions.
Sulfur's Role in Various Fields
Sulfur's unique properties, stemming directly from its 16 protons, contribute to its significance across various fields:
1. Industrial Applications:
-
Sulfuric Acid Production: Sulfuric acid (H₂SO₄), a crucial industrial chemical, is produced primarily from sulfur. Its vast applications span fertilizer production, metal refining, and numerous other industrial processes. The efficiency and effectiveness of these processes are directly linked to the chemical properties of sulfur.
-
Rubber Vulcanization: Sulfur plays a vital role in the vulcanization of rubber, a process that strengthens and improves the elasticity of rubber. This industrial application is critical for various products, from tires to other rubber goods.
-
Detergents and Pharmaceuticals: Sulfur and sulfur compounds are used in the production of detergents, fungicides, and certain pharmaceuticals. The chemical reactivity and bonding properties of sulfur are essential for the effectiveness of these products.
2. Biological Significance:
-
Amino Acids: Sulfur is an essential component of several amino acids, including methionine and cysteine, which are crucial for protein synthesis and various biological processes. The chemical properties of sulfur in these amino acids contribute to protein folding and enzyme activity.
-
Enzymes: Sulfur-containing enzymes play essential roles in various metabolic pathways, catalysing crucial biological reactions. The presence of sulfur in these enzymes enables their specific catalytic functions.
-
Vitamins: Some vitamins contain sulfur, underlining its significance in maintaining human health.
3. Environmental Concerns:
-
Air Pollution: Sulfur dioxide (SO₂) is a significant air pollutant, contributing to acid rain and respiratory problems. The understanding of sulfur's chemical behavior is crucial in developing strategies to mitigate sulfur dioxide emissions.
-
Geochemical Cycles: Sulfur participates in various geochemical cycles, impacting both terrestrial and marine environments. Studying sulfur's role in these cycles contributes to environmental monitoring and management.
Conclusion: The Importance of Atomic Number in Defining Sulfur
The number of protons in a sulfur atom—16—is not just a numerical value; it is the cornerstone defining its identity, chemical behavior, and extensive applications. From its vibrant yellow color to its crucial roles in industrial processes and biological systems, the 16 protons fundamentally dictate sulfur's unique characteristics. Understanding this fundamental aspect of sulfur's atomic structure enables a deeper appreciation for its role in the world around us. Further research into sulfur's properties, driven by our knowledge of its atomic number, continues to uncover new applications and deepen our understanding of its multifaceted significance. The simple number 16 opens up a vast and complex world of chemistry and biological function.
Latest Posts
Latest Posts
-
Type Of Molecule With An Uneven Distribution Of Electrons
Apr 06, 2025
-
Differential Rate Law Vs Integrated Rate Law
Apr 06, 2025
-
How Do You Test An Equation For Symmetry
Apr 06, 2025
-
Circuit Diagram With 3 Resistors In Series
Apr 06, 2025
-
Art Of The 1920s In America
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Is The Number Of Protons For Sulfur . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
