What Would The Potential Of A Standard Hydrogen Electrode
Muz Play
Apr 02, 2025 · 6 min read
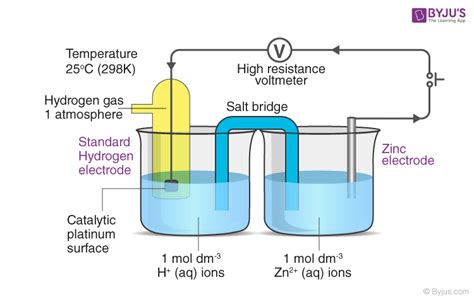
Table of Contents
The Untapped Potential of the Standard Hydrogen Electrode (SHE): A Deep Dive
The Standard Hydrogen Electrode (SHE), often considered the cornerstone of electrochemistry, is far more than a simple reference electrode. Its potential extends far beyond its foundational role in determining the reduction potentials of other half-cells. This seemingly simple device holds immense potential for advancements in various scientific fields, from energy storage and fuel cell technology to environmental monitoring and analytical chemistry. This article delves deep into the SHE's capabilities, exploring its applications, limitations, and the ongoing research pushing its boundaries.
Understanding the SHE: More Than Just a Reference
The SHE, as its name suggests, is a standard electrode against which the reduction potentials of all other half-cells are measured. It consists of a platinum electrode immersed in an acidic solution with a hydrogen gas pressure of 1 atm. The platinum acts as an inert catalyst, facilitating the reversible reaction:
2H⁺(aq) + 2e⁻ ⇌ H₂(g)
This simple reaction, under standard conditions (298 K, 1 atm H₂ pressure, 1 M H⁺ concentration), defines a potential of exactly 0 volts. This zero-point allows for the systematic measurement of reduction potentials of other electrodes relative to the SHE. This relative measurement, crucially, is independent of the specific experimental setup, providing a consistent and universally understood standard.
The SHE's Crucial Role in Electrochemistry
The SHE's importance in electrochemistry cannot be overstated. It forms the bedrock of the electrochemical series, a vital tool for predicting the spontaneity of redox reactions. By comparing the reduction potential of any half-cell to the SHE, we can determine whether a reaction will proceed spontaneously or require an external energy input. This capability is indispensable in various applications, including:
- Battery design and optimization: Understanding the relative reduction potentials of electrode materials is fundamental to designing efficient and high-capacity batteries.
- Corrosion prevention: The SHE aids in predicting corrosion rates by identifying materials susceptible to oxidation in a given environment.
- Electroplating and electrosynthesis: Controlling the reduction potentials allows for precise control over the deposition of metals and the synthesis of organic compounds.
Beyond the Basics: Exploring Advanced Applications of the SHE
While the SHE's role as a reference electrode is foundational, its potential extends far beyond this fundamental application. Ongoing research explores innovative ways to leverage the SHE's principles and functionalities in various fields.
1. Fuel Cell Technology: Harnessing Hydrogen's Power
Fuel cells, which directly convert chemical energy into electrical energy, rely heavily on the SHE's principles. The hydrogen oxidation reaction at the anode mirrors the reverse of the SHE reaction. Improving the efficiency and durability of fuel cells necessitates a deep understanding of the electrode kinetics involved, including the influence of factors such as catalyst materials, temperature, and pressure. The SHE serves as an invaluable tool in characterizing and optimizing fuel cell performance.
Optimizing Catalyst Efficiency: Research focuses on developing highly efficient and durable catalysts for the hydrogen oxidation reaction. The SHE provides the benchmark for evaluating the performance of these catalysts, helping to identify materials that minimize overpotential losses and maximize energy conversion efficiency.
Understanding Membrane Performance: The performance of proton exchange membranes (PEMs) in fuel cells is crucial for efficient proton transport. The SHE's potential can be used to investigate the ionic conductivity and permeability of these membranes under different operating conditions, leading to the design of more effective fuel cell components.
2. Environmental Monitoring: Detecting Pollutants and Assessing Water Quality
The SHE, combined with other electrochemical techniques, plays a significant role in environmental monitoring. Its use in sensors for detecting pollutants, like heavy metal ions or organic contaminants, is gaining traction. The principles underpinning these sensors rely on the specific electrochemical reactions of pollutants, often involving redox processes that are measured against the SHE's reference potential.
Heavy Metal Detection: Electrochemical sensors incorporating the SHE can be designed to detect the presence of heavy metal ions in water samples. These sensors often utilize a working electrode capable of selectively interacting with the target pollutant, with the SHE providing the stable reference potential needed for accurate measurement.
Organic Pollutant Analysis: Similarly, the SHE can be incorporated into sensors for detecting organic pollutants. The selective oxidation or reduction of these pollutants at a working electrode can be precisely quantified using the SHE as a stable reference, enabling sensitive and reliable environmental monitoring.
3. Analytical Chemistry: Precise and Sensitive Measurements
The SHE's precise and stable potential makes it an essential tool in various analytical chemistry techniques. It finds applications in techniques such as potentiometry, voltammetry, and amperometry. These techniques allow for the quantitative analysis of various substances, relying on the SHE to establish a reliable and reproducible reference potential.
Potentiometry: Potentiometric measurements rely on the difference in potential between a working electrode and the SHE to determine the concentration of a specific ion in solution. The SHE's stability is crucial for accurate measurements.
Voltammetry and Amperometry: These techniques involve applying a variable potential to a working electrode and measuring the resulting current. The SHE provides the stable reference potential needed for accurate measurements of redox reactions and the determination of various species.
Limitations and Advancements in SHE Technology
Despite its significance, the SHE presents certain limitations that researchers are actively addressing:
-
Practical Challenges: The SHE is not easily portable or readily used in field applications due to its reliance on a continuous flow of hydrogen gas and its sensitivity to oxygen contamination. This has led to the development of alternative reference electrodes, like the Silver/Silver Chloride (Ag/AgCl) electrode, which is more practical for everyday use. However, even these alternatives are ultimately calibrated against the SHE.
-
Oxygen Sensitivity: The SHE is highly sensitive to oxygen contamination, which can significantly alter its potential. Strict precautions are needed to exclude oxygen, impacting the ease of use in some applications.
-
Temperature Dependence: The potential of the SHE is slightly temperature-dependent. Accurate temperature control is therefore crucial for precise measurements.
Researchers are actively working on overcoming these limitations:
-
Miniaturization: Miniaturized versions of the SHE, suitable for microfluidic devices and portable applications, are being developed.
-
Improved Stability and Durability: Research aims to develop more robust and stable SHE designs that are less prone to oxygen contamination and drift.
-
Development of Alternative Reference Electrodes: While other reference electrodes exist, research continues into the development of highly stable and easily implemented reference electrodes that can serve as accurate replacements for the SHE in certain contexts.
The Future of the SHE: Unfolding Potential
The SHE, despite its simplicity, holds vast untapped potential. Its importance extends far beyond its fundamental role as a reference electrode. As research continues to refine its design, explore its applications in emerging technologies, and address its limitations, the SHE promises to play an increasingly significant role in diverse scientific and technological advancements. From improving fuel cell efficiency and developing more sensitive environmental monitoring tools to advancing analytical chemistry techniques, the SHE's influence on the future of science and technology is undeniable. The ongoing pursuit of understanding and optimizing the SHE’s functionality underscores its enduring importance and its continuing contribution to progress across multiple scientific disciplines. The seemingly simple standard hydrogen electrode is, in reality, a powerful tool with a wealth of potential yet to be fully realized.
Latest Posts
Latest Posts
-
Evaluating Functions Linear And Quadratic Or Cubic
Apr 03, 2025
-
What Are The Reactants In The Calvin Cycle
Apr 03, 2025
-
What Happened To Islam After The Death Of Muhammad
Apr 03, 2025
-
Which Of These Diagrams Is A Convex Mirror
Apr 03, 2025
-
Which State Of Matter Has A Definite Shape
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Would The Potential Of A Standard Hydrogen Electrode . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
