What Is The Solute In Salt Water
Muz Play
Apr 03, 2025 · 5 min read
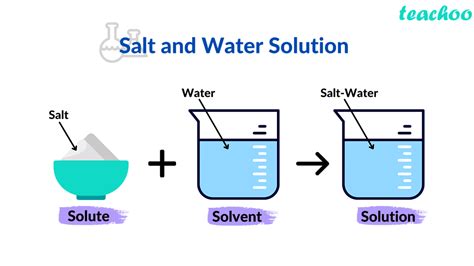
Table of Contents
What is the Solute in Salt Water? A Deep Dive into Solutions and Solutes
Understanding the composition of saltwater is fundamental to various fields, from oceanography and chemistry to cooking and even medicine. A key concept in understanding this composition is the distinction between a solute and a solvent. This article will explore what constitutes a solute, specifically in the context of saltwater, and delve into the broader implications of this concept.
Defining Solute and Solvent
Before we dive into the specifics of saltwater, let's establish clear definitions. In a solution, we have two primary components:
-
Solute: This is the substance that dissolves in a solvent. It's typically present in a smaller amount compared to the solvent. Think of it as the substance that gets broken down and distributed throughout the solution.
-
Solvent: This is the substance that dissolves the solute. It's usually present in a larger amount. It's the medium in which the solute disperses.
The combination of solute and solvent creates a homogeneous mixture, meaning the composition is uniform throughout.
Saltwater: A Case Study
Now, let's apply this to saltwater. Saltwater, as its name suggests, is a solution of salt (primarily sodium chloride, NaCl) dissolved in water (H₂O). Therefore, to answer the question directly:
The solute in saltwater is salt (primarily sodium chloride, NaCl).
However, it's crucial to understand that "salt" in this context isn't just one single type of molecule. Ocean water, for instance, contains a complex mixture of salts. While sodium chloride is the dominant component, other salts like magnesium chloride (MgCl₂), potassium chloride (KCl), and calcium sulfate (CaSO₄) are also present in significant quantities. These contribute to the overall salinity and properties of the saltwater.
Understanding the Dissolution Process
The dissolving of salt in water is a fascinating process driven by the polar nature of water molecules. Water molecules (H₂O) are polar, meaning they have a slightly positive end (hydrogen atoms) and a slightly negative end (oxygen atom). Salt, on the other hand, is an ionic compound, composed of positively charged sodium ions (Na⁺) and negatively charged chloride ions (Cl⁻).
When salt is added to water, the polar water molecules surround the ions. The slightly negative oxygen ends of the water molecules attract the positive sodium ions, while the slightly positive hydrogen ends attract the negative chloride ions. This process, known as hydration, effectively weakens the electrostatic forces holding the sodium and chloride ions together in the salt crystal. The ions become surrounded by water molecules, breaking away from the crystal lattice and dispersing uniformly throughout the water, forming a homogeneous solution.
Factors Affecting Solubility
Several factors influence the solubility of salt in water, including:
-
Temperature: Generally, the solubility of most salts in water increases with temperature. Warmer water can dissolve more salt than colder water. This is why you might find it easier to dissolve salt in hot water compared to cold water.
-
Pressure: While pressure has a less significant effect on the solubility of salts in water compared to gases, it does play a role, especially at high pressures.
-
Presence of other solutes: The presence of other dissolved substances in the water can affect the solubility of salt. Certain ions might interact with the salt ions, reducing or increasing their solubility.
Beyond Sodium Chloride: The Complexity of Seawater
Seawater is far more complex than a simple solution of sodium chloride in water. It's a dynamic mixture containing a vast array of dissolved substances, including:
-
Major ions: These include sodium (Na⁺), chloride (Cl⁻), magnesium (Mg²⁺), sulfate (SO₄²⁻), calcium (Ca²⁺), and potassium (K⁺). These ions contribute significantly to the salinity and overall properties of seawater.
-
Minor ions: These are present in smaller concentrations but still play important roles in various marine processes. Examples include bromide (Br⁻), strontium (Sr²⁺), and borate (BO₃³⁻).
-
Nutrients: Seawater contains essential nutrients like nitrates (NO₃⁻), phosphates (PO₄³⁻), and silicates (SiO₄⁴⁻) that are crucial for marine life.
-
Dissolved gases: Gases like oxygen (O₂), carbon dioxide (CO₂), and nitrogen (N₂) are dissolved in seawater, supporting marine life and influencing ocean chemistry.
-
Organic matter: Seawater contains dissolved organic matter, originating from decaying organisms and other biological processes. This organic matter significantly impacts the water's properties and the marine ecosystem.
Salinity and its Implications
Salinity is a crucial characteristic of seawater, reflecting the total concentration of dissolved salts. It's usually expressed in parts per thousand (ppt) or practical salinity units (PSU). Salinity varies significantly depending on location and factors like evaporation, precipitation, and river runoff. Different levels of salinity affect marine life, influencing the distribution and abundance of various species. Some organisms thrive in high-salinity environments, while others are adapted to lower salinity conditions.
Practical Applications and Significance
Understanding the solute in saltwater has profound implications across various fields:
-
Oceanography: Understanding the composition of seawater is crucial for studying ocean currents, marine ecosystems, and the impact of climate change on the oceans. Oceanographers routinely measure salinity and other parameters to monitor ocean health and predict future changes.
-
Chemistry: Saltwater serves as an excellent example to demonstrate fundamental chemical concepts like solubility, solutions, and ionic interactions. Studying saltwater helps to illustrate the principles governing the behavior of solutions and the properties of various ions.
-
Aquaculture: Maintaining the appropriate salinity levels is crucial for successful aquaculture operations. Different marine species have specific salinity requirements, and deviations from these levels can negatively impact their growth and survival.
-
Desalination: Desalination processes, which aim to remove salt from seawater to produce fresh water, rely on a thorough understanding of the properties of saltwater and the interactions of its solutes.
Conclusion: More Than Just Salt and Water
While the primary solute in saltwater is indeed sodium chloride, it's crucial to recognize the complexity of this seemingly simple solution. Seawater is a dynamic and multifaceted mixture containing a vast array of dissolved substances that influence its properties and play critical roles in various ecological and geological processes. A deep understanding of the solutes and their interactions within this solution is vital for various scientific disciplines and practical applications. From studying the vastness of our oceans to ensuring the success of aquaculture practices, understanding the composition of saltwater remains a crucial endeavor.
Latest Posts
Latest Posts
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
-
What Is The Net Change In Math
Apr 04, 2025
-
Why Cant The Subscripts Be Changed In A Chemical Equation
Apr 04, 2025
-
How Many Bonds Does Boron Form
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Solute In Salt Water . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
