What Makes Something A Strong Acid
Muz Play
Mar 31, 2025 · 7 min read
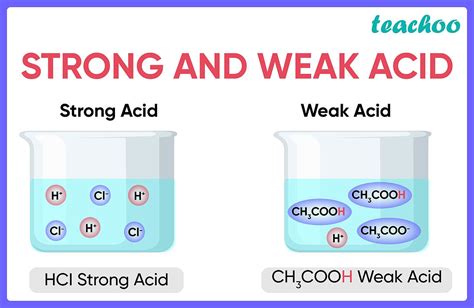
Table of Contents
- What Makes Something A Strong Acid
- Table of Contents
- What Makes Something a Strong Acid? Understanding Acidity and Dissociation
- The Nature of Acids: A Brief Overview
- The Brønsted-Lowry Definition: The Heart of Acidity
- Strong Acids vs. Weak Acids: A Critical Distinction
- The Equilibrium Constant: Ka and its Significance
- Factors Determining Acid Strength
- 1. Bond Strength: The Role of Polarity and Electronegativity
- 2. Stability of the Conjugate Base: A Crucial Factor
- 3. Solvent Effects: The Influence of the Medium
- Common Examples of Strong Acids
- Measuring Acidity: pH and its Relevance
- Safety Precautions When Handling Strong Acids
- Conclusion: Understanding the Power of Strong Acids
- Latest Posts
- Latest Posts
- Related Post
What Makes Something a Strong Acid? Understanding Acidity and Dissociation
Acids are ubiquitous in our world, playing crucial roles in everything from the digestive processes in our bodies to industrial manufacturing. Understanding what constitutes a strong acid, as opposed to a weak one, is fundamental to chemistry and numerous applications. This article delves deep into the properties that define strong acids, exploring the underlying concepts of dissociation, pH, and the factors influencing acid strength.
The Nature of Acids: A Brief Overview
Before diving into the specifics of strong acids, let's establish a baseline understanding of acids in general. Acids are substances that donate protons (H⁺ ions) when dissolved in a solution, typically water. This proton donation is the key characteristic that defines acidic behavior. The ability of an acid to donate protons dictates its strength. This ability is related to the stability of the conjugate base that forms after proton donation.
The Brønsted-Lowry Definition: The Heart of Acidity
The Brønsted-Lowry definition of acids is particularly useful for understanding acid strength. It defines an acid as a proton donor, and a base as a proton acceptor. When an acid dissolves in water, it donates a proton to a water molecule, forming a hydronium ion (H₃O⁺) and the conjugate base of the acid. This reaction is an equilibrium process, meaning it proceeds in both directions.
Example: The dissociation of hydrochloric acid (HCl) in water:
HCl(aq) + H₂O(l) ⇌ H₃O⁺(aq) + Cl⁻(aq)
In this reaction, HCl acts as a Brønsted-Lowry acid, donating a proton to water, which acts as a Brønsted-Lowry base. The resulting hydronium ion is indicative of the acidic nature of the solution.
Strong Acids vs. Weak Acids: A Critical Distinction
The critical difference between strong and weak acids lies in the extent of their dissociation in water. Strong acids essentially completely dissociate into their constituent ions when dissolved in water. This means that nearly all the acid molecules donate their protons, leading to a high concentration of hydronium ions (H₃O⁺).
Weak acids, on the other hand, only partially dissociate. A significant portion of the weak acid molecules remain undissociated in solution, resulting in a lower concentration of hydronium ions compared to a strong acid of the same concentration.
The Equilibrium Constant: Ka and its Significance
The extent of dissociation of a weak acid is quantified by its acid dissociation constant (Ka). Ka is the equilibrium constant for the dissociation reaction of an acid in water. A larger Ka value indicates a greater extent of dissociation and thus a stronger acid.
Example: Consider the dissociation of a weak acid, HA:
HA(aq) + H₂O(l) ⇌ H₃O⁺(aq) + A⁻(aq)
The Ka expression is:
Ka = [H₃O⁺][A⁻] / [HA]
For strong acids, Ka is extremely large, essentially approaching infinity, making it impractical to use Ka directly to compare strong acids.
Factors Determining Acid Strength
Several factors contribute to the strength of an acid:
1. Bond Strength: The Role of Polarity and Electronegativity
The strength of the bond between the hydrogen atom and the rest of the acid molecule plays a significant role. A weaker bond facilitates easier proton donation, leading to a stronger acid. This bond strength is influenced by factors such as the electronegativity of the atom bonded to hydrogen. A more electronegative atom pulls electron density away from the hydrogen, weakening the bond and making it easier to donate the proton.
For instance, the electronegativity difference between hydrogen and chlorine in HCl is quite high, leading to a weaker H-Cl bond and thus the strong acidic nature of HCl.
2. Stability of the Conjugate Base: A Crucial Factor
The stability of the conjugate base formed after proton donation is another crucial determinant of acid strength. A more stable conjugate base means the acid is more likely to donate its proton, resulting in a stronger acid. Stability is often linked to factors like resonance, inductive effects, and size.
-
Resonance: If the conjugate base can delocalize the negative charge through resonance, it becomes more stable, making the corresponding acid stronger. This is evident in carboxylic acids.
-
Inductive Effects: Electron-withdrawing groups attached to the conjugate base can stabilize the negative charge by pulling electron density away from it. This increases the stability of the conjugate base and makes the acid stronger.
-
Size: Larger anions tend to be more stable because the negative charge is spread over a larger volume. This is particularly noticeable in the hydrohalic acids (HF, HCl, HBr, HI), where the acid strength increases down the group due to the increasing size of the halide ion.
3. Solvent Effects: The Influence of the Medium
The solvent in which the acid is dissolved also affects its strength. The properties of the solvent, such as its polarity and ability to stabilize the ions formed during dissociation, influence the equilibrium of the dissociation reaction. In different solvents, the same acid might exhibit different levels of strength. Water is the most common solvent used to study acids, but other solvents can affect the apparent strength.
Common Examples of Strong Acids
Several acids are universally recognized as strong acids due to their near-complete dissociation in water. These include:
- Hydrochloric acid (HCl): A highly corrosive acid used in various industrial processes and found in gastric juices.
- Hydrobromic acid (HBr): Another strong corrosive acid, similar in properties to HCl.
- Hydroiodic acid (HI): Similar to HBr, it's a very strong acid.
- Nitric acid (HNO₃): A strong oxidizing acid widely used in the production of fertilizers and explosives.
- Sulfuric acid (H₂SO₄): A highly corrosive and viscous acid, a crucial industrial chemical used extensively in various processes, including the production of fertilizers and detergents. It's a diprotic acid, meaning it can donate two protons. However, the second proton dissociation is weaker than the first.
- Perchloric acid (HClO₄): A very strong acid, often considered the strongest common acid in aqueous solution.
It's important to note that the strength of an acid is a relative term, always considered within the context of a particular solvent (usually water).
Measuring Acidity: pH and its Relevance
The acidity of a solution is commonly expressed using the pH scale, which is a logarithmic scale that ranges from 0 to 14. A pH of 7 is considered neutral, a pH less than 7 is acidic, and a pH greater than 7 is basic (alkaline). The pH is directly related to the concentration of hydronium ions (H₃O⁺) in the solution.
pH = -log₁₀[H₃O⁺]
Strong acids, due to their complete dissociation, have significantly lower pH values than weak acids at the same concentration. For example, a 0.1 M solution of a strong acid will have a pH of 1, while a 0.1 M solution of a weak acid will have a higher pH value, reflecting the lower concentration of hydronium ions.
Safety Precautions When Handling Strong Acids
Strong acids are highly corrosive and can cause severe burns to skin and eyes. It's imperative to handle them with extreme caution and always follow appropriate safety measures:
- Wear appropriate personal protective equipment (PPE): This includes safety goggles, gloves, lab coats, and possibly respirators, depending on the concentration and application.
- Work in a well-ventilated area: Some strong acids release hazardous fumes.
- Always add acid to water, never water to acid: Adding water to acid can cause a violent exothermic reaction.
- Neutralize spills immediately using appropriate procedures: Consult the safety data sheet (SDS) for the specific acid.
- Dispose of strong acids properly according to local regulations: Never pour them down the drain without proper neutralization.
Conclusion: Understanding the Power of Strong Acids
The strength of an acid is a crucial concept in chemistry with significant implications in various fields. Understanding the factors that govern acid strength, including bond strength, conjugate base stability, and solvent effects, is essential for predicting and controlling the reactivity of acids. The complete dissociation of strong acids, leading to high concentrations of hydronium ions and low pH values, makes them powerful reagents with diverse applications. However, their corrosive nature mandates careful and safe handling procedures to prevent accidents and injuries. This comprehensive overview provides a strong foundation for anyone seeking a deeper understanding of the fascinating world of acids.
Latest Posts
Latest Posts
-
How Does The Predator Prey Relationship Affect A Population
Apr 03, 2025
-
The Axons Of Parasympathetic Postganglionic Neurons Are
Apr 03, 2025
-
Is Hydrogen A Metal Nonmetal Or Metalloid
Apr 03, 2025
-
How Does Molecular Biology Support The Theory Of Evolution
Apr 03, 2025
-
Not A And Not B Truth Table
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Makes Something A Strong Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
